Patient Autonomy in Medical Education: Navigating Ethical Challenges in the Age of Artificial Intelligence
Abstract
Introduction
Materials and Methods
Search Strategy and Inclusion
Databases Search Protocol and Keywords
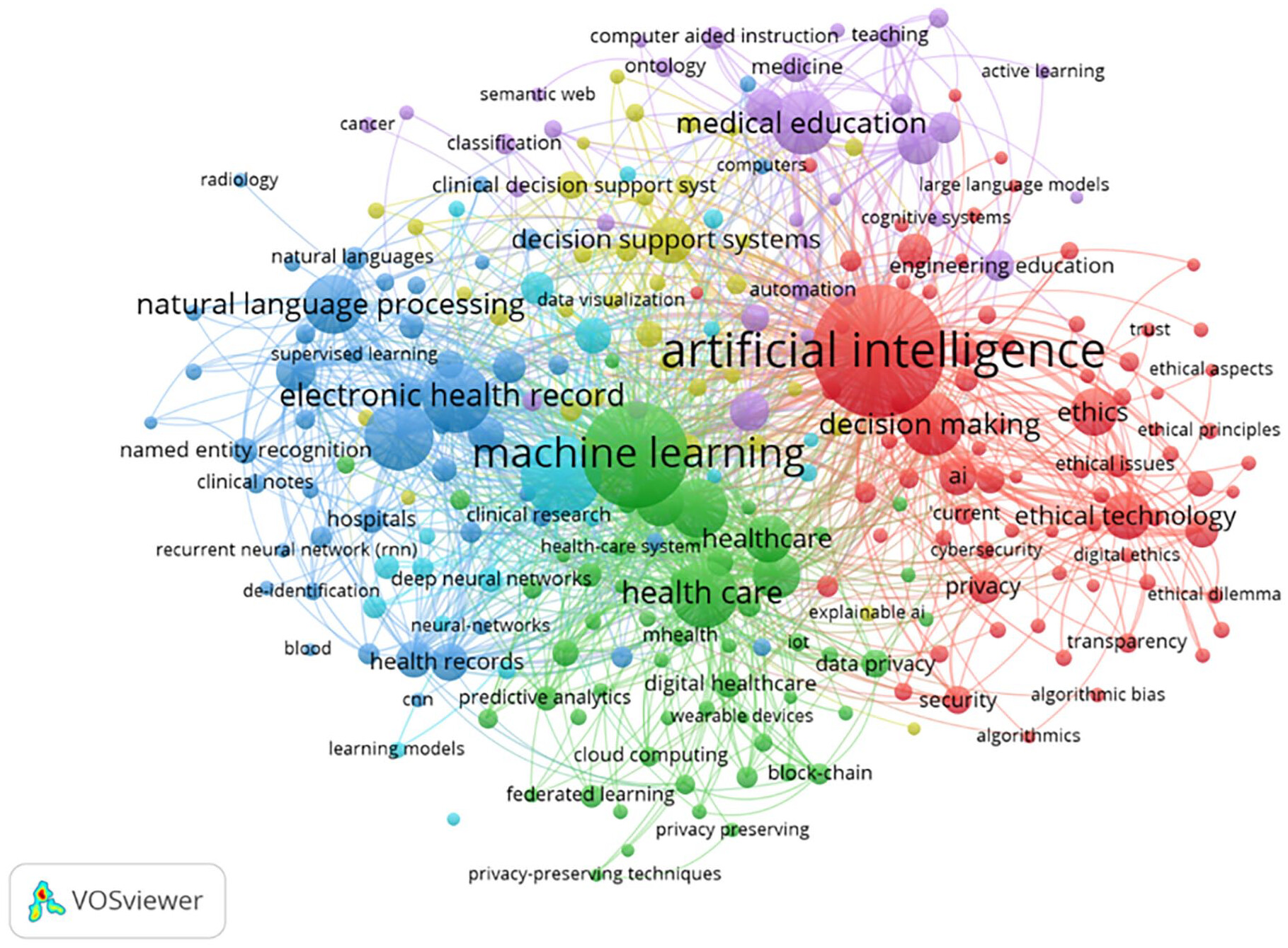
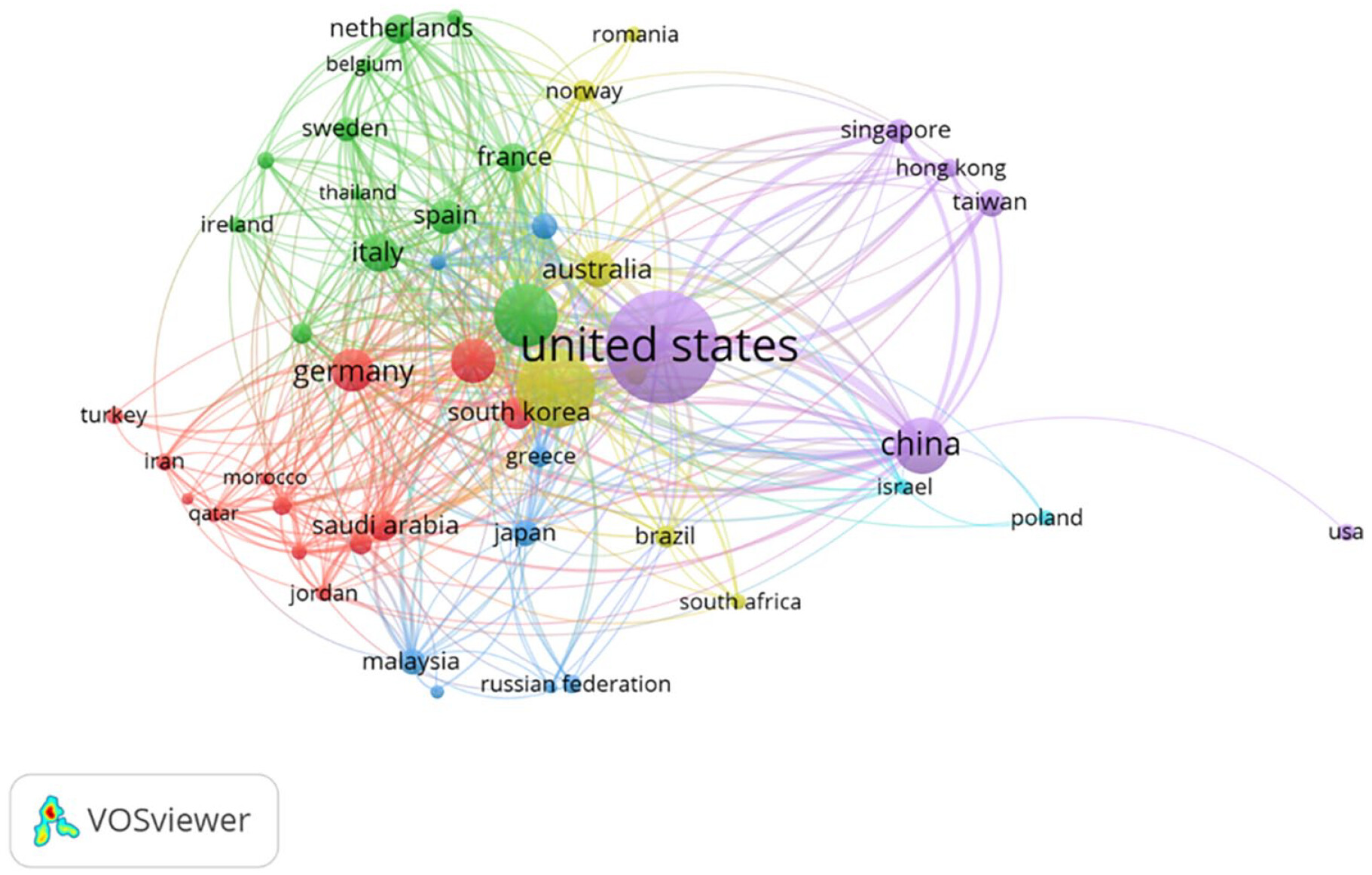
Keywords Co-occurrence Analysis
Data Extraction
The Importance of Patient Autonomy
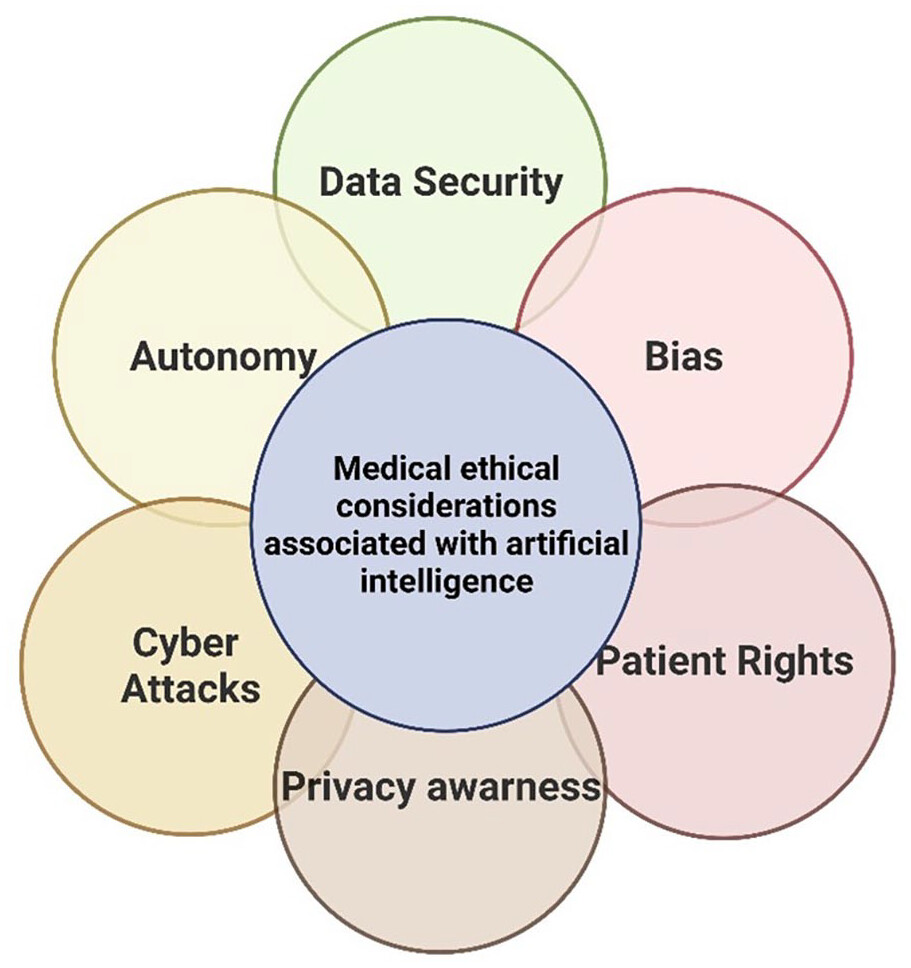
Ethical Challenges Faced
Cultivating Ethical Decision-Making Skills in Students
Data Protection Laws in Medical Education
| Aspect | HIPAA (United States) | GDPR (Europe) |
|---|---|---|
| Purpose | Protects the privacy of personal health information and provides rights to patients. | Ensures the protection of personal data and empowers individuals’ data rights |
| Scope | Applies to healthcare providers, health plans, and healthcare clearinghouses. | Applies broadly to any organization processing personal data. |
| Patient Rights | - Right to access and request their medical records. - Right to request corrections to inaccurate health information. - Right to privacy and control over disclosure of health information. | - Right to access and obtain a copy of their personal data. - Right to rectify inaccurate data. - Right to erasure (right to be forgotten). - Right to data portability. |
| Data Protection Officer (DPO) | Not mandated, but some covered entities appoint a Privacy Officer. | Mandatory for certain organizations. |
| Data Breach Notification | Requires covered entities to notify affected individuals and the HHS within 60 days of a breach. | Requires organizations to report data breaches to the appropriate data protection authority within 72 h. |
| Penalties for Non-Compliance | Civil penalties, ranging from fines from $100 to $50 000 per violation, with an annual maximum of $1.5 million. | Fines of up to 4% of global annual turnover or €20 million, whichever is higher, for severe violations. |
| International Applicability | Applies primarily within the United States but has implications for global healthcare organizations. | Applies to organizations outside the EU that process EU residents’ data. |
