Introduction
Veno-venous (V-V) and veno-arterial (V-A) extracorporeal membrane oxygenation (ECMO) are increasingly being used as a last-resort mechanical support for patients with severe refractory respiratory and/or cardiocirculatory failure.
1 Despite this, mortality rates remain high up to around 50%, depending on ECMO mode and indication.
2 Although most deaths occur while on ECMO, a substantial number of patients decease after elective weaning of support.
2,3 The etiology of these late deaths, which are commonly referred to as the “ECMO gap”,
4,5 is multifactorial. Yet, it seems in part related to the occurrence of weaning-related shock (WRS), a phenomenon characterized by unanticipated and severe hemodynamic instability shortly after decannulation.
6,7,8Clinically, new-onset fever and vasoplegia are commonly observed in this context, leading to the hypothesis that new or worsening inflammation or infection may contribute to WRS.
6,7,9 Alternatively, weaning from V-A ECMO could unmask or provoke progression of underlying heart failure.
10 However, despite its great clinical significance, the etiology of WRS remains largely elusive.
4,5Improved understanding of the pathophysiological processes underlying WRS is essential to develop novel therapeutic targets as well as prognostic biomarkers. Furthermore, distinguishing between septic, cardiogenic, and hemorrhagic shock is essential to initiate an appropriate treatment response. In this exploratory study we investigated the potential etiologies of WRS by examining sequential plasma biomarkers reflecting inflammation, platelet–endothelial cell adhesion, and cardiac injury and overload responses during the period immediately before and after ECMO decannulation.
Results
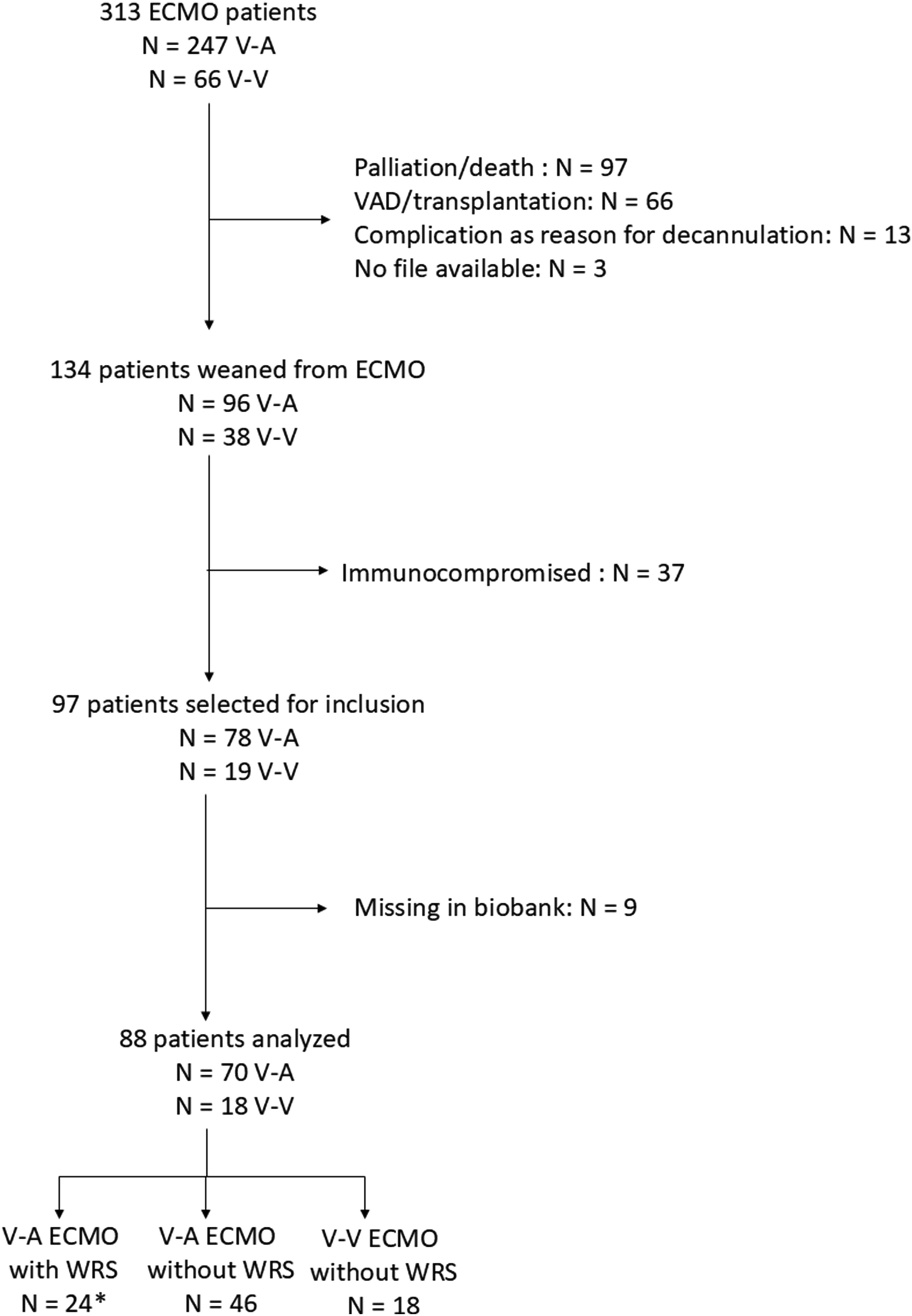
Among 97 patients eligible for study inclusion, 88 (91%) subjects were analyzed, including 70 V-A and 18 V-V ECMO cases (see flowchart
Figure 1). A total of 24 (27%) patients developed WRS, all within the V-A ECMO group. WRS occurrence in V-A ECMO patients was associated with higher ICU mortality (
