Key Takeaways
• Having a preoperative diagnosis of Mirizzi syndrome significantly lowers postoperative complications
• A majority of the time preoperative diagnosis is made by ERCP, and biliary stenting should be considered. This allows patients with more severe disease (type II through type V) to delay definitive cholecystectomy without increasing postoperative risk.
• For early Mirizzi syndrome (type I), laparoscopic cholecystectomy is a safe approach and can be performed by an acute care or hepatopancreatobiliary specialist.
Introduction
Mirizzi syndrome is uncommon with an incidence of 0.3% to 1.4% of all gallstone disease, yet it carries significant clinical importance due to its challenging operative management and frequent need for biliary reconstruction.
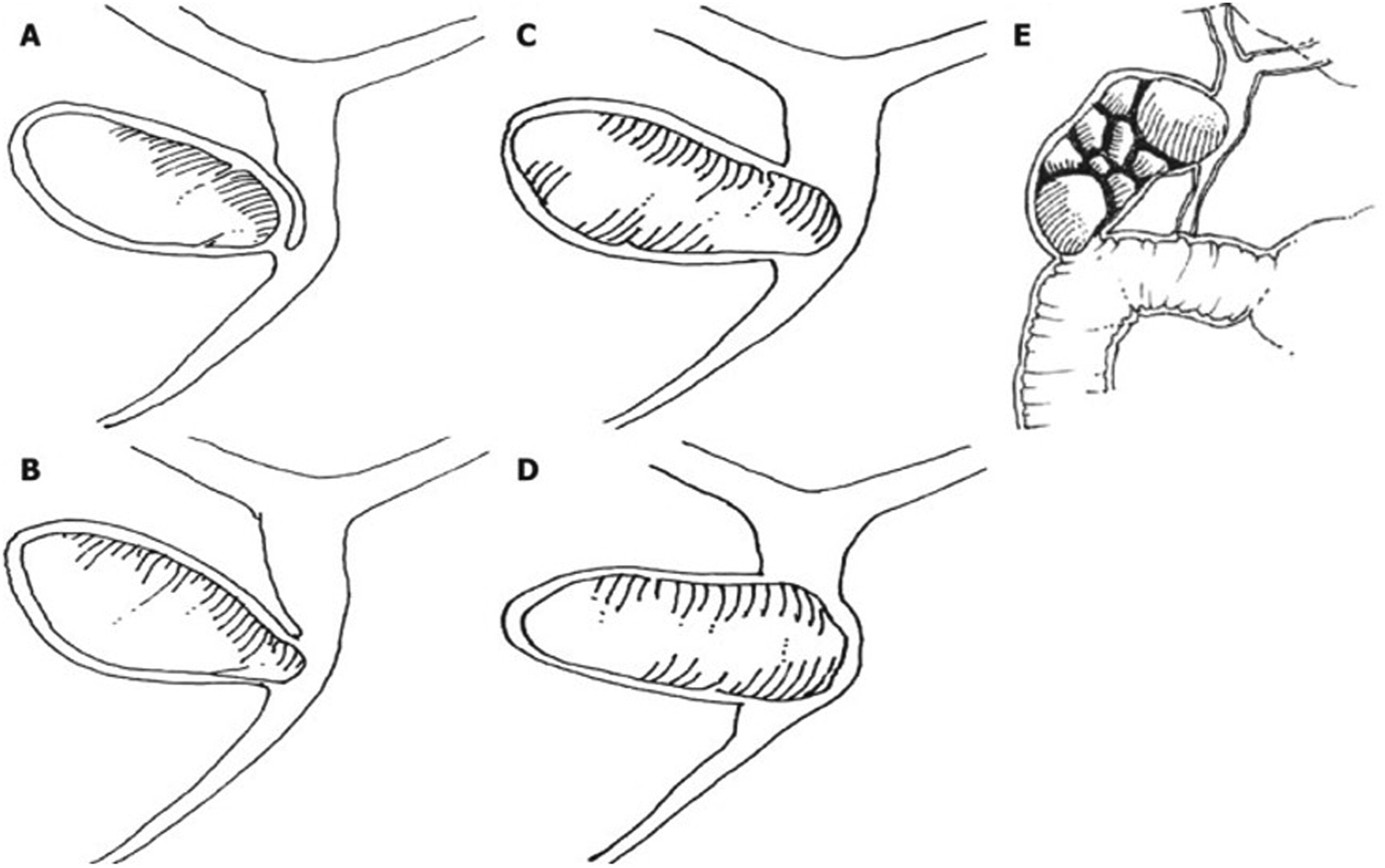
1 Mirizzi syndrome is defined as obstruction of the common bile duct or common hepatic duct by extrinsic compression from an impacted gallstone in the cystic duct or gallbladder infundibulum. The degree of inflammatory erosion and biliary involvement has been described by two major classification systems: the McSherry classification (1982) and the more widely used Csendes classification (1989), the latter dividing disease severity into five categories (types I–V).
