Introduction
The consumption of energy in buildings accounts for about 40% of European energy consumption and 36% of total European CO
2 emissions.
1 One of the most effective ways to reduce the greenhouse gas emissions is by improving building insulation levels. There is a large range of thermal insulation boards, batts and rolls, which have been tested by this laboratory, offering nominal thermal conductivity performances ranging from 0.018 W/m·K to 0.020 W/m·K (phenolic foams), 0.020 W/m·K to 0.026 W/m·K polyisocyanurate foams (PIR), 0.030 W/m·K to 0.038 W/m·K (expanded polystyrene (EPS) foams), 0.032 W/m·K to 0.044 W/m·K (mineral fibre), 0.035 W/m·K to 0.040 W/m·K (cellulose fibres), 0.038 W/m·K to 0.050 W/m·K (wood fibre), up to 0.038 W/m·K to 0.070 W/m·K (cork).
One of the advantages of EPS foam is that while there are short-term changes resulting from the initial blowing process, the thermal insulation performance quickly establishes a stable long-term value. The foam consists of approximately 98% air and 2% polystyrene. Grey or silver EPS products attribute their distinctive colour to a small quantity of carbon or graphite contained within the polymer matrix of the rigid foam. The carbon particles absorb infrared (IR) radiation energy, thereby minimising the contribution of radiant heat transfer to thermal conductivity, while retaining the other performance benefits normally found in standard white EPS rigid foams.
The air entrapped within the cells has a low thermal conductivity and so plays a decisive role in providing the foam with its thermal insulation properties. Unlike foams retaining low conductivity heavy molecular gases, the air stays in the cells and does not age except for blowing agent diffusion during the initial manufacturing process. The insulation performance effectively remains constant over the economic lifetime of the product, but there are short-term thermal conductivity effects arising from the initial production processes.
EPS is obtained by polymerising styrene and introducing small amounts of blowing agent such as pentane. The expanded material is made using a steam process, the high temperature softening the beads and the blowing agent expanding them up to 50 times their original size. Initial blowing agent contents might typically be up to 6 wt%
2 which results in a pentane/air gaseous mixture having a thermal conductivity value less than that of air. The pentane quickly diffuses out the foam to less than 1 wt% content over a period of about 2–3 weeks after manufacture.
3 The remaining pentane continues to diffuse but is eventually dissipated to insignificant levels such that the thermal conductivity effectively reaches a long-term stable level within the first few weeks after production.
If the thermal conductivity is measured too soon after production, the residual pentane found in freshly manufactured EPS can give an optimistically low value which does not reflect the long-term stabilised value. Typical changes in thermal conductivity during factory processing of EPS products are given in SG19 Guidance
4 (Q.57). For grey products, a typical increase would be 0.001 W/m·K, while for white products, the increase would typically be much lower. While the graphite minimises radiative heat transfer in grey EPS, it reportedly holds pentane for longer than white EPS and takes longer to diffuse out of moulded products.
Although a first-term change after production of 0.001 W/m·K is relatively small (3%), there is a need to declare a stabilised thermal conductivity value that is constant throughout the economic life of the product. To declare thermal properties that reflect the values during the expected lifetime of the product, the manufacturer must ensure that measurements are not carried out until the thermal conductivity has reached its final level, or that the conditioning effect is taken into account, for example, by an accelerated procedure
4 (Q.27).
There is a requirement in the European product standard for EPS board to condition at elevated temperatures (see
Appendix 1), which is assumed to be sufficient preconditioning to determine the final level of thermal conductivity. EPS boards are to be conditioned to constant mass at 70°C
5 to ensure sufficient reduction of pentane before thermal conductivity testing. Elevated temperatures raise the partial pressure of pentane and increase the rate of diffusion out of the cells. One of the concerns of some manufacturers has been that their products may not be tolerant of elevated temperature conditioning if they contain residual blowing agent with a possible risk of material changes. Foam products should not be continuously exposed to temperatures in excess of 70°C. Beads containing blowing agent may soften and expand in the range 60–101.7°C.
6When the wording of a standard is ambiguous or requires clarification, the working group SG19 (sector group for thermal insulation) for Notified Bodies try to bring an interim and practical position to allow for appropriate assessment and declaration. The guidance is given in the form of agreed answers to questions raised within SG19. In this approach, thermal conductivity change in EPS is deemed insignificant if, following measurement after 70°C conditioning, the measurement is repeated after reconditioning at 60°C and the difference does not exceed 1%. For un-elastified EPS samples of 50 mm thickness and a density range of 15–25 kg/m
3, the above procedure is deemed sufficient to determine the final level of thermal conductivity
4 Q.27.
The SG19 procedure requires repeated conditioning at elevated temperatures which accelerates thermal conductivity ageing. It is not made clear in this procedure why initial conditioning and reconditioning are not at the same temperature. This requires correction or clarification, and at the time of writing, the procedures are under review by SG19 and EN standards. An interim position by The European Manufacturers of Expanded Polystyrene specifies conditioning and reconditioning at elevated temperatures of 60°C or 50°C, depending on the product type.
