Introduction
Accumulating evidence has suggested that peripheral and neuroinflammation are both potential biomarkers as well as risk factors for Alzheimer's disease (AD) and related dementias (ADRD).
1,2 Systemic inflammation has been shown to lead to worsening neurological function and increase the risk of cognitive impairment
3 and may be involved early in the disease process, even before amyloid-β (Aβ) accumulation.
4,5 Aβ can also trigger the production of pro-inflammatory cytokines, exacerbating inflammatory burden.
6 In this paper we focused on peripheral inflammation as a starting point, and investigated the relationship between inflammatory markers, cognitive impairment, and clinical and demographic features in a Canadian cohort.
The term “inflammaging” has been used to describe the process linked to chronic low-grade inflammation, which is accompanied by cellular senescence, immunosenescence, organ dysfunction, and age-related diseases.
7 Multiple mechanisms (e.g., pro-inflammatory cytokines and immune cells) and risk factors (e.g., diabetes, arthritis, and alcohol intake) are associated with peripheral inflammation. The presence of disorders such as hypertension, hyperlipidemia, and obesity have been linked to vascular dysfunction via inflammation and oxidative stress.
8 Certain diets have been shown to play an important role in increasing or reducing peripheral inflammation and affecting neurodegenerative processes.
9,10,11 Quality and duration of sleep, sex, and history of smoking have also been linked to inflammatory processes.
12–16 Many of those factors are also associated with aging, which increases the difficulty in elucidating their impact across dementing disorders.
Interleukin-6 (IL-6) is an inflammatory marker central in inflammaging, which induces the expression of a variety of proteins that support inflammatory processes. IL-6 also stimulates the production of C-reactive protein (CRP), an acute phase protein secreted by the liver, commonly regarded as an inflammatory biomarker and used to assess the presence and severity of low-grade inflammation. IL-6 and CRP are easy to detect in serum and are secreted in large amounts during infections, making them arguably the most commonly assayed biomarkers of peripheral inflammation in humans.
17Peripheral inflammation has been linked to cognitive impairment in multiple diagnostic groups.
18–21 Research suggests IL-6 levels are closely linked to the size of brain regions, including the temporal and frontal lobes, hippocampal volume, and total gray matter.
22,23,24,25 Elevated IL-6 levels may contribute to neurodegenerative diseases by promoting tau and Aβ protein accumulation,
26–28 and via dysfunction in the blood–brain barrier (BBB).
29 Vascular dysfunction plays a critical role in compromising the integrity of the BBB, and IL-6 is upregulated in response to vascular injury and systemic inflammation. Disruption of vascular homeostasis—often driven by endothelial cell senescence, oxidative stress, and degradation of tight junction proteins—leads to increased BBB permeability, facilitating the infiltration of neurotoxic substances and immune cells into brain tissue.
30,31It should be noted that the concept of inflammaging is not universally accepted. A few factors should be considered such as substantial variability which has been described in different populations.
32 Additionally, there is not full agreement on what constitutes normal levels of IL-6 in the elderly, and what represents chronic low grade inflammation, as many aging related conditions can result in IL-6 upregulation.
33–35 Similarly, different upper limits of CRP are used to assess the risk of developing different conditions, such as cardiovascular disease, or to determine viral versus bacterial infection.
36,37In this study, our goal was to shed light on the relationship between inflammation, aging, neurodegeneration, clinical and lifestyle features of elderly Canadian individuals. We investigated peripheral inflammation (measured by IL-6 and CRP levels) across a cohort of subjects with various degrees of cognitive impairment. We hypothesized that higher levels of peripheral inflammation would be present in a subset of elderly individuals, but higher levels of inflammation would be linked to features of lifestyle, sex and clinical history. We also hypothesized that inflammation would be linked to brain health and cognition, therefore levels indicative of chronic inflammation would be increasingly present in individuals with AD, Mixed dementia (Mixed), mild cognitive impairment (MCI), and vascular MCI (V-MCI) when compared with cognitively unimpaired (CU) individuals. We further predicted a relationship between levels of inflammation and cognitive function, whereby higher levels of inflammation are related to lower cognitive scores independent of the diagnostic groups.
