Normative Data and Diagnostic Cutoffs for Igg4-related Disease in North Indian Men
Abstract
Background:
Immunoglobulin G4-related disease (IgG4-RD) is a fibroinflammatory condition with multisystem involvement. Given the heterogeneity of clinical presentations and the limitations of tissue biopsy, there is a growing need for reliable, non-invasive biomarkers. Serum IgG4 has emerged as a valuable serological marker, but its diagnostic precision is limited by considerable variability.
Objective:
To establish normative reference ranges for serum IgG4 in healthy adult North Indian males and propose diagnostic thresholds for IgG4-RD.
Methods:
This single-centre, observational study enrolled 203 healthy North Indian males (aged 20–60) to establish age-stratified normative IgG4 intervals by nephelometry. Retrospective analysis of 20 biopsy-confirmed IgG4-RD cases enabled assessment of diagnostic performance and determination of optimal serum IgG4 cutoffs.
Results:
The mean serum IgG4 concentration in healthy individuals was 767.1±462.7mg/L, with a median of 678mg/L and an IQR of 411.0–1031.8 mg/L. Median IgG4 values ranged from 569 to 806mg/L, with no age-related trend. In contrast, patients with IgG4-RD had a significantly higher mean IgG4 concentration of 5,518.4±7,205mg/L (P = .01). ROC curve analysis demonstrated an AUC of 0.693 with 95% confidence interval [CI]: (0.56–0.83), highlighting only ‘moderate’ diagnostic accuracy. At a cutoff of 1262mg/L, serum IgG4 had a sensitivity of 70% and specificity of 58% for identifying IgG4-RD.
Conclusion:
Serum IgG4 levels exhibit wide interindividual variability but minimal age-related trends. A diagnostic threshold of 1262mg/L offers moderate accuracy for identifying IgG4-RD. These normative reference intervals enhance the diagnostic utility of serum IgG4 in clinical practice.
Introduction
Immunoglobulin G4-related disease (IgG4-RD) is a chronic, immune-mediated fibroinflammatory condition characterised by tumefactive lesions, dense lymphoplasmacytic infiltrates rich in IgG4-positive plasma cells, obliterative phlebitis and storiform fibrosis, often accompanied by elevated serum IgG4 levels.1 Although initially described in the context of autoimmune pancreatitis, it is now recognised as a multisystem disorder encompassing a wide spectrum of clinical presentations, often mimicking malignancy or infection.
With increasing awareness and improved diagnostic modalities, the recognition of IgG4-RD is steadily rising across specialities.2 Serum IgG4 is a well-established erological marker and forms an integral component of most diagnostic frameworks for IgG4-RD.3,4 Although numerous studies have demonstrated an association between elevated serum IgG4 levels and the presence of IgG4-RD, there is wide variation across different populations and clinical settings.5–9 Previous studies have shown that serum IgG4 concentrations are significantly elevated in patients with IgG4-RD, making it a valuable tool not only for diagnosis but also for assessing disease activity and predicting treatment response to steroids or rituximab.10,11 However, elevated serum IgG4 levels are not exclusive to IgG4-RD and can be seen in various other conditions like repeated infections, autoimmune diseases, malignancies, as well as in primary immune deficiencies and other non-IgG4-RD conditions,12 thereby reducing its specificity and positive predictive value despite its high sensitivity and negative predictive value.13
Establishing normative reference ranges and disease-pecific cutoffs for serum IgG4 is crucial to improving its diagnostic accuracy, particularly in differentiating IgG4-RD from conditions with overlapping clinical features. Despite the widespread use of serum IgG4 in diagnostic algorithms, current cutoffs are largely derived from small, organ-pecific studies with limited ethnic diversity, leading to inconsistent thresholds and diagnostic uncertainty. This highlights a significant research gap: the absence of well-defined, population-specific normative data and validated cutoffs for IgG4-RD diagnosis. Therefore, this study was designed to address this gap by establishing population-specific normative reference intervals for serum IgG4 in healthy North Indian males and to evaluate its diagnostic performance in biopsy-confirmed IgG4-RD.