2,3 See
Figure 1 illustrating this.
3 Increasing severity, particularly in types III–V, often necessitates complex biliary reconstruction such as Roux-en-Y hepaticojejunostomy, typically performed by hepatopancreatobiliary (HPB) specialists.
4Accurate preoperative diagnosis of Mirizzi syndrome aides in preoperative planning and is commonly diagnosed via endoscopic retrograde cholangiopancreatography (ERCP), although magnetic resonance cholangiopancreatography (MRCP) and right upper quadrant abdominal US (RUQ US) are useful diagnostic tools.
5 Diagnosis is essential, whether preoperatively or intraoperatively, in order to avoid bile duct injury and decrease morbidity and mortality.
3 Traditionally, laparoscopic approaches have been reserved for select type I cases; however, emerging evidence suggests that laparoscopic and even robotic approaches may be safe for some type II cases.
6–8 These approaches frequently involve laparoscopic bile duct exploration, often utilizing a transinfundibular approach to avoid choledochotomy.
6 Comparative studies have demonstrated equivalent postoperative outcomes between laparoscopic and open approaches for type I disease, while data for type II disease remain mixed. There has also been increasing evidence of using ERCP therapeutically. Some programs have used it for decompression to bridge to definitive surgery via stenting or even nasogastric drainage, while there exist case reports of ERCP and lithotripsy only.
9,10 Overall, standardized surgical guidelines are currently lacking, which continues to hinder the establishment of optimal operative strategies tailored to disease severity.
Therefore, the aim of this study was to characterize current diagnostic and operative practice patterns across 3 Los Angeles County hospitals, one of the largest urban public health systems in the United States, and to develop a consistent operative strategy aligned with disease severity. Specifically, we evaluated the diagnostic modalities used for Mirizzi syndrome and assessed whether the timing of diagnosis influenced postoperative outcomes. Additionally, given the increasing use of preoperative ERCP and biliary stenting, the optimal timing of definitive surgical intervention remains unclear; thus, we compared outcomes between index-admission and delayed surgical intervention.
Methods
This study was approved by the Institutional Review Board at the Lundquist institute affiliated with Harbor UCLA Medical Center. This was a multi-center retrospective chart review across 3 hospitals. Patients were identified by querying the electronic medical record for all cholecystectomies performed between 2014 and 2023 using relevant Current Procedural Terminology (CPT) codes within this time period (laparoscopic cholecystectomy with and without cholangiography (CPT 47562,47563), open cholecystectomy with and without cholangiography (CPT 47600,47605), cholecystectomy and common bile duct exploration (CPT 47610, 47612), percutaneous biliary catheter placement (CPT 47534), and cholecystostomy/choledochotomy (CPT 47480, 47420)). Additional extracted variables included date of service, procedure start/end time, procedure duration, procedure location, and admit/discharge date. Data was compiled into a Microsoft Excel database.
Subsequently, each operative report was screened, and preoperative and postoperative diagnoses were extracted. From these diagnoses, any operative report containing the word “Mirizzi” was isolated and a detailed chart review was performed. Extracted demographic data included age, sex, body mass index (BMI), and laboratory values at presentation (white blood cell count, alkaline phosphatase, aspartate aminotransferase, alanine aminotransferase, total bilirubin, and direct bilirubin).