Discussion
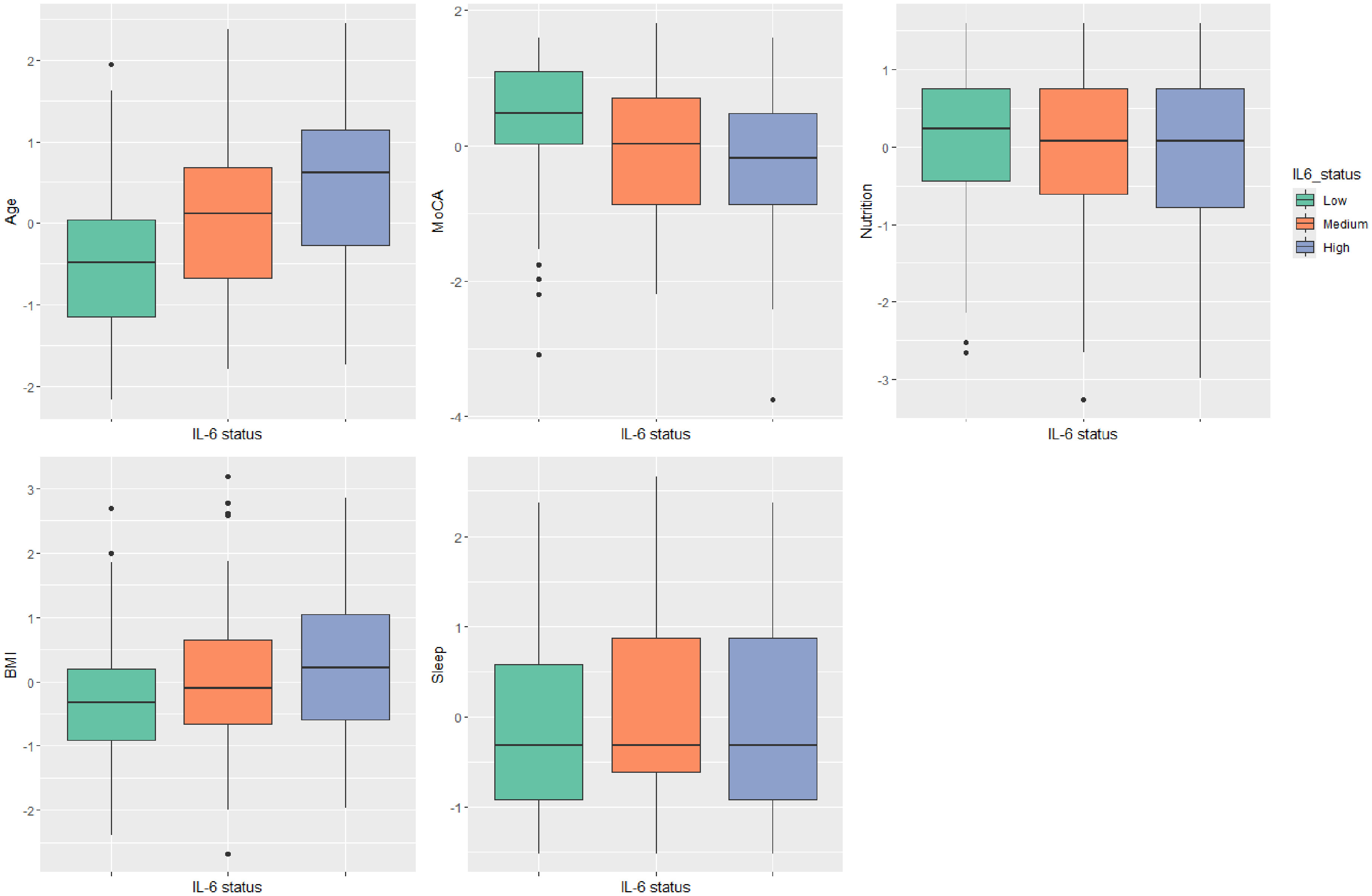
This study utilized a well-characterized cohort of an older Canadian sample with and without cognitive impairment and dementia to investigate levels of peripheral inflammation, aging, lifestyle, clinical features, and different types of cognitive impairment. Our results are in agreement with previous literature indicating a relationship between inflammation and aging. We have found that peripheral inflammation is common but by no means ubiquitous across neurodegenerative disease, and demonstrated that some variables are more strongly correlated with inflammation markers (aging and elevated BMI).