7 The objective of this article is to examine and compare the effect of room temperature and elevated temperature conditioning on the thermal conductivity of identical products of 50 mm silver grey EPS boards from the same batch sampled at the point of production. Two boards were conditioned at 70°C, and two conditioned at 23°C/50% RH where there is no risk of heat conditioning damage. The thermal conductivity of the boards was initially measured within a few hours of manufacture and then retested at frequent intervals over a period of 262 days thereafter.
The results of a programme of retesting of ambient conditioned retained grey EPS samples, in some cases several years after the initial tests, have also been included in order to determine the time at which initial measurements after 23°C/50% RH conditioning reflect a stabilised thermal conductivity value that is constant throughout the economic life of the product.
The heat transfer processes in porous materials and applicable theoretical models for EPS foams have been considered. Scanning electron microscopy (SEM) of the cell structures of the grey EPS samples by Salford Analytical Services has been included.
Test samples
Four test samples of an identical product of silver EPS board of nominal density 17 kg/m3 were collected at the manufacturer’s factory from the same batch of material, approximately 1 h after production, having been cut to the required size by the manufacturer. The product consisted of a collection of beads moulded together under compression to produce a near-homogeneous EPS board. These samples were then given unique identification marks, and their dimensions and mass were recorded before being sealed in a gas-tight plastic envelope for their journey to the test laboratory. From the four samples, two were to be conditioned in a sealed and controlled environment of 23 ± 2°C and 50 ± 5% RH, with the other two samples being conditioned at 70 ± 2°C in a temperature-controlled oven. For both conditioning criteria, each sample was labelled A and B.
After transportation from the factory to the laboratory, the unconditioned samples were unwrapped, and the dimensions and mass were recorded again. Samples labelled A, each from the 23°C and 70°C selections, were tested immediately for thermal conductivity on arrival, at approximately 6 h after being collected from the factory. The samples labelled B were placed on arrival in their selected conditioning environment overnight and tested the following morning around 22 h after being collected. Each test was set to run for approximately 2 h in order for the sample to achieve an adequately stable thermal equilibrium and the samples were repeatedly tested at regular intervals of between 16 h and 24 h of conditioning for approximately 2 weeks, after which there were increasing conditioning intervals in between the testing. The method of testing the thermal conductivity is described in more detail in the fifth section.
Heat transfer processes in porous materials and applicable theoretical models
Thermal insulation materials are usually highly porous and consist of a solid matrix full of small voids that comprise the bulk of the total volume. These voids normally contain air or some other low conductivity gas. The thermal conductivity of the material is the result of various basic heat transfer mechanisms: solid and gas conduction, gas convection and long-wave radiation within the voids.
8,9 The apparent conductivity depends on the kind of solid, bulk density, temperature, water content, thickness and age. Factors such as cell shape and diameter and arrangement of any fibres or particles, transparency to thermal radiation or type and pressure of the gas come into play.
Air convection cannot occur in pores smaller than a few millimetres. EPS cell sizes may typically be in the range 50–350 µm
10 and the convection component of total thermal conductivity is negligible. The thermal conductivity of EPS cellular foams is therefore determined by conductive and radiation contributions.
Thermal conductivity due to conductive heat transfer λ c (depends on the structural composition and the combined effects of conduction through solid phase λ s, and conduction through gas phase λ g). The thermal conductivity due to radiation energy transfer is λ r.
In the absence of convection and for separable conduction and radiative processes, the total thermal conductivity λ of a porous material may be written as
It should be noted that the EPS boards tested consisted of a collection of beads moulded together under compression to produce a near-homogeneous material with negligible inter-bead voids and convection. For loose fill EPS beads, which are often used in cavity insulation applications, there are interconnected voids between the individual beads, forming a tortuous but continuous air path through the material. Consequently, there can be a small but continuous convective path through loose beads, dependent on the bead and void size. Tests in this laboratory indicate that the thermal conductivity of loose EPS beads can be up to 5% more than that for EPS boards of a similar density.
Figure 1 shows a digital microscopy photograph of bonded beads in the EPS boards received for testing. These are compared with a sample of loose EPS beads of similar density in
Figure 2. The scale mark in red in the photographs represents a distance of 1 mm.
Radiative heat transfer
Radiative heat transfer is dependent on the fourth power of the absolute temperature, and as the temperature is increased, the radiative component can increase dramatically in comparison to that due to conduction processes. At ambient temperatures and for materials of low densities such as those used in the thermal insulation in buildings, the radiation contributions can be lower than the conduction part, but still significant. For a given temperature, the main factor controlling the radiative contribution is the number of radiation barriers within the insulation which scatter or absorb the radiation, and it generally decreases with increasing density.