n = 13, 54%) compared with both V-A ECMO patients without WRS (
n = 9, 20%) and V-V ECMO patients (
n = 1, 6%) (
p = 0.009 and
p = 0.004, respectively). Among those who ultimately died in the ICU, median time from ECMO weaning to death was 7 (IQR 4–12) days for WRS patients, compared to 15 (IQR 6–27) days for non-WRS patients.
Following clinical adjudication, WRS was believed to be attributable to hemorrhagic, cardiogenic, and septic shock in 0 (0%), 8 (33%) and 8 (33%) cases, respectively. The remaining 8 (33%) cases remained unclassifiable. V-A ECMO duration and cumulative fluid balance prior to weaning did not differ significantly between patients with and without WRS (
p = 0.21 and
p = 0.053, respectively). WRS occurred more frequently in older patients (
p < 0.01), those with diabetes mellitus (
p = 0.02) and in subjects who had received postcardiotomy ECMO support (
p < 0.01 (
Table 1). Among these postcardiotomy patients, WRS occurred in 3 of 8 patients (38%) who underwent isolated coronary artery bypass grafting surgery, and 13 of 25 patients (52%) who underwent valve, aortic root or combined procedures (
supplemental Table 1). Patients with WRS more frequently received inotropic support at the time of decannulation than those without WRS (dobutamine use 36% versus 5%, and milrinone use 45% versus 19%, respectively.
In the first 72 h after decannulation, we observed an overall increase in CRP and temperature, but not leukocytes (
Figure 2). New-onset clinical inflammation (rise in CRP, leukocytes, and/or new fever) was observed in 66 (75%) patients overall, including 22 (92%) V-A patients with WRS versus 36 (78%) V-A patients without WRS (
p = 0.84), and 8 (44%) V-V patients without WRS (
p = 0.01). Among WRS patients, new-onset clinical inflammation occurred at similar rates across all subgroups of cardiogenic (
n = 6, 75%), septic (
n = 7, 88%) and unclassifiable shock (
n = 8, 100%) (
p = 0.32).