| Country | Law/Regulation | Purpose |
|---|---|---|
| Australia | AI Safety Framework54 | Establishes standards and principles for safe and effective AI applications in healthcare |
| Therapeutic Goods (Medical Devices) Regulations 200255 | Supports clinical decision-making with software | |
| Brazil | Brazilian Artificial Intelligence Bill No.21/202056 | Develops and implements AI across various sectors in Brazil |
| Lei Geral de Proteção de Dados (LGPD) (General Personal Data Protection Law)57 | Aligns with GDPR standards to address comprehensive data protection needs | |
| Canada | Consumer Privacy Protection Act (CPPA)58 | Modernizes the framework for protecting personal data in the digital age |
| Digital Charter Implementation Act, (Bill C-27) 202259 | Protects personal information and health records, ensuring AI does not cause substantial direct harm to patients | |
| China | Announcement by the State Council on the Release of the New Generation Artificial Intelligence Development Plan, State Council Document No. 35, 201760 | Establishes comprehensive national regulations covering all aspects of AI, enhancing oversight and risk management |
| Cybersecurity Law of China61 | Maintains the sovereignty of cyberspace and protects national security | |
| European Union | Regulation by the European Parliament and the Council establishing unified regulations on AI Act and modifying certain legislative acts of the Union62 | Enables and fosters innovation in AI while developing trustworthy AI applications |
| In Vitro Diagnostic Medical Devices Regulation (IVDR)63 | Protects the well-being of patients and users, ensuring the quality and safety of in vitro medical devices | |
| Medical Devices Regulations 2017/745(MDR)64 | Ensures the safety of patients using medical devices and secures the data generated through these devices | |
| General Data Protection Regulation (GDPR)65 | Enhances and unifies data protection for all individuals within the EU | |
| Civil Law Rules on Robotics66 | Implements AI robotics | |
| Germany | Patient Data Protection Act (PDPA)67 | Protects personal and sensitive patient data, promoting digital health records |