Many approaches at treating the scattering mechanisms have been based on a set of differential equations by Hamaker.
11 His analysis has been extended by Larkin,
12 Larkin and Churchill,
13 Simpson,
14 and Simpson and Stuckes.
9 Hamaker’s model has been adopted by several investigators
15,16 for fibrous insulation. The more rigorous Hamaker two-flux model treats the net radiative transfer as the difference between the radiant fluxes (scattered, absorbed and re-emitted) in the forward and backward directions. The Hamaker model was extended for materials where scattering is the dominant mechanism,
13 as in EPS foams without graphite IR attenuators.
Doherty et al.
17 suggested that the walls in many plastic foams are insufficiently thick to absorb all the radiation impinging on them. Skotchdopole,
18 and to a lesser extent, Stephenson and Mark,
19 found that a linear dependence existed between cell size and the measured thermal conductivity of polystyrene foams. The magnitude of the slope was substantially larger than would be predicted by an equation given by Doherty et al. which considers all radiation as being obstructed at each cell boundary then re-emitted, thereby indicating the transparency of polystyrene foams to IR radiation.
Radiative heat transfer through porous materials can occur by direct transmission through the cells, by absorption and re-radiation, and by scattering. While absorption occurs primarily in transmission through the solid material, scattering occurs when IR waves encounter a discontinuity in the refractive index; for foamed materials, this may occur at the surface of the cells. Where the reflecting surfaces are small enough, that is of the same order as the dominant IR wavelength (3–30 µm at room temperatures), scattering becomes an important consideration.
Cell sizes in EPS foams may be in the range 50–350 µm.
10 The cell dimensions would appear to offer very little scattering potential, however, Lanceley et al.
20 found the extensive intercell skeletal strands were small enough for scattering. Microscopic measurements indicated that although the face thickness of the cell membrane was only about 0.4 µm, the skeletal strands were up to 3.2 µm thick.
For white EPS products in the built environment, the radiative component has more significance where there is no opacifier to restrict the transfer of IR radiation. For carbon- or graphite-enhanced EPS, the radiative contribution can be further reduced and dominated by IR absorbing graphite particles.
Conductive heat transfer λc in porous materials
Most porous materials may be considered as mixtures of gas (usually air) of thermal conductivity λ g and solid λ s. In general, the thermal behaviour of a material lies between that of its components and depends on the volume fraction of each and its distribution and shape. Due to the complexity of the structure of real materials, there are few cases where rigorous calculations are possible.
Simpson and Stuckes
8 considered a general equation for multiphase materials based on the equation for the dielectric constant of mixtures by Reynolds and Hough.
21 The models consider a spheroidal shape of the dispersed phase in a continuous matrix material. The phases are assumed isotropic and homogeneous throughout. The general formula for the thermal conductivity
λ of two-phase media is
Subscript 0 refers to the continuous matrix phase, and subscript 1 refers to the dispersed phase, where Vi, λi and fi are the volume fraction, thermal conductivity and spheroidal field factor, respectively, for each phase.
The cell structure of EPS foam is similar to that of soapy water, consisting of an array of air-filled, closed plastic membrane polyhedral cells.
20 Spheres provide a good approximation to the shape of the gas-filled cells in a polystyrene solid matrix. For spherical shapes, the equation can then be simplified to two limiting cases.
Maxwell
22–Eucken
23 model for spheres of phase 1 dispersed in continuous phase 0
Brailsford and Major
24 model for spheres of phase 0 and phase 1 embedded in a mixture having the thermal conductivity
λ of that being calculated
The Brailsford and Major model gives a good estimate of the conductivity of a mixture if neither phase is continuous.
The mean of the Maxwell–Eucken and Brailsford–Major models should provide a more realistic model intermediate between the upper and lower cases of (i) pores of phase 1 dispersed in continuous solid of phase 0, and (ii) pores and solid phase embedded in a mixture having the λ of that being calculated. For the purposes of modelling the conductive processes in the mixture of gas and solid, the combined thermal conductivity of the mixture is therefore taken to be
Conductive heat transfer in gases λg
Gas conductivity and pore size
For pores smaller than a few millimetres, air convection cannot occur and heat transfer through the gas depends on collisions between the molecules within the pores and the conductivity is normally that of still air.
For air-filled super insulation materials with a predominance of pores of <0.1 µm in size, the conductivity may be effectively reduced below that of still air by restricting collisions between gas molecules. It has been shown by Kistler
25 that for a given pressure, the mean length of the free path
Lp for molecules confined within the pores of an insulation is given by
where d is the mean free path of the gas in a free state and L 0 is the average pore dimension. According to kinetic theory, the thermal conductivity of a gas is proportional to the mean free path of the molecules. Consequently, the effective conductivity of the gas contained within the pore is given by
where λ g is the conductivity of free gas.