Materials and Methods
This single-centre, prospective, non-interventional observational study was conducted at the Department of Endocrinology, in collaboration with the Department of Immunopathology and Histopathology, PGIMER, Chandigarh. Between January 2023 and June 2024, 203 apparently healthy adult males aged 20–60 years, who were attendants of patients with migraine or irritable bowel syndrome in the general medicine outpatient department (OPD), were recruited as the healthy cohort. These conditions were selected as index illnesses to minimise confounding from systemic inflammatory or autoimmune disorders. Participants were stratified into five-year age groups. Inclusion criteria included apparently healthy adult males providing informed consent. Exclusion criteria comprised unwillingness, medicolegal issues, known IgG4-RD, active infections, autoimmune or malignant disease within 5 years, and current use of glucocorticoids or immunosuppressants. Subjects with IgG4 levels >97th percentile were further investigated and reclassified accordingly.
erum IgG and IgG4 levels were measured using an Optilite nephelometer (The Binding Site Group, UK) with BS-NIA IgG1-4 antibodies. IgG4/IgG ratios were calculated. Percentile distributions (3rd, 10th, 25th, 75th, 90th, 95th, 97th) were used to define normative ranges. For comparison, retrospective data from biopsy-proven IgG4-RD cases were collected, including serum IgG4/IgG levels and organ involvement (IgG4-RD cohort). These served as a reference to construct receiver operating characteristic (ROC) curves and identify optimal diagnostic cutoffs.
The study was approved by the Institutional Ethics Committee (Intramural), Postgraduate Institute of Medical Education and Research (PGIMER), Chandigarh (Reference No. IEC-INT/2023/MD-1257), and was conducted in accordance with the principles of the Declaration of Helsinki. For the retrospective IgG4-RD cases, the requirement for written informed consent was waived by the Institutional Ethics Committee due to the retrospective nature of the study and use of anonymised data.
tatistical Analysis
ample size estimation for the control (reference) cohort was based on a precision-based single-proportion formula. Assuming an outpatient population of approximately 10,000 individuals, an expected prevalence of 10%, a 95% confidence level (Z = 1.96), and an absolute allowable error of 5%, the minimum required sample size was 137. Allowing for ~50% attrition, the target was set at 205, with 203 controls finally included.
The IgG4-RD group consisted of 20 consecutive, histologically confirmed cases available during the study period and was included for exploratory diagnostic performance (ROC) analysis.
Continuous variables were summarised as mean ± SD or median (IQR) depending on distribution (Shapiro-Wilk test). Percentile distributions (3rd-97th) were calculated to define normative IgG4 reference intervals, and the IgG4/IgG ratio was described using central measures and after harmonising units (mg/L). Categorical variables were expressed as frequencies and proportions and compared using the chi-square test. Group comparisons between cases and controls were performed using the t-test or Mann-Whitney U test as appropriate.
The diagnostic accuracy of serum IgG4 was assessed using ROC analysis. Area under the curve (AUC), sensitivity, specificity, and the optimal cut-off (determined by the Youden index) were estimated. ROC analysis was first performed using all cases and controls. To assess robustness, a sensitivity analysis was conducted by randomly selecting 20 cases on each occasion and repeating the ROC analysis. This random sampling was repeated 20 times, and the resulting AUC values ranged from 0.599 to 0.707. The resampled control sets yielded mean IgG4 ~1217.5 mg/L (SD 350.6) and mean age ~39.2 years (SD 10.2), consistent with the overall healthy cohort values (767.1 mg/L, SD 462.7; 41.0 years, SD 11.3). Statistical analyses were performed using SPSS Statistics version 22.0 (IBM Corp., Chicago, IL, USA), with a two-tailed P < .05 considered significant.