We specified groups with low, medium, and high levels of inflammation represented by the sample tertiles. Using this approach, we observed the expected cumulative relationship between levels of IL-6 and aging, BMI, nutrition, sleep, and MRI white matter changes (Fazekas scores). There was also a relationship between levels of IL-6 and cognitive function, represented by MoCA scores. Finally, there was a relationship between levels of IL-6 and diagnosis of MCI and Mixed, when compared to cognitively unimpaired older adults. We also found a relationship between CRP and age, BMI, and sex. These results have confirmed and expanded previous findings in the literature, as discussed below. Importantly, we found only a small percentage of individuals with IL-6 and CRP levels above the normal upper limit, demonstrating that few participants showed signs of clinical inflammation (i.e., approximately 8% individuals had IL-6 levels >7 pg/ml, as per the clinical assay specifications
49). Within these participants, the highest occurrence of inflammation was in the Mixed and V-MCI groups.
To our knowledge, this is the first study to investigate IL-6 and CRP levels in AD, MCI, V-MCI, Mixed dementia, and cognitively unimpaired older adults in the same cohort. The difference between diagnostic groups is a novel and important finding in our study. We found increased levels of IL-6 to be present in the cohort of subjects with mixed dementia, but not AD, when both groups were compared to cognitively unimpaired controls. This is consistent with the idea that vascular factors mediate the effect of peripheral inflammation on cognitive impairment.
Mounting evidence suggests that the pathophysiology of ADRD is marked by more than neurological changes, but also by burden in the periphery.
1851–53 Although a specific relationship between ADRD and peripheral inflammation has not been established, vascular disease is a known point of convergence between inflammation and cognitive impairment. Cerebrovascular disorders such as stroke and cerebral small vessel disease are known to disturb neurovascular function and affect the BBB, which may trigger neuro-inflammation.
54,55Vascular dysfunction is often present in AD, but the participants in the COMPASS-ND study go through rigorous clinical and imaging scrutiny in order to obtain a final diagnosis, and the absence of significant vascular disease on MRI scanning is a core criteria for the diagnosis of AD. The scarcity of increased IL-6 levels in the AD cohort when compared to the mixed dementia group is also consistent with the idea that vascular dysfunction can compromise the integrity of the BBB and mediate the effect of peripheral inflammation on cognitive impairment.
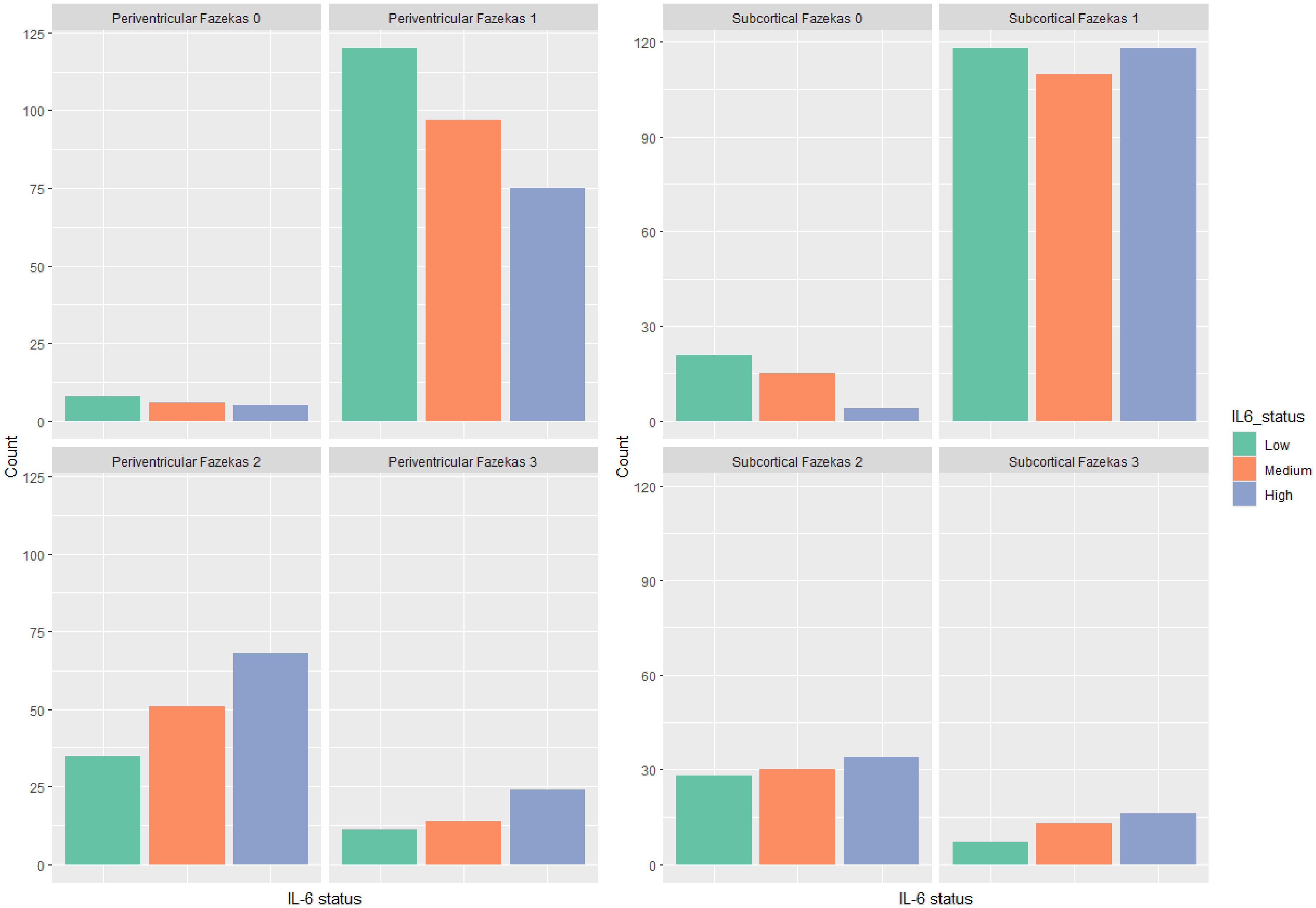