The quantity
λp is plotted against pore size for the conductivity of free air
λ g = 0.0251 Wm K at 10°C and at atmospheric pressure, given a mean free path of 68 nm, in
Figure 3. In nanoporous materials where the size is less than 0.1 µm, the effective conductivity of air is significantly less than that of free or still air. However, EPS foam cell sizes are generally two orders of magnitude larger and the conductivity of the air in the cells remains that of free air.
Gas mixture thermal conductivity λg
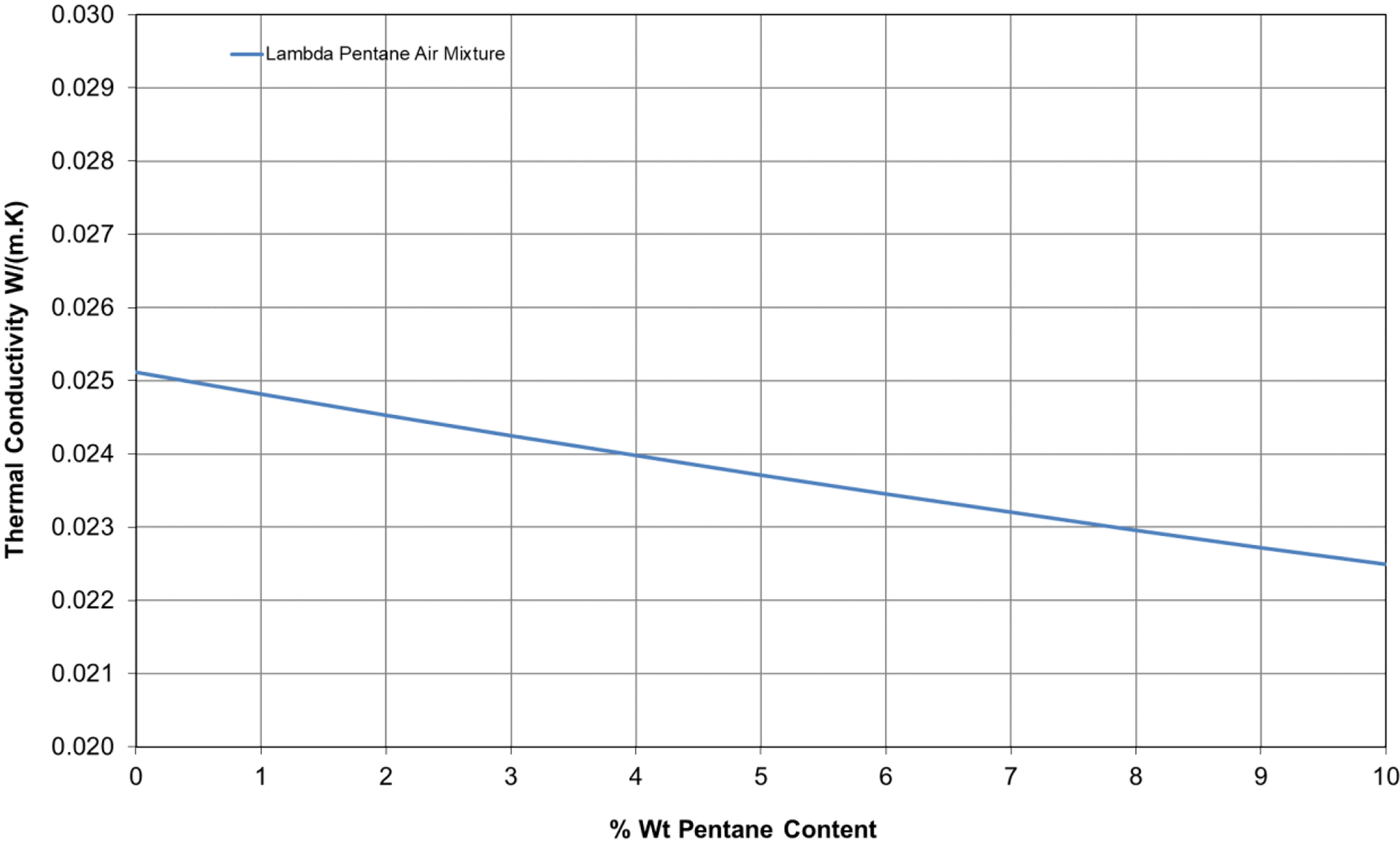
Materials such as polyurethane foam depend on low conductivity heavy molecular gases being trapped in the cells. When blowing EPS foam with pentane, the gas will diffuse out quickly and the air diffuses in. After a short curing time, the cells will contain only air and the thermal conductivity of air is 0.0251 W/m·K at a mean temperature of Tm = 10°C.
In the initial production stages that EPS cells contain a mixture of air and pentane gases and the thermal conductivity of the gas mixture can be expressed as
26where
x pentane and
x air are mole fraction components at time
t,
λ pentane and
λ air are thermal conductivity of the gas components and
λ g is shown in
Figure 4 for a pentane/air mixture. Molar mass of pentane is 72.17 kg/kmol and air 28.97 kg/kmol. The thermal conductivity at 10°C of normal/isopentane is taken to be 0.013 W/m·K
27 and air 0.0251 W/m·K.