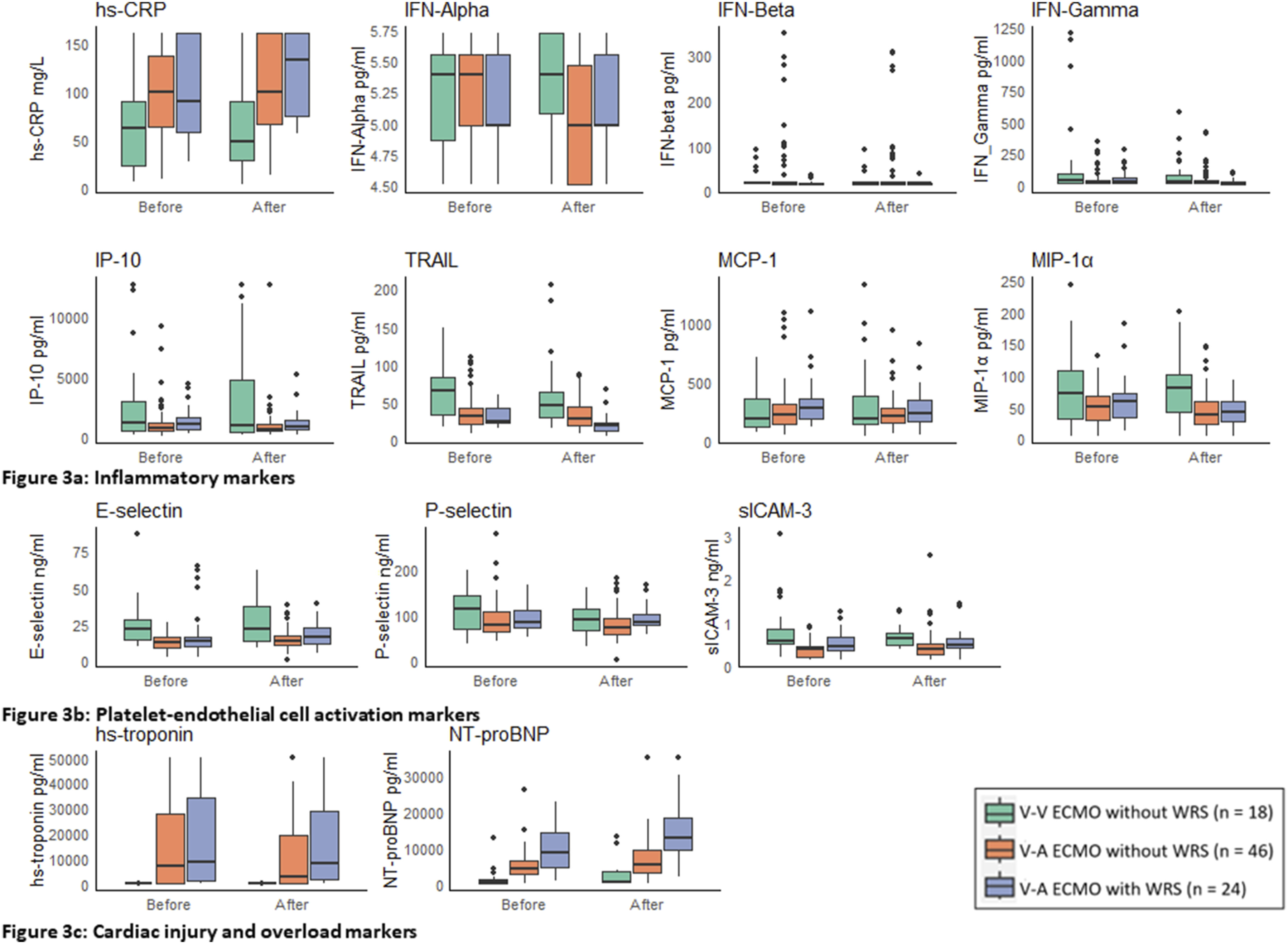
We observed elevated plasma concentrations for hs-CRP, IP-10, MCP-1, and P-selectin, and suppressed TRAIL levels compared to the reference ranges for these biomarkers in healthy individuals, at all timepoints, across all groups (
Figure 3(a)–(b)). However, we did not observe any differences in trends for these biomarkers over time when comparing patients with and without WRS (
p > 0.05 for all between-group comparisons)
. Likewise, elevated levels of HsTnI were observed in V-A (but not V-V) ECMO patients compared to reference standards in healthy individuals. However, no differences in HsTnI trajectories between patients with and without WRS were observed after decannulation (
p > 0.05 for all between-group comparisons) (
Figure 3(c)). In contrast, NT-proBNP levels rose significantly more after decannulation in WRS patients compared to V-V and V-A ECMO patients who did not develop WRS (
p = 0.003 and
p = 0.02, respectively). However, NT-proBNP values were already higher in WRS patients prior to decannulation (
p < 0.001 for all between-group comparisons) (
Figure 3). These observations were robust in a sensitivity analysis that excluded patients who already had established AKI at time of decannulation (
Supplement Figure 2). Strikingly, the rise in NT-proBNP levels was most pronounced not in patients with clinically suspected cardiogenic shock, but in those with presumed septic or unclassifiable shock (
Supplement Figure 3). After introduction of weaning protocols in 2018, there was a significant decrease in cumulative fluid balance prior to weaning (median of +845 ml before 2018 and a median of +85 ml thereafter,
p = 0.04), but no significant differences in the incidence of WRS (
Supplement Table 2,
p = 0.1).
Discussion
A significant proportion of patients who are being weaned from ECMO develop unanticipated WRS after decannulation, with older individuals and those supported with V-A ECMO following cardiac surgery or acute myocardial infarction being particularly at risk. Although subsequent fever and CRP responses could suggest that many of these WRS cases may be related to new-onset inflammation, this notion is not supported by the observed temporal dynamics of inflammatory and platelet-endothelial cell adhesion markers in our study. Instead, most individuals exhibiting WRS displayed markedly elevated NT-proBNP values before decannulation, with further increases thereafter. This finding suggests cardiac overload as a main determinant of WRS.
