Introduction
Periodontitis is associated with chronic inflammation characterized by a complex interaction between periodontopathic bacteria and the host immune response, which results in the release of pro-inflammatory mediators that lead to the destruction of soft and hard structures that support the teeth.
1,2 Periodontitis affects nearly 15% to 50% of the global population, and it is more prevalent in the elderly (82%), compared to the younger population (59%).
3,4 Periodontitis is complex, involving the secretion of cytokines (IL-1β, IL 6, IL-17, TNF-ɑ), chemokines (IL-8, MCP-1, MIP1α, RANTES), prostaglandins, proteolytic enzymes (MMPs), elastase, mast cell tryptase, dipeptidyl-peptidase, plasminogen activators, lysosomal cysteine proteinases, cathepsins, and protease. All of these mediators contribute to further degradation of the periodontal tissue and resorption of alveolar bone by several mechanisms.
5 The subgingival plaque contains more than 700 species of bacteria that are related to this pathological condition.
6 Porphyromonas gingivalis,
Tannerella forsythia, and
Treponema denticola are categorized as red complex bacteria, which play a significant role in periodontal disease progression.
7 Meanwhile, the orange complex bacteria which includes
Prevotella intermedia, Prevotella nigrescens, Prevotella micros, and Fusobacterium nucleatum are considered as etiological agents that, in the course of the disease, lead to a shift toward the red complex bacteria.
8 Besides these complexes, a member of the Pasteurellaceae family,
Aggregatibacter actinomycetemcomitans also has a significant role in localized aggressive periodontitis.
9The physiological and pathological aspects of periodontal disease have been studied more widely in rats, such as the laboratory rat (
Rattus norvegicus), which belongs to the Muridae family, than in other rodents.
10 There are several inbred strains commonly used in periodontal research, such as the Wistar, Sprague Dawley, Lewis, Norwegian Grey, and Rice rats.
10 Anatomically, rodent dentition consists of 1 rootless incisor and 3 molar teeth in each quadrant.
11 Rodents share several anatomical, bacterial, and periodontal pathogenic features that resemble those found in humans, and the alteration of their genome permits the researcher to study various hypotheses.
12 They share similar anatomical structures in the molar area with human teeth, which includes a shallow gingival sulcus and junctional epithelium attachment with the tooth surface that serves as a pathway for foreign substances, exudation of inflammatory cells, and bacterial endotoxins. However, there appears to be a difference in the keratinization of the crevicular epithelium and the relationship between gingival and junctional epithelium with desmosomal contact between the most superficial cells.
13,14 When compared to mice, accessibility to the molar teeth is better in rats due to their bigger body size. Rats are used in studies of gene expression, mechanisms of inflammatory regulation, resolution, and healing processes in periodontal and peri-implant research. In addition to studies involving teeth, rodents are considered ideal candidates for investigating the relationship between soft and hard tissue associated with inflammatory conditions.
15,16In the rat model, periodontitis is mainly induced by 3 widely accepted methods: placement of ligature by suture or wire around the maxillary or mandibular teeth, oral bacterial inoculation, and lipopolysaccharide (LPS) injection.
6,17 The ligature method in the rodent was first described in 1966,
18 while oral inoculation with
P.
gingivalis model was developed in 1994
19 to induce alveolar bone resorption.
20 The ligature acts as a gingival irritant that induces the accumulation of plaque and bacterial colonization, which leads to epithelial migration and tissue destruction.
21 The oral bacterial inoculation model uses a predefined number of bacteria that are inoculated orally by gavage or topical application.
6 On the other hand, LPS injection induces rapid cytokine release from inflammatory cells, osteoclastic-cell differentiation, and alveolar bone resorption.
22The aim of this review is to demonstrate the various methods of induction used for experimental periodontitis in rats and to highlight their advantages and disadvantages in order to guide researchers in selecting the most effective methods in future study designs.