28Initial blowing agent contents are typically up to 6 wt%
2 which results in a pentane/air gaseous mixture having lower thermal conductivity (0.0235 W/m·K) than that of air-filled cells. Pentane quickly diffuses out of EPS foam to less than 1% over a period of about 2–3 weeks after manufacture.
3 The remaining pentane continues to diffuse slowly over longer periods but is eventually dissipated to insignificant levels such that the thermal conductivity reaches a long-term stable level within the first few weeks after production.
Solid nonporous thermal conductivity
The thermal conductivity of solid nonporous polystyrene is taken to be 0.150 W/m·K and density 1050 kg/m
3 at 283 K.
26,29 Styrene polymer is inherently thermally insulating whereas carbon or graphite is a conductive filler which can potentially raise the solid conductivity component of grey EPS foams. The carbon content contained within the grey EPS foam samples examined was not disclosed by the manufacturer for confidentiality reasons. It is however understood to be relatively small. Vo et al.
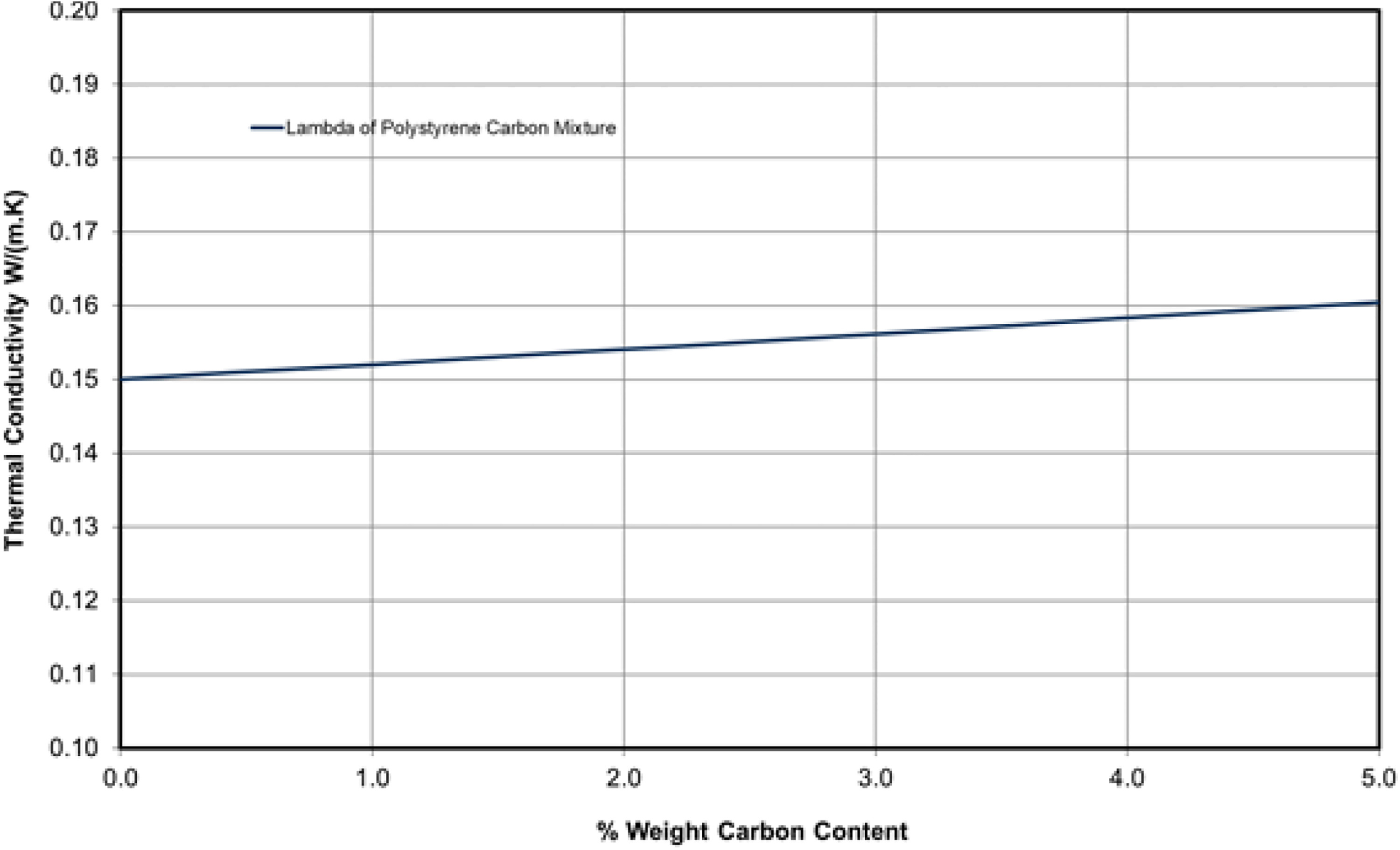
26 calculate that the increase in the thermal conductivity of the solid blend is small for the addition of 1% by weight of carbon black or graphite.