Results
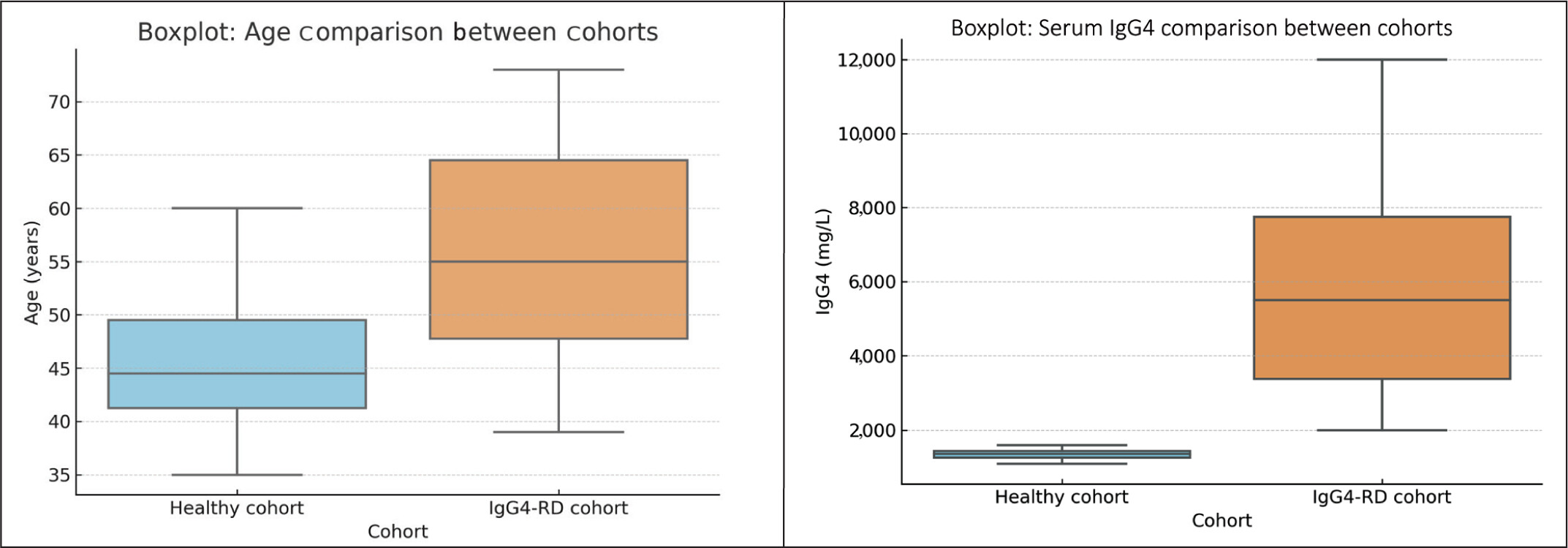
A total of 203 apparently healthy North Indian adult males aged 20–60 years were enrolled. The mean age was 41±11.3 years, with average height and weight being 172.0±6.8cm and 76.5±10.6kg, respectively. The mean BMI was 25.8±3.3kg/m2, placing the majority of participants in the overweight category. The mean serum IgG4 concentration in this healthy cohort was 767.1 ± 462.7 mg/L, with a median of 678 mg/L and an interquartile range (IQR) of 411.0–1031.8 mg/L (IQR width 620.8 mg/L), highlighting significant interindividual variability. Total IgG levels had a mean of 1338.9±352.4mg/dL, and the mean IgG4/IgG ratio was 0.0564, indicating that IgG4 accounted for approximately 5.6% of total IgG. The ratio ranged from 0.0049 to 0.1672, further supporting the heterogeneity in IgG4 expression among healthy individuals.
To define age-specific normative data, IgG4 percentiles were calculated across eight 5-year age groups. The 50th percentile (median) IgG4 values ranged between 569 and 806mg/L, while the 97th percentile values ranged from 1,358.6 to 1,920.2mg/L. Importantly, no consistent age-related trend was observed, supporting the use of a single reference range across the adult male population aged 20–60 years. Based on this, the 97th percentile value may be considered as a conservative upper limit for normal serum IgG4 in this demographic.
For comparison, retrospective data were analysed from 20 biopsy-confirmed IgG4-RD patients. All 20 patients demonstrated single-organ involvement. The most frequent sites were the sphenoid sinus, retroperitoneum, kidney, and gall bladder. Other sites included skin, larynx, neck, lacrimal gland, bone, bile duct, prostate, liver, and subcutaneous tissue. This highlights the heterogeneous distribution of IgG4-RD across organ systems, with no clear organ predominance in this cohort. The mean serum IgG4 level in this group was significantly elevated at 5,518.4 ± 7,205 mg/L, with a median of 2,271.3 mg/L and an IQR of 785.5–6,142.3 mg/L (IQR width 5,356.8 mg/L). Despite similar age distribution between groups (P = .18), IgG4 levels were significantly higher in IgG4-RD patients compared to healthy controls (P = .01).