Materials and Methods
A narrative literature search was conducted on published research articles from the previous 15 years on PubMed, Google Scholar, and Semantic Scholar. The following terms were used: periodontitis induction on a rat model; periodontitis induction on an animal, periodontitis induction on a laboratory animal; periodontal disease on animals; periodontal pathogen. The authors used the following inclusion criteria to select the studies: periodontitis induction by ligature method, oral bacterial inoculation, LPS injection, experimental periodontitis on rats, periodontitis on laboratory animal models, and periodontal pathogens. The exclusion criteria were clinical studies, case reports, comments, and any literature with data that could not be extracted.
Periodontitis Induction by Ligature Method
The ligature method is one of the more commonly used procedures for the experimental induction of periodontal disease in rats.
23 The placement of a ligature, preferably using a suture or wire, acts as a local irritant, which contributes to the accumulation of dental plaque and ulceration of the sulcular epithelium, which then enables the invasion of periodontal pathogens into the connective tissue.
24 Bacterial plaque formation due to ligature placement initiates gingival inflammation, which leads to periodontal tissue destruction and initiates alveolar bone resorption, which is similar to what happens in humans.
24 However, some researchers claimed that the absence of bacteria in the ligature precludes periodontal disease, and in such cases, bone loss has been observed as a result of direct mechanical trauma.
23,25 In response to this argument, studies have shown that the orthodontic wire works as a plaque retentive factor to initiate gingival inflammation.
21 Recently, a study showed that the placement of orthodontic wire causes the accumulation of endogenous oral microbiota, which then leads to gingival inflammation and alveolar bone loss.
26 However, in another study, it was shown that no gingival tissue or bone damage was observed in the micro-CT image after 24 h of ligature wire placement, which indicates low possibility of mechanical trauma induction.
26 Nevertheless, in the ligature model, the bone destruction process is rapid, severe, and usually observed to be greater than in other areas. Thus, several researchers have considered the ligature technique to be an acute model of periodontal disease induction.
25,27Non-absorbable sterile 2/0
a, 3/0
b, 5/0
c cotton, 4/0 silk
d and nylon sutures are usually used, and these are placed around the maxillary or mandibular teeth in the suture-ligation technique. Upon placement, the suture is applied and tied gently to prevent damage to the periodontal tissue.
