Bypassing Prescribers and Pharmacists: Online Purchasing of Semaglutide and Tirzepatide “For Research Purposes”
Abstract
Legitimate Semaglutide and Tirzepatide Products Are Scarce and Expensive
Our Assessment of Websites Online That Sell Semaglutide or Tirzepatide Products
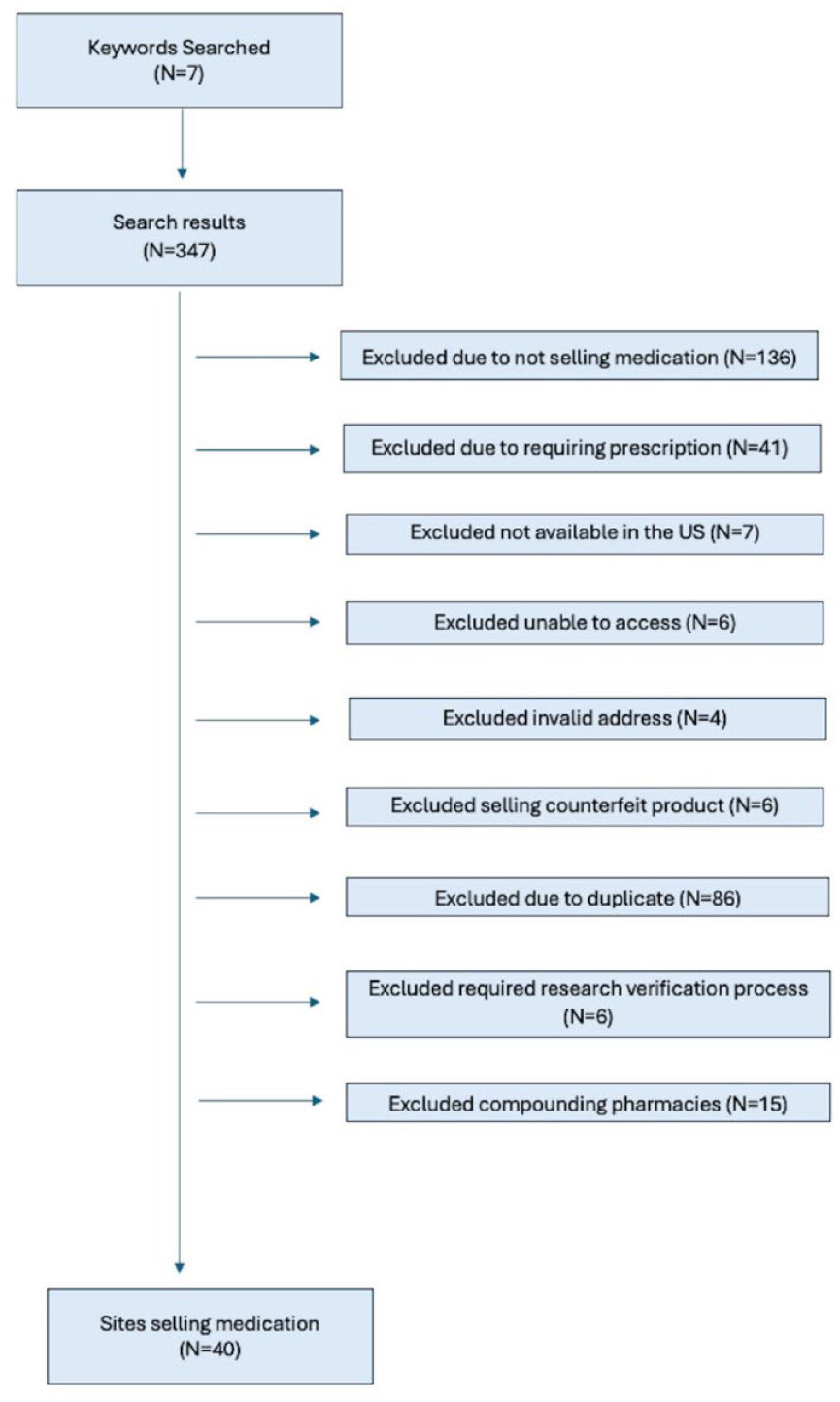
Loophole Assessment
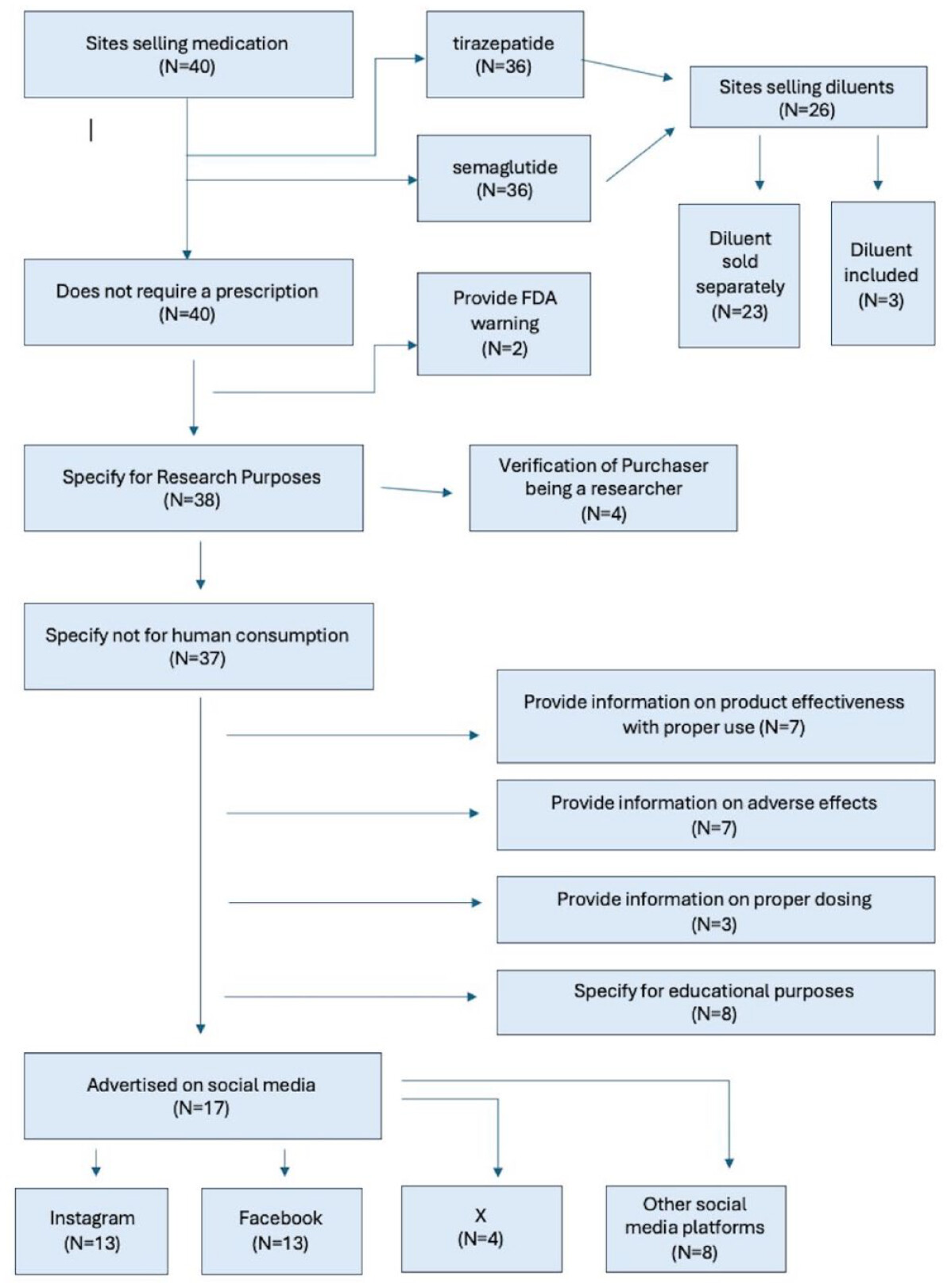

Safety Assessment
Health Implications for Consumers
Quality Issues and Product Access
Bypassing Health Professional Issues
What Can Health Professionals Do?
Close the Loophole and Support the FDA
Alerting Consumers
Declaration of Conflicting Interests
Funding
ORCID iD
References
Cite
Cite
Cite
Download to reference manager
If you have citation software installed, you can download citation data to the citation manager of your choice
Information, rights and permissions
Information
Published In
Keywords
Article versions
Authors
Metrics and citations
Metrics
Publication usage*
Total views and downloads: 10871
*Publication usage tracking started in December 2016
Altmetric
See the impact this article is making through the number of times it’s been read, and the Altmetric Score.
Learn more about the Altmetric Scores
Publications citing this one
Receive email alerts when this publication is cited
Web of Science: 5 view articles Opens in new tab
Crossref: 4
- What’s a ‘black box’ warning? A pharmacologist explains how these labels protect patients
- Clinical Outcomes of Glucagon-Like Peptide-1 Receptor Agonist (GLP-1 RA) and Glucagon-Like Peptide-1/Glucose-Dependent Insulinotropic Polypeptide Receptor Agonist (GLP-1/GIP RA) Exposures Presenting to the Emergency Department
- Characterization of glucagon-like peptide-1 (GLP-1) agonist exposures reported to a single United States poison center
- Buyer beware: Off-brand Ozempic, Zepbound and other weight loss products carry undisclosed risks for consumers
- Compounded Semaglutide and Tirzepatide Products Use Unique Formulations but Efficacy and Safety Largely Unknown
- Transdermal “Natural GLP-1” Dietary Supplements Violate Law and Place Patients at Risk
Figures and tables
Figures & Media
Tables
View Options
View options
PDF/EPUB
View PDF/EPUBAccess options
If you have access to journal content via a personal subscription, university, library, employer or society, select from the options below:
loading institutional access options
Alternatively, view purchase options below:
Purchase 24 hour online access to view and download content.
Access journal content via a DeepDyve subscription or find out more about this option.