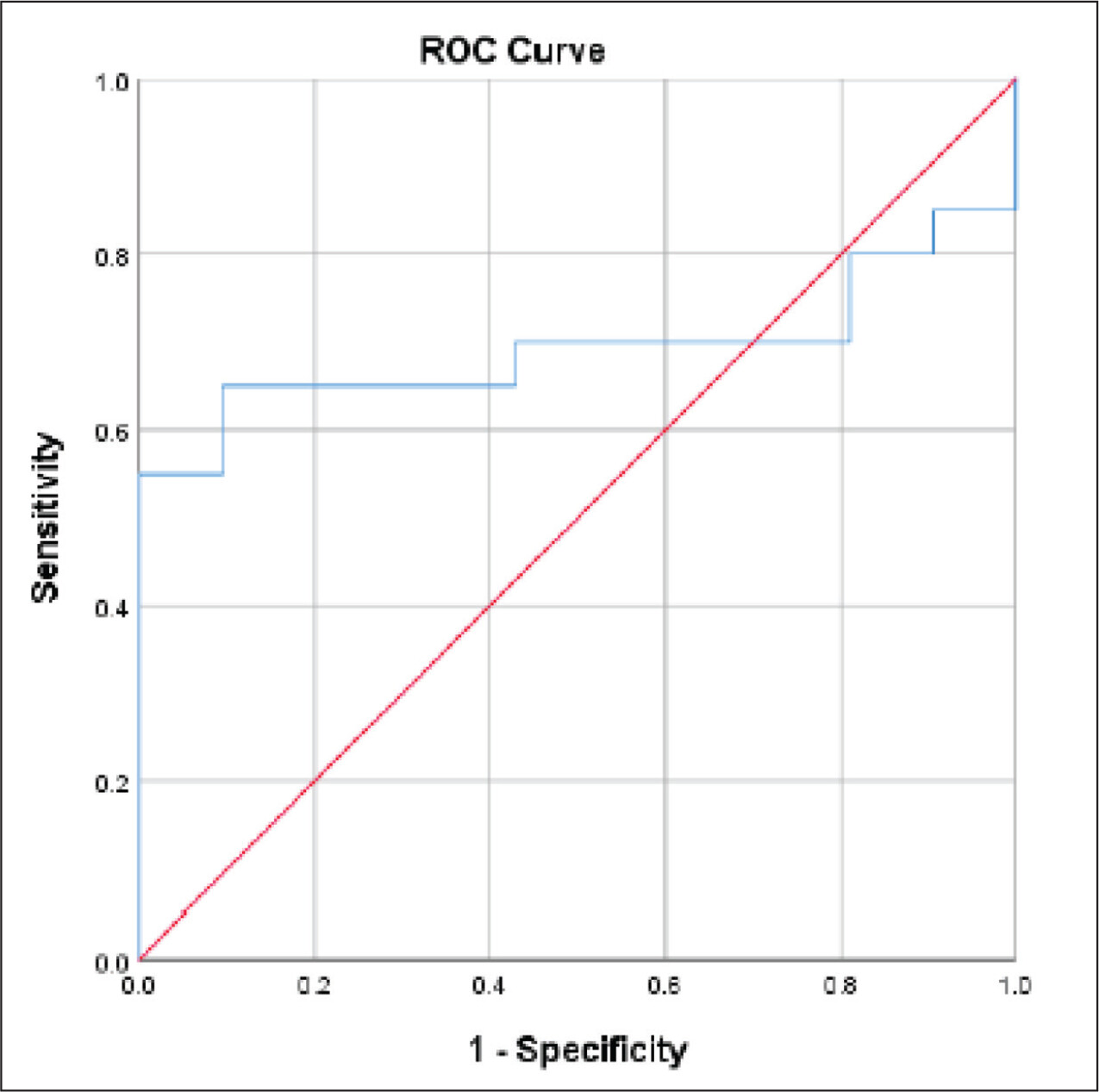
ROC curve analysis demonstrated a moderate diagnostic performance of serum IgG4 for IgG4-RD, with an AUC of 0.693 (95% confidence interval [CI]: 0.56–0.83). At a cutoff of 1,262mg/L, sensitivity was 70% and specificity of 58%. Increasing the cutoff to 1,657mg/L improved specificity substantially to 95%, but at the expense of sensitivity, which declined to 65%. This trade-off suggests that while higher cutoffs improve diagnostic confidence (rule-in), they may miss milder or early disease presentations, reinforcing the need for contextual interpretation in clinical decision-making.