In our study, 75% of weaned patients exhibited clinical signs of new-onset inflammation, such as increasing CRP, leukocyte counts, or fever, following decannulation. Interestingly, while this was most common in WRS patients (92%), similar inflammatory responses were observed in both V-A and V-V ECMO patients without WRS (78% and 44%, respectively). These findings are consistent with existing literature, which report that up to 60% of weaned patients display symptoms such as fever and leukocytosis.
7,17,18 Notably, despite the increase in clinical signs of inflammation in weaned patients, there was no correlation with rising levels of inflammatory biomarkers after decannulation in our study.
This observation can be explained in several ways. First, CRP is a nonspecific marker involved in the acute phase response to a multitude of stimuli and clinical conditions.
19,20 The lack of significant changes in key inflammatory cytokines typically associated with infection, such as the interferons involved in immune responses against intracellular pathogens, suggests that factors other than infection are driving the observed increase in CRP. Second, the increased incidence of fever after decannulation does not necessarily indicate inflammation, as ECMO actively maintains body temperature around 36°C. It has been postulated that a shift in hypothalamic setpoint after a period of active temperature management during ECMO support may explain the elevated temperatures observed post-decannulation.
9 However, others argue that active cooling does not directly affect the thermoregulatory center but rather triggers defensive mechanisms that generate and conserve heat.
21 It is possible that these compensatory responses persist after decannulation, resulting in fever without an inflammatory or infectious cause. This phenomenon bears similarities to fever observed after targeted temperature management for cardiac arrest, known as rebound hyperthermia.
22,23 Although several theories have been proposed to explain rebound hyperthermia, the underlying pathophysiological mechanisms remain unclear.
22,24,25The most striking observation in our study was the highly significant increase in NT-proBNP concentrations in patients with WRS. This finding suggests that increased myocardial loading conditions after ECMO decannulation are an important contributor to its development. Notably, this rise was most pronounced in patients who clinically appeared to have septic, rather than cardiogenic, shock. Patients with limited myocardial reserve may be particularly susceptible to the adverse effects of inflammation on cardiac function once circulatory ECMO support is discontinued.