The initial diagnostic modalities were extracted including RUQ US, MRCP, ERCP, or intraoperative diagnosis. For patients undergoing preoperative ERCP, biliary stent placement was noted. Operative reports were manually reviewed and coded by trained staff for surgical approach (laparoscopic, robotic-assisted, open, or conversion to open), use of intraoperative cholangiography, and performance of common bile duct (CBD) exploration. Based on the operative report details, a Mirizzi syndrome type was assigned according to the definition laid out by Csendes classification. Cases lacking sufficient documentation to permit classification were excluded. Surgeries were categorized as being performed during index admission or in delayed surgical setting if the patient was discharged home and returned for an elective procedure. The operating service was identified as the acute care surgery (ACS) team or a hepatopancreatobiliary specialist (HPB).
Primary outcomes included postoperative complications: iatrogenic bile duct injury, postoperative bile leak, biloma, abdominal abscess, need for postoperative percutaneous drain placement, and hospital readmission. These outcomes were stratified by Mirizzi Type and then compared between patients diagnosed preoperatively vs intraoperatively, those undergoing index-admission vs delayed surgery, and those operated on by ACS vs HPB services. Statistical analysis was performed using Fisher’s exact test with a significance level of α = 0.05. Analyses were conducted in Python (version 3.8.8) using the SciPy (version 1.10.1) and Pandas (version 1.2.4) packages.
Results
From 2014 to 2023, a total of 14 486 cholecystectomies were performed across 3 Los Angeles County hospitals. Of these, 68 cases of Mirizzi syndrome were identified, 65 of which met inclusion criteria, yielding an incidence of 0.45%. Inclusion criteria were any mention of the word “Mirizzi” or “Mirizzi syndrome” in the operative report findings or body of the report. Two cases were excluded due to insufficient documentation for classification, and one was excluded due to post-cholecystectomy Mirizzi syndrome. One case of gallbladder cancer was included, as malignancy was diagnosed on postoperative pathology rather than preoperatively. Per Csende’s classification, there were 39 (60%) type I, 6 (9%) type II, 16 (25%) type IV, and 4 (16%) type Va cases; no type III cases were identified.
For type I disease, the mean age was 49 years; 22 patients (56%) were female. The cohort was predominantly Hispanic (79.5%), with smaller proportions identifying as Asian (2.6%), Black (2.6%), or other ethnicities (15.4%). Demographic characteristics were similar across Mirizzi types (
Table 1). For laboratory values on presentation to the emergency room, median white blood cell count (WBC) was within normal limits across all Mirizzi types (<10,000/μL
3). There was a positive association between elevation in liver enzymes and disease severity; for type I Mirizzi syndrome the median total bilirubin (T. Bili) was 1.1 mg/dL while the median T. Bili for type Va was 6.9 mg/dL. The trends in aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (Alk Phos) were similar across Mirizzi type, with type I exhibiting the lowest values (52 U/L, 72 U/L, and 121 U/L, respectively) and type Va exhibiting higher values (213 U/L, 253.5 U/L, and 428 U/L, respectively). These findings are shown in
Table 1.
Preoperative diagnosis was established using MRCP, ERCP, or RUQ ultrasound. For type I, 15 (38.4%) cases were diagnosed by preoperative MRCP, 14 (35.9%) diagnosed by preoperative ERCP, and 9 (23.1%) were diagnosed intraoperatively. The most common method for type II was preoperative ERCP, 3 cases (50%). For type IV, 5 (31.3%) were identified by preoperative MRCP and 5 (31.3%) by preoperative ERCP. For type Va, preoperative ERCP was most commonly used, 2 (50%). The remaining methodologies used for diagnosis across all types are seen in
Table 2.
The operative approach to cholecystectomy varied when categorized by the Csendes classification. Out of 39 type I cases, 17 (43%) laparoscopic cholecystectomy was completed, while 13 (33%) cases began laparoscopic and then converted to open. 8 (20%) type I cases began as an open cholecystectomy. Out of 16 type IV cases, 9 (56%) were laparoscopic converted open, while the remaining 7 (44%) were open cholecystectomies. There were two robotic-assisted laparoscopic Mirizzi cases, 1 type I and 1 type Va. The more severe type was converted to open. The remaining distribution of operative approaches can be seen in
Table 3. Additionally, we noted the frequency of intraoperative cholangiogram performed, which occurred in 13% of type I cases and 25% of cases in types IV and Va. The frequency of CBD exploration and hepaticojejunostomy was also higher as disease severity progressed, as noted in
Table 4.