Discussion
This study is the first to generate normative reference data for serum IgG4 concentrations in a healthy adult male population from North India. Among 203 participants aged 20–60 years, the mean serum IgG4 level was 767.1mg/L (SD: 462.7), with a median of 678mg/L and wide interindividual variability (IQR: 411.0–1,031.8 mg/L) (Table 1). These findings underscore the substantial physiological variation in serum IgG4 levels even among healthy individuals, a fact that aligns with previously reported global data. A study by Aucouturier et al., using competitive ELISA, demonstrated a similar variability in healthy adults, with IgG4 levels ranging from 10 to 1,870mg/L (mean: 465mg/L) in males.14 Another large-scale Chinese study using nephelometry by Xia et al. reported a median IgG4 level of 450mg/L (IQR: 220–920) in 1,248 adults, again reflecting high variability.15
| Parameter | Mean (SD) |
|---|---|
| Age (years) | 41.0 (11.3) |
| Height (cm) | 172.0 (6.8) |
| Weight (kg) | 76.5 (10.6) |
| BMI (kg/m²) | 25.8 (3.3) |
| Pulse (bpm) | 74.6 (7.3) |
| ystolic BP (mmHg) | 126.3 (10.0) |
| Diastolic BP (mmHg) | 80.1 (6.9) |
| Haemoglobin (g/dL) | 14.4 (1.2) |
| Urea (mg/dL) | 23.1 (6.2) |
| Creatinine (mg/dL) | 0.82 (0.17) |
| AST (U/L) | 33.4 (13.9) |
| ALT (U/L) | 37.6 (16.8) |
| Total protein (g/dL) | 7.78 (0.38) |
| Albumin (g/dL) | 4.68 (0.37) |
| RBS (mg/dL) | 121.7 (23.6) |
| IgG4 (mg/L) | 767.1 (462.7) |
| IgG (mg/dL) | 13,38.9 (352.4) |
A key strength of our study was the stratification of participants across eight 5-year age groups. The median IgG4 values in each group (ranging from 569 to 806mg/L) showed no consistent trend with increasing age, suggesting no significant age-related effect on IgG4 levels in this population (Table 2 and Figure 1). Although some Western studies, such as the Spanish cohort by Carballo et al., have reported a decline in serum IgG4 levels with advancing age,16 others, like Aucouturier et al., have found the opposite trend.14 Given these conflicting observations and the absence of robust, consistent evidence across populations, adopting a single reference range for adult males aged 20–60 years appears justifiable.