20 Indeed, circulating proinflammatory cytokines have been shown to exert negative inotropic effects on cardiac myocytes by altering intracellular Ca
2+ homeostasis.
26,27 Moreover, increasing temperatures and/or the development of fever post-decannulation can exacerbate heart failure by increasing heart rates and metabolic demands.
28In contrast to NT-proBNP, there was no significant surge in troponin levels in V-A ECMO patients with WRS compared to V-A ECMO patients without. This suggests that myocardial loading, but not injury plays a key role in WRS. The release of troponin could be the result of increased inflammation and coagulation, as studies indicate that troponin release is common in patients with activation of these pathways.
29,30The intricate interactions between inflammatory processes, increased cardiac wall stress, and cardiomyocyte dysfunction complicates the determination of their precise contributions to WRS development.
20 However, in the absence of clear differences in measured biomarkers of inflammation and platelet-endothelial cell activation between patients with and without WRS, the observed change in NT-proBNP concentrations suggests that it is not inflammation, but cardiac overload what distinguishes patients with WRS from those without. Moreover, previous studies did not find new-onset inflammation after decannulation to be associated with increased mortality.
7,9,17 This substantiates the notion that the high mortality rates observed in WRS patients are primarily due to cardiac failure.
Patients who developed WRS exhibited significantly higher NT-proBNP values even before decannulation, as well as a (non-significant) trend towards higher fluid balances. This could indicate suboptimal timing of ECMO decannulation in some. Indeed, others have previously suggested that NT-proBNP could be a valuable prognostic for evaluating V-A ECMO patients prior to weaning.
31 Moreover, the increased use of inotropic support prior to decannulation among WRS patients may reflect a more limited myocardial reserve which, together with the vasodilatory effects of these drugs, warrants particular attention during weaning decisions. Currently, there are no standardized guidelines for assessing cardiac function after ECMO decannulation.
32 Consequently, cardiac biomarkers and echocardiographic parameters are not systematically monitored, even though CRP, leukocyte counts, and temperature are evaluated daily. This may lead physicians to disregard potential cardiac dysfunction when clinical signs of inflammation arise. Therefore, we stress the need for prompt evaluation of cardiac function and volume status in patients who develop WRS even if clinical signs suggest an inflammatory cause for hemodynamic instability.
Our study has several limitations. The sample size was low, specifically in the V-V ECMO group, and the patients heterogeneous, which may limit generalizability of our findings and underscores the need for larger, multicenter studies to confirm the results of this exploratory study. Furthermore, patients were enrolled over a 12-years time-period during which ECMO care evolved considerably, including a growing use of percutaneous closure devices for decannulation and the adoption of stricter weaning protocols from 2018 onwards. Following these changes, we observed a significantly reduced cumulative fluid balance and a non-significant trend reduction in WRS incidence after decannulation. Since our findings suggests that cardiac overload contributes significantly to WRS development, future weaningmight be further improved by incorporating comprehensive hemodynamic monitoring data (e.g., pulmonary artery catheter measurements, central venous pressure and echocardiographic data). Lastly, sample degradation over time may have affected biomarker stability, potentially influencing the findings.
33 However, limited knowledge exists on this issue,
34,35 and duplicate measurements for each timepoint partly mitigated these effects.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DD reports research cooperation with Getinge-Maquet Critical Care AB, Solna, Sweden and Sonion BV, Hoofddorp, The Netherlands; DD reports research consultancy for HBOX Therapies, Aachen, Germany, DD reports educational consultancy to Abiomed, Aachen, Germany. All financial benefits are paid to the University of Twente, Enschede, The Netherlands, no personal fees are received. OC received in kind and in cash contributions to research from ImmuneXpress Inc., Seattle, WA, Abionic SA, Epalinges, Switzerland, Prolira BV, Utrecht, Netherlands, and Presymptom Health Ltd., Porton Down, UK. For the remaining authors, no conflicts of interest were declared.