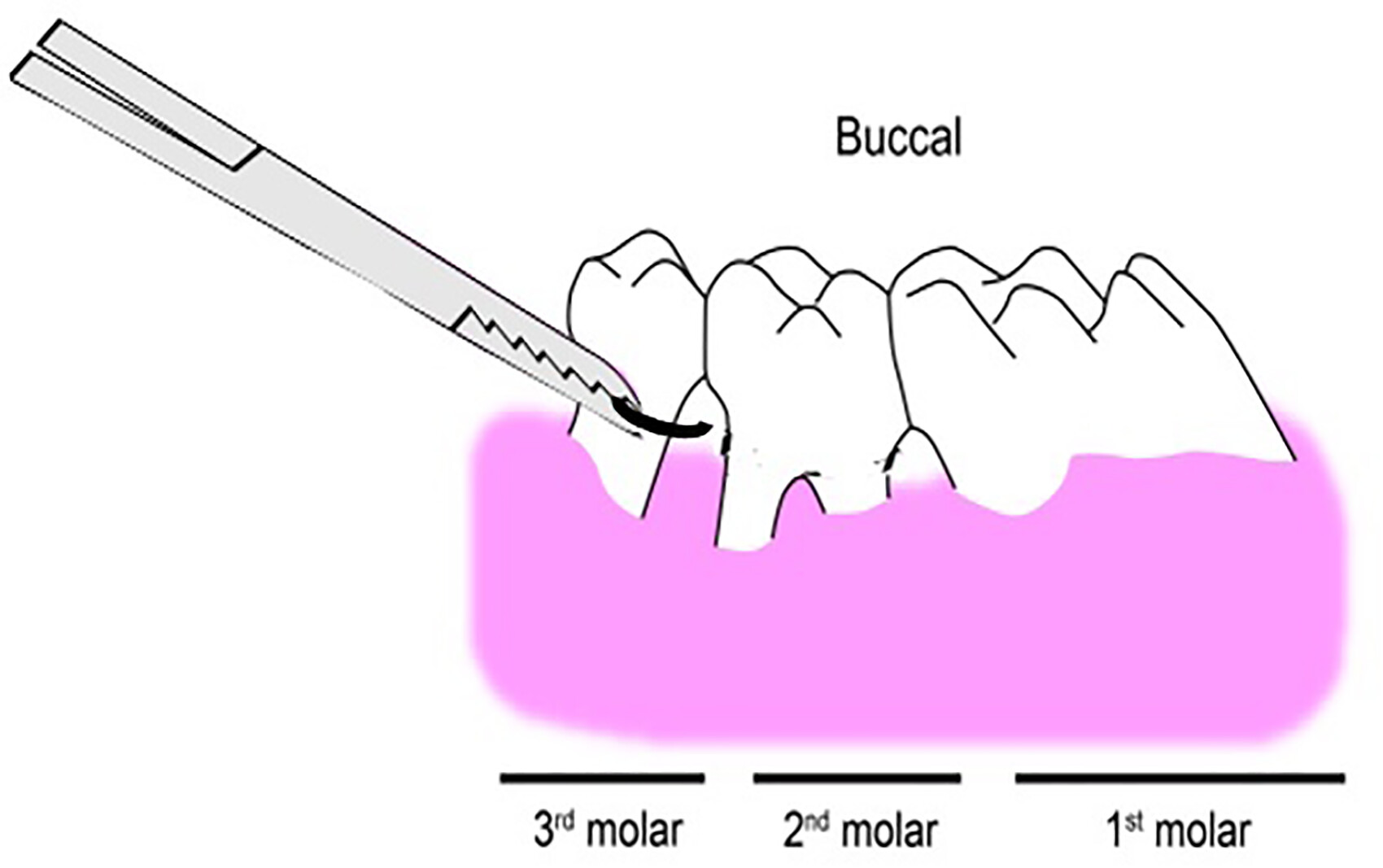
21 Usually, a ligature is placed through the proximal contacts of the selected tooth (
Figure 1a). The suture is passed around the crown of the tooth sub gingivally (
Figure 1b) and tied firmly with a double knot on the buccal side, labial side, or mesial side.
28 The ligature method is the most popular for experimental periodontitis induction because it is inexpensive and suture materials are easily available. Moreover, periodontitis can be promptly induced in a specific site of the oral cavity.
29 However, special skill is needed to apply this method on rats as inserting the ligature into the interproximal spaces between rat molars can be challenging due to the small size of the tooth, limitation in viewing the sulcus region, and the tiny oral space in which to perform knotting and insertion. Additional instruments are also needed, for example a mouth gag, to keep the mouth cavity open for ligature placement. A major drawback of this technique is that the suture is easily torn due to the force applied to place the ligatures. Moreover, frequent monitoring is needed because it could be easily displaced due to tongue movement during mastication. While the ligature method by suture is still commonly used, several studies have recently focused on using orthodontic wire instead of suture. Orthodontic wire (0.2 mm
e, 0.4 mm
f, 0.5 mm
f) can be placed into the interdental space by using a needle holder and forceps (
Figure 2). Before insertion of orthodontic wire, it is necessary to make a space in the interdental area to prevent damage to the gingival sulcus. A study has demonstrated a modified method for wire placement, where #8 and #10 endodontic files
g are bent 90° from the tip and inserted carefully into the interdental area by push and pull movements to make a space for wire insertion. Compared to passing the sutures into the interdental space, the wire can be easily inserted into a similar site.
26 Orthodontic ligature wire is also economical, feasible and can be placed by a single person within a short period of time. Moreover, the wire can be clamped with the tip of the needle holder properly and is less likely to be displaced by tongue movement. Also, less technical skill is required for wire placement, and maintaining position of the wire is much easier compared to using a suture. However, the wire needs to be carefully inserted, as any excessive force could damage the gingival tissue and lead to bleeding.
Table 1 shows the different ligation techniques to induce experimental periodontal disease in rats.
Periodontitis Induction by Oral Bacterial Inoculation
In the oral inoculation method, monomicrobial or polymicrobial mediums are used for periodontal pathogen inoculation.