Based on a thermal conductivity for carbon black of 21.7 W/m·K,
26 the authors have calculated the thermal conductivity of a nonporous solid mixture of polystyrene/carbon for up to 5% by weight of carbon black (density 2200 kg/m
3) using equation (
5). The thermal conductivity of the solid blend is predicted to increase by approximately 1.4% for a 1 wt% increase in carbon (
Figure 5). This has a negligible effect on the overall thermal conductivity of low-density EPS insulating boards, consisting of approximately 98% air and 2% polystyrene. Assuming the above carbon content range for the samples under investigation (density 17.3 kg/m
3), the solid polystyrene content would only be about 1.6% and carbon less than 0.08%.
Composite gas/solid conductive heat transfer
The composite gas/solid conductive heat transfer in the silver EPS of density 17.3 kg/m
3 has been calculated in
Table 1 for initial (6 wt%) and final (0 wt%) pentane contents in the cells, based on equation (
5).
Thermal conductivity ageing and diffusion of blowing agent
The λ g value is affected by EPS foam ageing as it depends on the gas mixture composition which starts to change as soon as the processing is finished and continues until a new equilibrium with air is reached. The changes are due to the diffusion of O2 and N2 into the cells and the blowing agent pentane out of the foam cells. The diffusion of gas through a polymeric membrane increases with temperature and relative humidity and the ageing process is accelerated at elevated temperatures.
The thermal conductivity of PIR foams has been found
30 to age according an empirical relationship using a modified form of a sigmoid (S-shaped) function, in which the power is proportional to the log of time. In equation (
9),
λ is the thermal conductivity at a given time, the subscript
i denoting the initial thermal conductivity,
t is the time in days. Δ
λ represents the incremental increase in thermal conductivity from the initial value to the final plateau value.
C is a time constant which determines the ageing rate which is dependent on several factors such as ageing temperature, relative humidity, product density and thickness and diffusion rates through the polymeric membranes. Theoretical evaluation of these relationships is beyond the scope of this investigation.
Modified sigmoid (function of log time)
The empirical equation was found to closely describe both 23°C/50% RH thermal conductivity ageing of polyurethane foams and accelerated ageing at an elevated temperature of 70°C. The thermal conductivity of the same product was measured at a mean temperature of 10°C after the samples had been aged for a time
t.
Figure 6 illustrates a schematic graph of the empirical relationship for the dependence of
λ on log of time of the same product aged at 70°C and at 23°C/50% RH from the time of manufacture. Typical
λ values for polyurethane foams may be 0.02 W/m·K, increasing by some 10–30% over a period of decades, the rate being dependent on product properties such as thickness, blowing agent, facings and density.
λ increases with ageing time as the higher conductivity air diffuses in and the lower conductivity blowing agent diffuses out. The loss of blowing agent increases with accelerated ageing at elevated temperature, resulting in the thermal conductivity increasing more rapidly with time. As the blowing agent dissipates to insignificant levels and is replaced by air, the thermal conductivity tends towards a constant plateau level which is the long-term final value. For a given product sample thickness, density and blowing agent, the time constant C was found to decrease strongly with the ageing temperature as diffusion increases. Since the diffusion of gas through a polymeric membrane is also dependent on relative humidity, the temperature dependence of C has not been evaluated for the ageing regimes described above.
This type of equation has been used to describe accelerated and room temperature thermal conductivity ageing with time of EPS foams. For EPS foams, the plateau level may effectively be reached within weeks after production, whereas polyurethane foams may not be fully aged after several decades. Initial blowing agent contents in EPS might typically be 6 wt% resulting in a pentane/air gaseous mixture having thermal conductivity values at the time of manufacture of less than that of air. The bulk of the pentane quickly diffuses naturally out of the foam to about 1% or less over a period of about 2–3 weeks after manufacture. The remaining pentane continues to diffuse slowly over longer periods but is eventually dissipated to insignificant levels such that the thermal conductivity reaches the plateau level. Consequently, after a relatively short curing time, the cells will contain air and the gas thermal conductivity tends to that of air 0.0251 W/m·K at a mean temperature of 10°C.
Material characteristics
The theoretical models in the previous section describe the heat transfer implications for EPS materials. Electron microscopy techniques using high-resolution SEM have been used to examine the cell structure.
It should be noted that the characterisation of porous solids conforms to the new pore size classification proposed by Mays.
31 The classification is based on a logarithmic scale (to base 10), and is summarised for the three main pore size ranges as:
Nanopore: pore size between 0.1 nm and 100 nm (1 nm = 10−9 m).