| India | Personal Data Protection Bill68 | Protects personal data and establishes a Data Protection Authority |
| Japan | Act on the Protection of Personal Information (APPI)69 | Regulates personal data protection with tighter controls on data transfers |
| Kingdom of Saudi Arabia | Medical Device/SFDA MDS-G23 software70 | Utilizes AI and Big Data in medical software to analyze and forecast patient health conditions |
| Singapore | Personal Data Protection Act (PDPA)71 | Enhances the protection of personal data and addresses management and security obligations |
| Qualification of Clinical Decision Support Software (CDSS) and Standalone Medical Mobile Applications(SaMD)72 | Design software to assist healthcare providers in clinical decisions | |
| South Korea | Personal Information Protection Act (PIPA)73 | Strengthens the use and protection of personal information |
| Medical Devices Act No. 15945, 11 December 201874 | Categorizes software as a medical device used in healthcare settings | |
| United Arab Emirates | AI in the Healthcare Sector of the Emirate of Abu Dhabi, Policy/AI/0.9, Version 0.975 | Monitors, analyzes, and observes public health within the healthcare system |
| United Kingdom | Medical Devices Regulation (Specific to AI) and Data Protection Act (DPA)76 | Integrates AI as a subcategory under software medical devices, providing guidelines for safety and performance |
| USA | HIPAA updates77 | Improves privacy protections and healthcare data interoperability under HIPAA |
| Part 2 Final Rule (42 CFR Part 2)78 | Enhances protections for substance use disorder patient records | |
| Health IT Interoperability and Algorithm Transparency79 | Ensures transparency in AI algorithms in healthcare IT, improving safety and effectiveness assessments |
Adherence to Treatment With the Help of AI and Machine Learning
Future Outlook
Study Limitations
Conclusion
Acknowledgments
Ethical Approval and Consent to Participate
Consent for Publication
Declaration of Conflicting Interests
Funding
ORCID iDs
Footnote
Data availability statement
References