| Age (Years) | 3% | 10% | 25% | 50% | 75% | 90% | 95% | 97% |
|---|---|---|---|---|---|---|---|---|
| 20–25 (n = 19) | 169.9 | 222.6 | 416.0 | 743.0 | 978.5 | 1,284.2 | 1,398.4 | 1,403.4 |
| 26–30 (n = 23) | 295.6 | 315.7 | 462.0 | 640.2 | 1,247.5 | 1,703.4 | 1,863.2 | 1,920.2 |
| 31–35 (n = 29) | 187.7 | 273.0 | 362.0 | 806.0 | 1,325.0 | 1,471.1 | 1,729.1 | 1,876.4 |
| 36–40 (n = 29) | 126.9 | 200.6 | 438.2 | 569.0 | 802.5 | 984.2 | 1,114.0 | 1,358.6 |
| 41–45 (n = 30) | 125.0 | 196.2 | 437.1 | 730.4 | 1,031.1 | 1,469.8 | 1,564.2 | 1,616.5 |
| 46–50 (n = 19) | 112.8 | 200.0 | 451.5 | 666.0 | 1,160.0 | 1,276.4 | 1,354.0 | 1,393.6 |
| 51–55 (n = 26) | 256.7 | 270.5 | 411.6 | 652.7 | 887.0 | 1,129.4 | 1,467.0 | 1,600.6 |
| 56–60 (n = 28) | 239.3 | 307.9 | 438.0 | 773.7 | 1,119.8 | 1,477.7 | 1,550.9 | 1,576.9 |
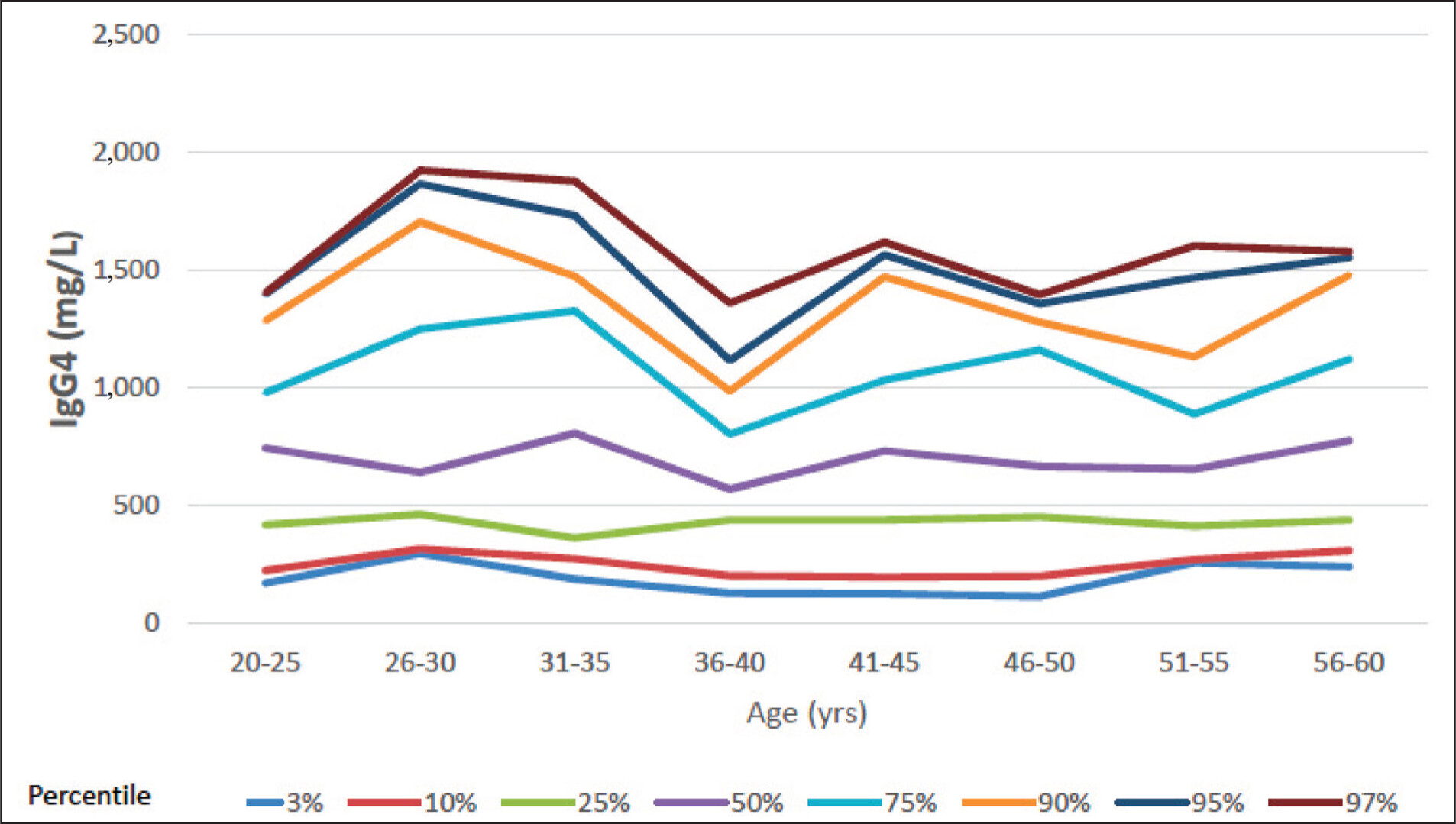
We also examined the IgG4-to-total IgG ratio, which averaged 0.0564, indicating that IgG4 constitutes approximately 5.6% of total IgG. This finding is consistent with reported physiological proportions in the literature, where IgG4 generally accounts for 0.8%–11.7% of total IgG.17 However, the wide range of ratios observed in our study (0.0049–0.1,672) highlights the need for careful interpretation when using this ratio clinically.
In the retrospective analysis of biopsy-proven IgG4-RD cases (n = 20), the mean serum IgG4 level was 5,518.4mg/L (SD: 7,205) and median 2,271.3mg/L, markedly elevated compared to healthy controls. Despite comparable age distributions (P = .18), the IgG4 levels were significantly higher in disease cases (P = .01), reinforcing the clinical utility of IgG4 measurement in suspected IgG4-RD (Table 3 and Figure 2).
