Introduction
The infra zygomatic crest (IZC) bone screws are effective skeletal anchorage devices used in orthodontics for en-mass retraction, intrusion, and distalization of the maxillary arch. It is also effective in treating borderline surgical cases to nonsurgical ones without compromising the results. However, IZC screws pose a few challenges, most common being uncontrollable soft tissue inflammation, gingival overgrowth on the screw, and early loosening of the screw.
1According to a study, 43% of individuals with implants suffer from peri-implantitis.
2 Peri-implant inflammation caused by plaque retention leads to mobility and loss of the orthodontic bone screws. In a study, Sato et al.
3 reported that an anaerobic environment in crevices around orthodontic anchor plates is favorable for the growth of anaerobes. The knowledge of quantitative and qualitative aspects of predominant microorganisms present in the peri-implant region is important for reducing inflammation.
4 Reports suggest gram-negative obligate anaerobes are predominantly comprised of bacterial flora in peri-implantitis.
5In the recent past to reduce or nullify the adhesion of microbes onto the surface of implants many methods like surface treatments such as polishing, coating, and modification of surface free energy, have been employed.
6In this regard, silver nanoparticles are of prime interest in surface coating procedures due to their high antimicrobial actions against both gram-positive and gram-negative bacteria, viruses, and other eukaryotic microorganisms compared to other metal nanoparticles.
7 Evidence from previous in-vitro
6, 8 and in-vivo studies
9–11 on AgNP-coated titanium mini-implant, orthopedic implants as well as stainless steel (SS) orthodontic brackets, and orthodontic band cement have confirmed that AgNP gives antibacterial and anti-adhesive characteristics. However, there are no reported studies comparing success rate as well as microbiota between AgNP-coated and uncoated SS IZC bone screws on clinical application. SS is the material of choice for bone screws rather than titanium or titanium alloy because of its toughness when placed in the cortical bone.
12 In the current literature the controversy remains regarding the effectiveness of in-vitro studies because of various limitations like the inability to reproduce oral environment, variation in bone density, and also various other hormonal and systemic conditions which will influence the outcome of the study.
13Therefore this study aimed to surface coat SS IZC screws with AgNP and evaluate the success rate as well as microbiota of silver nanoparticle coated and uncoated infra-zygomatic crest (IZC) screws on clinical application. The study proceeded with the assumption that there was no difference in success rate between AgNP-coated and uncoated IZC screws on clinical application.
Materials and Methods
The study was approved by the Institutional Ethical Committee (No:110/2017-18) to compare the success rate for AgNP-coated and uncoated IZC screws. This double-blind split-mouth study included 18 patients (4 male, 14 female; mean age 22.58 ± 3.52) undergoing fixed orthodontic treatment with MBT mechanotherapy (3M Unitek, 0.022″ slot) requiring bilateral IZC anchorage to correct Class II malocclusion. The patients were selected based on the following selection criteria, after obtaining informed consent:
•
Cases with congenitally missing third molars and/or samples where the third molars were removed before the treatment were considered for the study.
•
Patients with good oral hygiene and no evidence of periodontal bone loss after radiographic examination with post-alignment overjet of 4.8±0.6 (mm) and overbite of 5.8 ± 0.9 (mm).
Exclusion criteria include:
•
Subjects with systemic disease,
•
History of antibiotics intakes 3 months before collection of IZC screws.
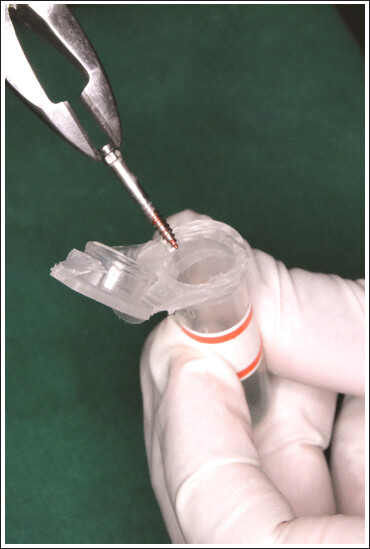
2 × 14 mm stainless steel IZCs (FavAnchor
TM SAS, S.H. Pitkar Orthotools Limited, Pune, India) (
Figure 1) were placed bilaterally in the IZC area. A total of 36 IZC screws were used in this study. A power analysis was established by G*power, version 3.0.1 (Franz Faul Universitat, Kiel, Germany). With a 95% confidence level and power of test as 80%, where C = 0.5* In[(1 + r)/(1–r)]=0.354, considering the mean difference of 27.9 and standard deviation of 3.01, the calculated sample size came up to 18 per group.