38 In this method, individual isolates of
P. gingivalish,i,j, A. actinomycetemcomitansk,l, F.
nucleatumm, T. forsythian, and
T. denticolao or a combination of different bacterial strains are commonly used for bacterial inoculation.
6 Among these bacterial species, inoculation of
P. gingivalis alone or in combination with other bacteria is considered a significant causative agent for the oral dysbiosis process because of its strong ability to initiate periodontal bone destruction.
39 This bacterium can locally invade periodontal tissue and is able to affect the host cell, immune cells and the cells responsible for bone remodelling.
40 It can survive in the oral environment via multiple virulence factors, which are fimbriae, gingipains, LPSs, capsules, and hemagglutinins.
40In the polymicrobial inoculation model, greater bone loss is observed compared to the monomicrobial inoculation model. A previous study demonstrated that polymicrobial infection by
P. gingivalis and
T. forsythia significantly increased maxillary, mandibular, and total vertical alveolar bone loss compared to monomicrobial infection.
41 Moreover, marked apical migration of junctional epithelium and enhanced alveolar bone destruction were observed in that previous study.
42 Another study reported that mixed inoculation with
P. gingivalis and
T. denticola induces marked interproximal as well as horizontal alveolar bone resorption compared to monomicrobial infection.
43 Meanwhile, a mixed oral infection model with
P. gingivalis,
T. forsythia and
T. denticola demonstrates a lower serum IgG level compared to oral monomicrobial infection, which suggests alteration of host responses, which is due to the evasion of protective immune responses. In addition, a significant amount of maxillary and mandibular bone resorption was reported by this bacterial consortium.
44 As shown in
Table 2, high bacterial concentrations were used, which varied from 10
8 to 10
12 cells/ml, depending on the preparation route, either mixed with 2% carboxymethylcellulose or phosphate buffered saline and administered via food and drinking water. This induction method does not require general anesthesia or a special operating procedure, which allows it to be performed by a single operator. In addition, minimal observation of the induction site is needed. However, one of the significant issues with this method is the complex bacterial interaction that can occur. For example, in the
P.
gingivalis monomicrobial infection, it was shown that this bacterium collaborates with other endogenous bacterial species and may modify the oral biofilm, which contributes to the periodontal destruction process rather than producing a direct effect on its own
.24 In addition, the induction period by this method requires a minimum of 4 weeks to produce some significant results.
In oral bacterial inoculation, the bacteria contributed to leukocyte migration into the gingival connective tissue. A study has reported that inoculation of bacteria induces migration of leukocytes (mononuclear cells and neutrophils) into the gingival connective tissue and apical migration of the junctional epithelium, as shown by histological examination (H&E staining).
49 An increased level of inflammatory cytokines, such as TNF-α, IL-12, IFN-γ, within the gingival tissue in response to oral bacterial inoculation was detected by another study via a real time PCR assay.
50 Moreover, research has shown that mono and mixed bacterial inoculation induces the secretion of IL-1beta, IL-6, IL-8, PGE2, MMP-9 in response to a bacterial challenge.
44Periodontitis Induction by Lipopolysaccharide Injection
LPS is a major constituent of the outer membrane of Gram-negative bacteria that is recognized by the immune system as a marker for bacterial pathogen invasion.
51 Several methods have been used for the isolation and purification of bacterial LPS, including trichloroacetic acid extraction at 4 °C, extraction in aqueous butanol, triton/Mg+2, cold ethanol, extraction in water 100 °C, phenol, chloroform, petroleum ether, and methanol.
52 Among these methods, hot phenol water and phenol chloroform extraction methods are widely used to extract LPS from various bacterial strains.
51 The LPS from periodontal pathogens is a potential endotoxin that provides persistent inflammatory stimulation.
53 It is a significant stimulus that triggers a natural immune response when injected into the gingival tissue.
24 Many studies have demonstrated that bacterial LPS induces the expression of inflammatory cytokines and initiates osteoclastic activity, thus leading to bone destruction, which is the key feature of periodontitis.