| Parameter | Healthy Resampled Cohort (n = 20) | IgG4-RD Cohort (n = 20) | P Value |
|---|---|---|---|
| Age (years), mean (SD) | 39.2 (10.8) | 44.4 (13.0) | .18 |
| IgG4 (mg/L), mean (SD) | 1,217.5 (350.6) | 5,518.4 (7205) | .01 |
ROC curve analysis yielded an AUC of 0.693, indicating moderate diagnostic discrimination. A cutoff of 1,262mg/L offered 70% sensitivity and 58% specificity, while a higher threshold of 1,657mg/L improved specificity to 95%, though at the cost of reduced sensitivity (65%) (Figure 3). These findings are consistent with the broader literature. For example, a meta-analysis by Hao et al., involving 1,235 IgG4-RD patients and 5,696 controls, reported a pooled sensitivity of 87.2% and specificity of 82.6% for serum IgG4, with cutoffs ranging from 135–144mg/dL. The analysis also concluded that raising the threshold increases specificity but reduces sensitivity—mirroring our results.18 North Indian cohort of patients with IgG4-related renal disease reported IgG4 levels ranging from 85 to 1,740mg/dL (mean: 252mg/dL), but no normative data for healthy individuals were available.19
Importantly, elevated serum IgG4 is not disease-pecific. Carruthers et al. reported that up to 5% of the general population may have elevated IgG4 levels without having IgG4-RD. In their study, among 190 individuals with high IgG4, 125 did not have IgG4-RD, with IgG4 levels averaging 2,340mg/L.5
erum IgG4 concentrations have been shown to correlate positively with the number of organs affected in IgG4-RD. In a study by Han DG et al., a statistically significant, moderate positive correlation was observed between the extent of organ involvement and serum IgG4 levels, supporting the utility of serum IgG4 quantification in disease assessment.20
A notable limitation in the diagnostic utility of serum IgG4 levels is that approximately 30% of patients with IgG4-RD may present with normal IgG4 concentrations,21 thereby necessitating reliance on comprehensive clinicopathological criteria for diagnosis, including clinical features, imaging, haematological parameters, and, critically, histopathological evaluation of affected tissue, which remains the gold standard.
This highlights the limited positive predictive value of serum IgG4 alone, especially in populations with low disease prevalence, and supports the need for population-specific reference ranges. Prior Indian data on IgG4 reference values are limited. The reference intervals established in this study will aid clinicians in interpreting IgG4 levels more accurately, while the IgG4 cut-off for IgG4 RD will help to refine its clinical utility across diverse clinical contexts and organ systems involved in IgG4-RD. However, the male-only sample, small disease cohort, modest AUC, and lack of organ-specific cutoffs limit generalizability and diagnostic precision; subclinical confounders may also have influenced results.
Conclusion
This study establishes the first age-stratified normative reference intervals for serum IgG4 in North Indian males, demonstrating wide interindividual variability but no significant age-related trends, implying a uniform adult reference range. It also identifies diagnostic cut-offs to enhance the clinical utility of IgG4. A threshold of 1262mg/L offers balanced diagnostic sensitivity and specificity, with an AUC of 0.693(95% CI: 0.56–0.83), which provides only moderate diagnostic accuracy and hence limits the utility of IgG4 as a single diagnostic test. Thus, clinical interpretation must remain contextual, integrated with clinical and radiological findings. Future multicentric studies involving larger and more diverse populations are warranted to refine these thresholds and improve diagnostic accuracy.
Ethics Approval
Ethical approval was obtained from the Institutional Ethics Committee of the institute (IEC-INT-2023/MD-1257), and all procedures adhered to the Declaration of Helsinki.
Informed Consent
Written informed consent was obtained from all participants prior to inclusion in the study, ensuring voluntary participation and understanding of the research procedures.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Data availability statement
All datasets generated and analysed in the current study are available from the corresponding author on reasonable request, and ome data may already be included in the published article or upplementary materials.
References
1. Mahajan VS, Mattoo H, Deshpande V, et al. IgG4-related disease. Annu Rev Pathol Mech Dis. 2014;9:315–347.
2. Floreani A, Okazaki K, Uchida K, et al. IgG4-related disease: changing epidemiology and new thoughts on a multisystem disease. J Transl Autoimmun. 2021;4:100074.
3. Umehara H, Okazaki K, Masaki Y, et al. Comprehensive diagnostic criteria for IgG4-related disease (IgG4-RD), 2011. Mod Rheumatol. 2012;22:21–30.
4. Wallace ZS, Naden RP, Chari S, et al. The 2019 American College of Rheumatology/European League Against Rheumatism classification criteria for IgG4-related disease. Ann Rheum Dis. 2020;79:77–87.
5. Carruthers MN, Khosroshahi A, Augustin T, et al. The diagnostic utility of serum IgG4 concentrations in IgG4-related disease. Ann Rheum Dis. 2015;74(1):14–18.
6. Yamamoto M, Tabeya T, Naishiro Y, et al. Value of serum IgG4 in the diagnosis of IgG4-related disease and in differentiation from rheumatic diseases and other diseases. Mod Rheumatol. 2012;22:419–425.
7. Li P, Chen H, Deng C, et al. Establishment of a serum IgG4 cut-off value for the differential diagnosis of IgG4-related disease in Chinese population. Mod Rheumatol. 2015;25:1–5.