Group I-silver nanoparticle (AgNP) coated (n = 18) and group II-uncoated (n = 18). Pairs of coated and uncoated screws were coded for the right and left sides and random allocation was done to ensure that equal numbers of each type were tested on the right and left sides in a uniform manner.
Coating of IZC screws was carried out using magnetron sputtering (Radio Frequency—ANELVA SPF-332H) at CeNSE, IISc-Bengaluru, India. Scanning electron microscope (SEM; JEOL - IT 300, IISc, and Bengaluru, India) with an energy-dispersive spectrometer (EDS) was used to assess the surfaces of the coated IZC screws.
Discussion
In the present study, silver nanoparticles were deposited onto the surface of 2×14 mm SS IZC screws using Radio Frequency magnetron sputtering. To increase the adhesive strength post-deposition heat treatment was done as AgNP particles were deposited onto a smooth, machined SS surface where adhesion was a big concern.
15 The EDS microanalysis confirmed that a higher atomic % (11.89%) of AgNPs was obtained by magnetron sputter coating.
During the clinical procedure, the IZC screws were initially autoclaved
15 and insertion was done under local anesthesia following which immediately loaded with an e-chain. The findings of the present study showed an overall success rate of 63.9% which was slightly lower when compared to previous studies by Chang et al.
12 and Uribe et al.
16 who had reported 78.2% and 93% success rates, respectively.
The stability of these IZC screws seems to be affected by bone stiffness and bone density.
17 Most of the patients in this study were in the growing age group. Chen et al.
18 reported that screw stability primarily depends upon the mechanical interlock between the alveolar bone and the screw. Therefore, cortical bone density and thickness are important factors in the success of IZC screws.
The split-mouth design success rate for AgNP coated and uncoated groups were 77.80% and 50%, respectively. A key advantage of this split-mouth design is that the sample size requirement is approximately half that of a parallel-group design when all other parameters are equal. This is because each patient acts as his/her control, so much of the inter subject variability is removed, resulting in increased study power or a decrease in the number of participants required compared with a study in which patients receive only one intervention.
19 The higher success rate for the AgNP-coated group when compared to the uncoated group may be mainly due to the antibacterial effect of AgNPs. This nanometal attaches to the surface of the bacterial cell membrane and drastically disturbs its permeability and respiration and thus has high antibacterial actions.
20 Binary logistic regression analysis shows no statistically significant association between age, sex, and site with success and failure of AgNP-coated and uncoated IZC screws. However, the present study sample size is small and statistically insignificant to evaluate and confirm the same.
This is probably the first in-vivo study that compared the efficacy of AgNPs coated and uncoated SS IZC screws, whereas the previous study was in-vitro and has shown similar findings.
6T-RFLP analysis, a DNA-based nonculture-dependent approach was used in this study for molecular analysis. This approach helped in the rapid assessment of the bacterial colonization around the AgNP-coated and uncoated IZC screw surfaces.
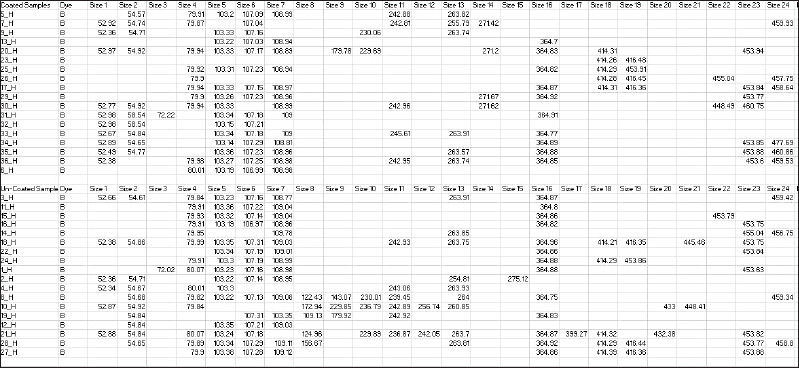
14 In the study TRFs detected per sample ranged from 2 to 12 in group I and 4 to 15 in group II. Among these, the TRF with a length of 107 base pair (briefly TRF107) was the most frequently encountered (88.9%) across samples followed by TRF103(86%), TRF109(80.5%), TRF80(66.7%), TRF365(63.9%), TRF55(55.5%) and TRF454(41.7%). These values indicate that TRFs were statistically significantly distributed between the 2 groups.