Cite
Cite
Cite
Download to reference manager
If you have citation software installed, you can download citation data to the citation manager of your choice
Information, rights and permissions
Information
Published In
Keywords
Data availability statement
Authors
Authors’ Contributions
Metrics and citations
Metrics
Journals metrics
This article was published in INQUIRY: The Journal of Health Care Organization, Provision, and Financing.
View All Journal MetricsPublication usage*
Total views and downloads: 4611
*Publication usage tracking started in December 2016
Publications citing this one
Receive email alerts when this publication is cited
Web of Science: 19 view articles Opens in new tab
Crossref: 21
- Cyberchondria and complexity: a systems-level exploration of anxiety and informational instability in the digital age
- Ethical concerns and strategies for implementing artificial intelligence in healthcare: a review of empirical studies
- Health sciences students’ attitudes toward artificial intelligence: predictors of ethical awareness, clinical decision-making, and public health perceptions-a cross-sectional study
- Artificial Intelligence in Healthcare and Biomedical Visualization
- Nanotheranostics in Metabolic Syndrome
- AI‐driven Innovations in Physiotherapy and Oncology 2
- The ethical challenges in the integration of artificial intelligence and large language models in medical education: A scoping review
- Dimensions of Artificial Intelligence Literacy: A Qualitative Synthesis of Contemporary Research Literature
- Artificial Intelligence for Patient Education: A Primer, Not a Proxy
- Leveraging 3D Cell Culture and Artificial Intelligence Technologies for Regenerative Medicine
- View More
Figures and tables
Figures & Media
Tables
View Options
View options
PDF/EPUB
View PDF/EPUBAccess options
If you have access to journal content via a personal subscription, university, library, employer or society, select from the options below:
loading institutional access options
Alternatively, view purchase options below:
Access journal content via a DeepDyve subscription or find out more about this option.