8. Yu KH, Chan TM, Tsai PH, et al. Diagnostic performance of serum IgG4 levels in patients with IgG4-related disease. Medicine (Baltimore). 2015;94:e1707.
9. Su YY, Sun W, Wang CQ, et al. Detection of serum IgG4 levels in patients with IgG4-related disease and other disorders. PLoS One. 2015;10:e0124233.
10. Tabata T, Kamisawa T, Takuma K, et al. Serial changes of elevated serum IgG4 levels in IgG4-related systemic disease. Intern Med. 2011;50:69–75. 10.2169/internal-medicine.50.4321
11. Khosroshahi A, Bloch DB, Deshpande V, Stone JH. Rituximab therapy leads to rapid decline of serum IgG4 levels and prompt clinical improvement in IgG4-related systemic disease. Arthritis Rheum. 2010;62:1755–1762. 10.1002/art.27435
12. Ebbo M, Grados A, Bernit E, et al. Pathologies associated with serum IgG4 elevation. Int J Rheumatol. 2012;2012:602809. 10.1155/2012/602809
13. Ngwa TN, Law R, Murray D, Chari ST. Serum immunoglobulin G4 level is a poor predictor of immunoglobulin G4-related disease. Pancreas. 2014;43:704–707. 10.1097/MPA.0000000000000118
14. Aucouturier P, Danon F, Daveau M, et al. Measurement of serum IgG4 levels by a competitive immunoenzymatic assay with monoclonal antibodies. J Immunol Methods. 1984;74(1):151–162.
15. Xia CS, Fan CH, Liu YY. Diagnostic performances of serum IgG4 concentration and IgG4/IgG ratio in IgG4-related disease. Clin Rheumatol. 2017;36(12):2769–2774.
16. Carballo I, Alvela L, Pérez LF, et al. Serum concentrations of IgG4 in the Spanish adult population: relationship with age, gender, and atopy. PLoS One. 2016;11(2):e0149330.
17. Meulenbroek A. Human IgG Subclasses: Useful Diagnostic Markers for Immunocompetence. CLB; 2002.
18. Hao M, Liu M, Fan G, Yang X, Li J. Diagnostic value of serum IgG4 for IgG4-related disease: a PRISMA-compliant systematic review and meta-analysis. Medicine (Baltimore). 2016;95(21):e3785.
19. Singh N, Nada R, Rawat A, et al. Spectrum of IgG4-related kidney disease at a tertiary care center. Indian J Nephrol. 2018;28(3):209–214.
20. Han DG, Ying CL, Cai ZP, Tong QY, Liu W. Comparative analysis of clinical characteristics in multi-organ and single-organ involvement of IgG4-related disease: a single-center retrospective study. Arch Iran Med. 2025;28(5):303–312. 10.34172/aim.34027
21. Guma M, Firestein GS. IgG4-related diseases. Best Pract Res Clin Rheumatol. 2012;26(4):425–438. 10.1016/j.berh.2012.07.001
Cite
Cite
Cite
OR
Download to reference manager
If you have citation software installed, you can download citation data to the citation manager of your choice
Information, rights and permissions
Information
Published In
Article first published online: March 26, 2026
Keywords
Data availability statement
Data is available for this article. View more information
History
Published online: March 26, 2026
Authors
Authors’ Contribution
Pinaki Dutta contributed to conceptualisation, methodology, validation, resources, visualisation, supervision and project administration. Dipika Bansal contributed to methodology and formal analysis. Shamsher Singh contributed to investigation, data curation, writing - original draft and writing - review and editing. Amal Shaharyar contributed to investigation, data curation, writing - original draft and writing - review and editing. Vivek Jha and Debajyoti Chatterjee contributed to the investigation. Ranjana W. Minz and Yashwant Kumar contributed to validation. All authors read and approved the final manuscript.
Metrics and citations
Metrics
Journals metrics
This article was published in Indian Journal of Rheumatology.
View All Journal MetricsPublication usage*
Total views and downloads: 41
*Publication usage tracking started in December 2016
Publications citing this one
Receive email alerts when this publication is cited
Web of Science: 0
Crossref:
There are no citing articles to show.
Figures and tables
Figures & Media
Tables
View Options
View options
PDF/EPUB
View PDF/EPUBAccess options
If you have access to journal content via a personal subscription, university, library, employer or society, select from the options below:
loading institutional access options
Alternatively, view purchase options below:
Access journal content via a DeepDyve subscription or find out more about this option.