Overall, 51/65 (78.4%) were diagnosed preoperatively while 14/65 (21.5%) were diagnosed intraoperatively. Complication rates were significantly lower in the preoperative diagnosis group, 25% (13/51) vs 57% (8/14) (
P = 0.05). Most cases 52/65(80%) were completed during index admission with a complication rate of 25% (13/52) vs 61% (8/13) in the delayed surgery group (
P = 0.02). For type II-Va cases, complication rates did not differ for surgery at index admission vs delayed surgery, 10/18 (55%) vs 3/8 (38%),(
P = 0.67). Acute care surgery team performed 25 (38%) and HBP performed 40 (61%) cases. Complication rates were 24% (6/25) for ACS and 40% (16/40) for HPB (
P = 0.28). The ratio of cases performed by ACS vs HPB varied across Mirizzi type: 16 vs 23 for type I, 3 vs 3 for type II, 4 vs 12 for type IV, and 2 vs 2 for type Va. Specifically for type I cases, the complication rate was 3/16 (19%) complications for ACS and 4/23 for HPB (17%) (
P = 1.0) (
Tables 5-
8).
Discussion
This study represents one of the largest U.S.-based retrospective analyses of Mirizzi syndrome and uniquely compares outcomes by disease subtype, operating service, and timing of surgery. The observed incidence of 0.45% aligns with prior reports and reflects the rarity of this condition, even within a large urban safety-net hospital system serving historically underserved populations.
1Most cases were diagnosed preoperatively, and as previous studies hypothesized, preoperative diagnosis was associated with lower complication rates overall. A notable proportion of patients underwent delayed surgery following preoperative ERCP and biliary stenting, particularly among higher-grade disease. Though there was a significantly lower complication rate in the index surgery group compared to delayed surgery, there was no difference in complications with more severe disease. Although delayed surgery did not significantly reduce complications, this strategy may allow for physiologic optimization, biliary decompression, and referral to appropriate HPB expertise without increasing postoperative risk. Within our system, ERCP with biliary stenting was the predominant preoperative diagnostic and therapeutic modality, particularly for advanced disease. This approach appears to facilitate safe delayed surgery and appropriate subspecialty referral, which may be especially valuable in resource-limited settings.
As disease severity increased, the likelihood of open surgery and biliary reconstruction also increased. While type I disease was frequently managed laparoscopically, advanced types often required Roux-en-Y hepaticojejunostomy. Importantly, postoperative outcomes were similar between ACS and HPB services across all disease grades, although the technical demands of biliary reconstruction underscore the importance of HPB involvement in advanced cases. This supports that through the existing practice patterns, Mirizzi syndrome is likely to be diagnosed preoperatively via ERCP, and if indeed type I, can be managed acutely by the ACS team and can be completed laparoscopically.
Despite representing one of the largest U.S.-based series to date, this study is limited by the inherent rarity of Mirizzi syndrome, which restricts statistical power and limits the ability to draw definitive conclusions or establish standardized surgical guidelines. Additionally, given the underserved population within this public hospital system, postoperative complications may be underreported if patients sought care outside the network. This limitation was partially mitigated using a multicenter design within a unified electronic medical record, though some loss to follow-up was unavoidable. To help power the study, especially with the more severe cases, this study could include additional centers with similar populations across the country. Further, only two robotic-assisted cases were reported in this study, but the use of robotics for Mirizzi syndrome has been increasing.
8 As robotic surgery becomes more commonly adept in treating biliary pathology, its role in complex gallbladder disease is an important area of further study.
Conclusions
Overall, our findings support existing practice patterns favoring preoperative diagnosis and early operative management of type I disease, while suggesting that delayed surgery following biliary decompression may be a safe strategy in select patients. Further prospective, multicenter studies are needed to better define optimal timing and operative strategies, particularly for higher-grade Mirizzi syndrome.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.