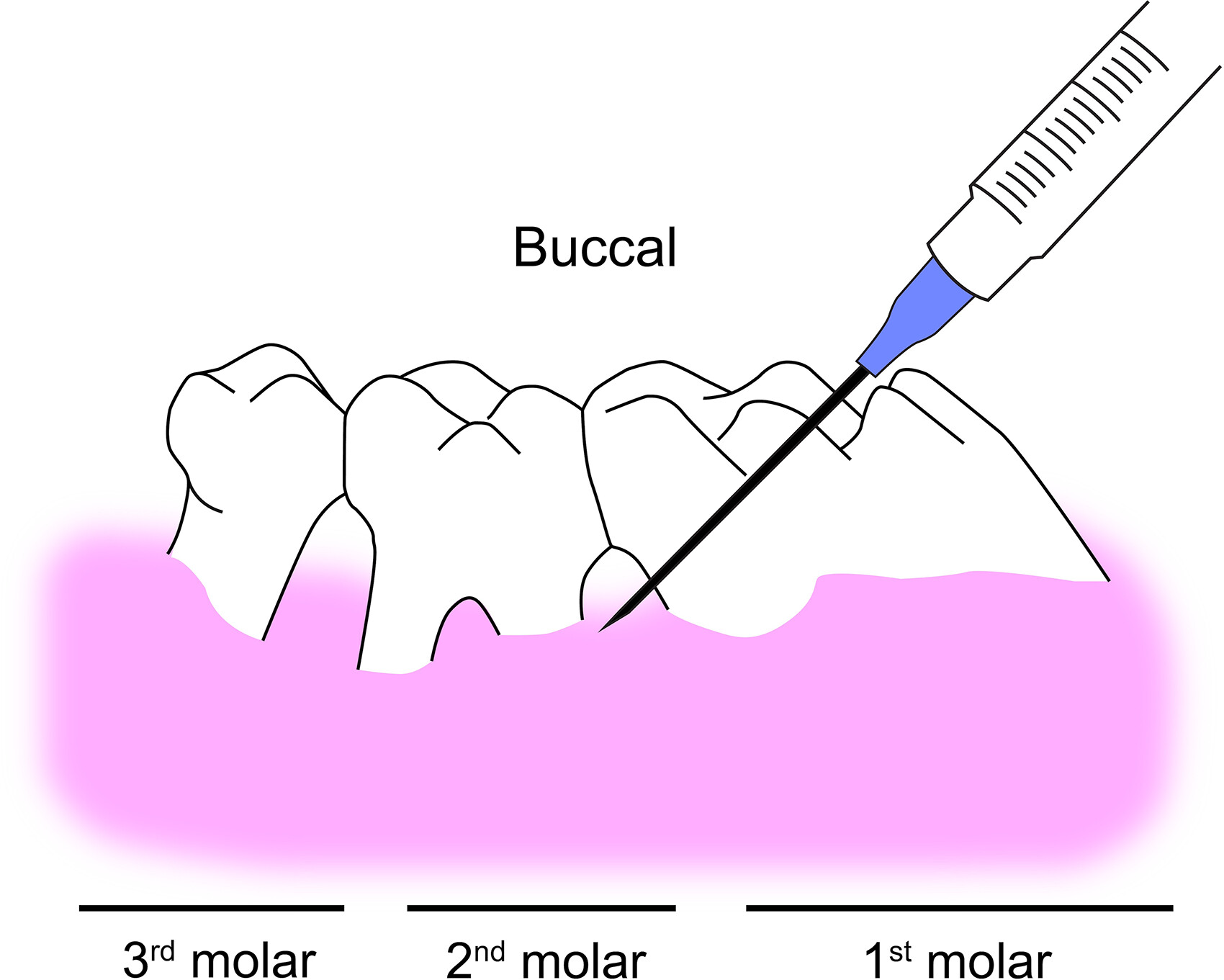
22In this technique, a micro syringe
p (28-33G) was used to inject a measured amount of purified bacterial LPS suspended in microvolumes (1-10 μl) into the gingival tissue (
Figure 3).
24 Preferred injection sites can be the palatal or lingual area of the molar region by approaching from the opposite direction for better access. The injection can be administered 1 to 3 times per week, depending on the experimental design. Bacterial LPS can be injected at single or multiple sites into the sulcus area based on the experiment protocol.
Inflammation and bone resorption have been observed after different induction times depending on the volume of bacterial suspension injected into the site, as shown in
Table 3. Several studies reported bone loss, which can be observed as early as 7 days after the first injection.