Micropore: pore size between 0.1 µm and 100 µm (1 µm = 10−6 m).
Millipore: pore size between 0.1 mm and 100 mm (1 mm = 10−3 m).
Each range covers three decades of pore size, with the lower boundary of the middle decade being the reference pore size for the range (1 nm, 1 µm and 1 mm for nanopores, micropores and millipores, respectively).
SEM and analysis
SEM micrographs of the conditioned silver EPS foam samples were obtained using system magnification of 1000×. Before examination, small sections of each type of conditioned sample were briefly cooled using liquid nitrogen and sliced using a razor blade to obtain a cross-sectional area of the inner surface.
Micrographs of the silver EPS foam sample used during thermal conductivity tests, after being conditioned at 23°C/50% RH and 70°C for 262 days, are shown in
Figures 7 and
8. The cell structure consists of an array of air-filled, polyhedral cells, with a broad range of cell dimensions from 15 µm to 115 µm in diameter. The cells were generally found to be microporous and therefore unlikely to exhibit the potential nanoporous (<0.1 µm) effect of the thermal conductivity of air being significantly less than that of free or still air. Where the reflecting surfaces are of similar same order as the dominant IR wavelength (3–30 µm at room temperatures), IR scattering becomes an important consideration. The relatively large cell dimensions would appear to offer limited scattering potential. The effect of graphite IR attenuator appears to be responsible for a relatively opaque cell membrane, restricting radiative heat transfer.
One of the concerns of some manufacturers is that EPS products may not be tolerant of conditioning at elevated temperatures. Although the cell walls in
Figures 7 and
8 appear to be generally closed, there are some small perforations in the membranes. Small perforations are unlikely to have a significant effect on the thermal conductivity, but it is worth noting that should the number and size of perforations become extensive, the possibility of a continuous open cell structure and risk of heat transfer by small convection currents increases. There is little increase in the number of perforations due to conditioning at an elevated temperature of 70°C, but the intercell skeletal strands and cell walls display some distortion or wrinkling.
In practice, test samples are only subjected to short-term conditioning of a few days at elevated temperatures. Consequently, additional silver EPS samples, less than 1-day-old and from the same production line, were acquired after completion of the thermal conductivity tests and subjected to short-term conditioning at elevated temperatures and 23°C/50% RH.
SEM micrographs after 7 days conditioning at elevated temperatures of 60°C and 70°C do not suggest increase in the number and size of perforations, but there is some indication of increase in wrinkling of the intercell skeletal strands and cell walls (
Figures 9 to
11). Although there is a possible risk of change to the cell walls after accelerated conditioning at elevated temperatures, the thermal conductivity is unlikely to be affected as there was no significant change in the cell size. It takes longer to eliminate the pentane gas by conditioning at 23°C/50% RH but there is no risk of material change to the cells. It should be emphasised that this is only a cursory SEM examination and a more rigorous and systematic study would be required to fully evaluate the effect of elevated temperature conditioning on EPS cell structure.
Conclusions
To declare thermal insulation properties that reflect the values during the expected lifetime of the product, a manufacturer must ensure that thermal conductivity measurements are not carried out until the value has reached its final level, or that the conditioning effect (diffusion of pentane out of the EPS cells) is taken into account, for example, by an accelerated procedure at an elevated temperature as described in product standard
5 or SG19 Guidance.
4 The criteria in SG19 Guidance
4 suggests that the measured thermal conductivity should be within 1% of the final value. A concern of some manufacturers has been that their products may not be tolerant of elevated temperature conditioning at 70°C.
The effect of elevated temperature conditioning at 70°C has been compared with ambient (23°C/50% RH) conditioning on the thermal conductivity of freshly manufactured identical products of 50 mm silver EPS boards from the same production batch. The thermal conductivity of the boards was initially measured within a few hours of manufacture and then retested at frequent intervals thereafter over a period of 262 days.
Theoretical models of porous or multiphase materials have been applied to the thermal conductivity results. The thermal conductivity performance as a result of pentane diffusion was found to be accurately described by a modified sigmoid function. An increase in thermal conductivity of 0.001 W/m·K was noted in accord with the changes during factory processing of grey EPS products suggested by SG19 Guidance.
4 The inclusion of carbon or graphite IR attenuator in 50 mm silver EPS board (17.3 kg/m
3) was found to offer a significant reduction (16%) in thermal conductivity compared to that of a white EPS of the same density and thickness.
The evidence from SEM demonstrated that the cell structure consists of an array of air-filled, polyhedral cells, with a broad range of cell dimensions from 15 µm to 115 µm in diameter. Elevated temperature conditioning at 70°C increases the rate of diffusion of the blowing agent. Although there is little evidence from SEM of an increase in perforation of the cell membranes, there is some indication of an increase in wrinkling of the walls and intercell skeletal strands at elevated temperatures. Changes to the cell walls are possibly due to the beads softening and expanding at elevated temperature coupled with an increased partial pressure of the blowing gas. Although there is a possible risk of material change after accelerated conditioning at elevated temperatures, the thermal conductivity is unlikely to be affected as there was no significant change in the cell size. It takes longer to eliminate the pentane gas by conditioning at 23°C/50% RH but there is no risk of material change to the cells.