TRF107 and 103 were the most frequent species in the uncoated group while in the coated group TRF107 and 103 were counted 15 times (out of 18). This indicates TRF107 and TRF103 were reproducibly present across 2 groups signifying the reproducibility and sensitivity of the T-RFLP protocol followed in the present study. T-RFLP data analysis results also showed that few peaks were present only in uncoated samples. These peaks included TRF122, TRF124, TRF125, TRF157, TRF144, TRF229,TRF237, TRF256,TRF254, TRF457,TRF278,TRF399.TRF433,TRF445,TRF255,TRF261,TRF239,TRF432,TRF453. Also, a few other peaks were found only in AgNP coated group which included TRF461, TRF460, TRF457, TRF478, TRF246, TRF271, and TRF272. The result suggests that the protocol was sensitive enough to identify the peaks.
The T-RFLP analysis in the present study revealed a statistically significant difference in the TRF value between AgNP-coated and uncoated IZC screws. The mean rank of TR-Fs was lower in AgNP coated group (14.92%) compared to the uncoated group (22.08%). The reason may be with higher atomic % of AgNP deposition on the surface of IZC screws. This finding was similar to an in-vitro study by Venugopal et al.
6 who concluded that the biopolymer modified AgNP coated titanium screws with higher atomic% exhibited greater antibacterial effects with clear zones of inhibition.
PCR-cloning sequencing protocol was used in the present study to identify the predominant species within the microbial population present between a coated and uncoated sample. Prevotella oris strain NCTC13071, Uncultured prevotella sp, and uncultured bacterium were predominant species in failed AgNP coated sample, while Capnocytophaga leadbetteri, Capnocytophaga sp. Uncultured bacterium clone, Uncultured bacterium clone 069096_35 was the predominant species in the failed uncoated sample.
Here the term “unculturable bacteria” is used to denote uncharacterized organisms that cause oral infections.
21 The predominant species detected in the present study might have been overlooked in an in-vitro culture as 50% of the oral flora is unculturable. The nonculturability in vitro may be due to the disruption of bacterial cytokine networks. Bacterial cytokines are thought to be mediators of bacterial signaling and may be particularly important in coordinating the growth of component organisms in bacterial biofilms such as dental plaque.
22This underlines the usefulness of nonculture-dependent molecular techniques (16S rDNA PCR and sequencing) used in the present research for studying samples. Apart from that, the predominant species identified in failed samples, both AgNP coated and uncoated, were found to be gram-negative. This result was found to be similar to that described in the earlier report by Kumar et al.
23 wherein using pyrosequencing, the investigators had confirmed that the failed IZC screws were dominated by gram-negative bacteria. AgNPs were found to have a stronger antibacterial effect on gram-negative bacteria compared to gram-positive bacteria.
8 Thus AgNPs incorporation around IZC screws would impart stronger antibacterial efficacy ensuring long-term stability and success.
In future studies more elaborate protocols of the Next Generation Sequencing based methodology may be employed. Through that we may effectively sequence and identify the complete metagenome of all organisms present. This would effectively find all minor species present or absent within samples. Also, the advantages of biogenic metallic nanoparticles over physio-chemically obtained nanoparticles may be considered for future research.
We had considered cases with congenitally missing third molars or samples where third molars were extracted before the orthodontic treatment in our study but standardization of the cases would be necessary to avoid selection bias in studying the antibacterial efficacy of AgNPs coated and uncoated IZC screws. This would be considered a limitation of our study.