24 The most important advantage of the LPS injection method is that periodontitis can be induced within a short time due to the direct delivery of virulent pathogenic stimuli into the tissue. The method also allows greater control over induction. Moreover, inflammation and bone resorption in the targeted area can be achieved due to specific site administration of LPS.
Periodontal Disease in Other Animal Models
Periodontal disease is the most common disease in cats and dogs, with incidence rates of 70% and 80%, respectively, in 2-year-old animals.
58 Gram-negative bacteria, such as
A. actinomycetemcomitans, T. forsythia, Campylobacter spp., Capnocytophoga spp., E. corrodens, P. gingivalis, P. intermedia, and
T. denticola, can contribute to subgingival plaque formation.
59 Although there are many bacterial species found in both dog and human dental plaque, there are considerable differences. While humans are known to carry the catalase-negative bacteria
P. gingivalis, dogs are thought to have catalase-positive
P. gingivalis-like species, for example
P. canoris, P. salivosa, P. denticanis, and
P. gulae.
60 Interestingly, a study using next generation sequencing (NGS) has found that Gram-negative bacteria are the most frequent in dogs with healthy periodontal status, whereas Gram-positive bacteria are mostly abundant in periodontitis, which is the opposite of the finding in humans.
61 Moreover, although dogs may be an excellent model for studying experimental periodontitis due to their similar histological traits to humans,
61 their cost and animal care requirements make their use in periodontal research less feasible.
13 In cats, anaerobic bacteria, mostly from the genera
Bacteroides and
Fusobacterium, dominate the typical oral microflora. However,
Porphyromonas spp. have been isolated from the dental plaque of cats with periodontal disease.
58 Furthermore, similar to dogs, certain studies have found that around 75% of subgingival bacteria in cats with good oral health are Gram-negative bacteria. The percent of Gram-positive bacteria only increased when periodontal health deteriorated to the disease stage.
62 Another study has reported that
Prevotella intermedia, A. actinomycetemcomitans, Porphyromonas gulae, and
Prevotella nigrescens were shown to be substantially more common in cats than in dogs.
59In horses, the naturally occurring oral diseases include buccal abrasion, calculus, gingival recession, and periodontal pockets.
13 According to a recent equine survey, the prevalence of periodontal pockets and gingival recession is higher in older horses and mostly associated with other dental disorders and tooth loss.
63 Horses are not a feasible model for basic science research on periodontitis or the testing of prospective therapeutics due to husbandry constraints. Meanwhile, periodontitis has been induced in minipigs within 4 to 8 weeks by ligatures in combination with bacterial inoculations of
P. gingivalis, S. mutans, and
A. actinomycetemcomitans.
64 Minipigs can be suitable for periodontal as well as oromaxillo-facial investigations. However, minipigs are rather costly, and again, husbandry concerns make this model less suitable. Nonhuman primates have human-like oral structures and teeth, as well as naturally occurring dental plaque, calculus, oral microbial infections (eg,
P. gingivalis), and periodontal disease.
65 Although periodontitis in primates most closely resembles the human disease, the expense and special husbandry requirements for these animals limit their use in periodontal studies.
Conclusion
There are 3 common methods to induce experimental periodontitis in rats: the ligature method, oral bacterial inoculation, and LPS injection. Among these 3 methods, the ligature model for induction of bone resorption is the most widely reported and frequently used in periodontal research. This method is convenient, requires simple instrumentation, and no special procedure is needed. Moreover, the achieved amount of bone loss by ligature placement is greater than other experimental models, and periodontitis can be observed within a short period of time. Also, the ligature needs to be applied only once, though its position needs to be maintained throughout the induction period. The ligature model allows analysis of most aspects of periodontal disease, including bacterial interactions and dysbiosis, periodontal inflammatory responses, and bone destruction.
Based on this review, the authors recommend the ligature method, specifically by orthodontic wire, as the best experimental method due to its advantages. It is less expensive, can be inserted into the interdental space in a convenient manner, and is simple to maintain. Moreover, this method is more suitable for the maxillary area because of less tongue movement. More studies need to be done in order to improve this method and make it more beneficial for studying periodontitis using the rat model.