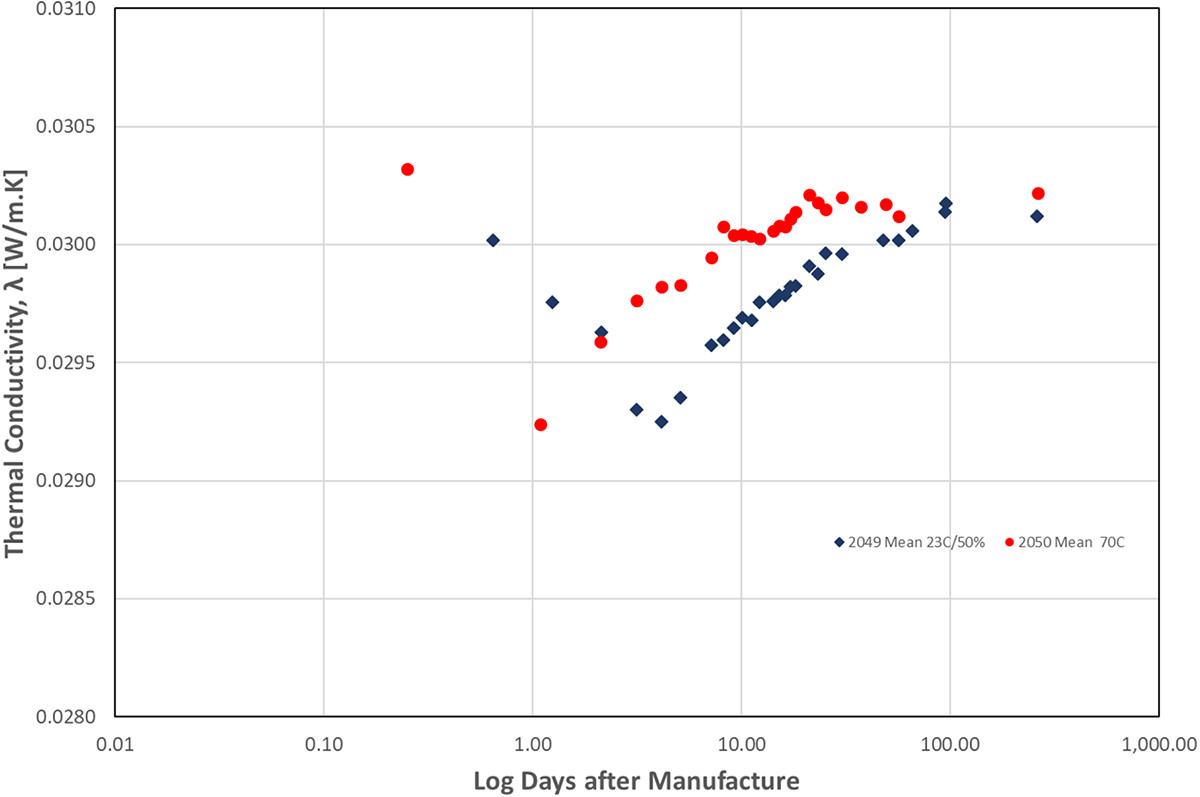
The time to an acceptable thermal conductivity level that is within 1% of the final value was found to be 5.1 days after conditioning at an elevated temperature of 70°C, whereas for conditioning at 23°C/50% RH the time taken was 23.1 days. The latter result was found to be in accord with the results of a separate programme of retesting retained grey EPS samples. Initial thermal conductivity tests 22 days from manufacture were found to be well within 1% of the final value after it was retested up to 9–12 years later.
In practice, EPS samples are likely to be tested at independent laboratories at least 3 weeks after manufacture, when the time taken to process, cut, deliver, prepare and condition to constant mass is included. It is suggested that measuring the thermal conductivity at least 3–4 weeks from a verifiable date of manufacture, after conditioning in the laboratory for at least 14 days at 23°C/50% RH (cf. EN13163:5.2), is an acceptable procedure for determining the final value that is constant throughout the economic life of the product, without material change to the EPS test sample.
Finally, it should be noted that industry has taken steps to clarify the situation regarding conditioning and reconditioning of EPS products at elevated temperatures, and a change of procedure is to be advised for the future. The apparent confusion around which conditioning temperatures are used has arisen due to changes in industry practice, such as the transition to improved EPS products containing IR absorbers. Details about these changes do not appear to be readily available in the public domain. EN standards and SG19 procedures are under review to resolve this, but it is expected that standards will be rewritten according to new requirements from the Commission and may therefore take some time to complete.