30,31It would be expected that if vascular factors mediate the effect of peripheral inflammation on cognitive impairment, then inflammation levels should bear a strong relationship with vascular findings on MRI, and this is indeed the case. We found a relationship between IL-6 and the group with Fazekas score of 1, whereby receiving a Fazekas score of 1 (effectively the lowest rating of white matter MRI changes) decreases the log cumulative odds of being in a higher IL-6 category. Because of their age, very few participants had a Fazekas score of 0, representing no white matter change. The large majority (90%) of the cohort had a Fazekas score of 1 (pencil thin lesions) or 2 (beginning confluence of lesions). A significant relationship was also found in the group with Subcortical Fazekas 1; however, all IL-6 status were overrepresented in that group, without indication of a continuous relationship between variables. These findings suggest that elevations in IL-6 may be largely contributing to cognitive impairment via vascular mechanisms. This conclusion is tentative, given that IL-6 is also elevated in MCI subjects.
Although we did not find a significant difference in IL-6 levels in the group with AD (in comparison with the cognitively unimpaired group), we did find elevated IL-6 in the group of subjects with MCI, in often cases a prodromal early phase of AD. A number of explanations can be proposed for this finding, not mutually exclusive. Perhaps peripheral inflammation is an early feature in the time-course of events whose cascade results in AD. Perhaps the evident drop in BMI in the AD group (poor nutrition is a characteristic of AD
56) results in a decrease in the degree of peripheral inflammation.
Notably, elevation in IL-6 was shown to be related to a diagnosis of neurodegenerative conditions and cognitive impairment as reflected in the MoCA scores, while CRP was not. In normal healthy conditions, the BBB regulates the passage of substances from the bloodstream into the brain, meaning that not much IL-6 crosses the barrier under normal circumstances. Under inflammatory conditions, the integrity of the BBB can be compromised. This increased permeability allows cytokines like IL-6 to cross more readily. Conditions such as infections, traumatic brain injury, or dementia can lead to elevated levels of IL-6 in the central nervous system (CNS).
31 CRP is a protein mechanistically downstream from IL-6, and may simply be a less sensitive biomarker. Although inflammatory conditions or brain injuries may compromise the integrity of the BBB and allow some proteins to cross more readily, the BBB restricts the passage of large molecules like CRP. Our findings suggest that IL-6 may exert a more robust effect from the periphery to the CNS.
Only a few individuals who were cognitively unimpaired (n = 11, or 12%) were present in the High IL-6 group. At the same time, there existed individuals in all dementia and pre-dementia categories who did not show peripheral inflammation. Elevated IL-6 is therefore not ubiquitous in dementia—perhaps peripheral inflammation defines a mechanistic subgroup within each dementia cohort.
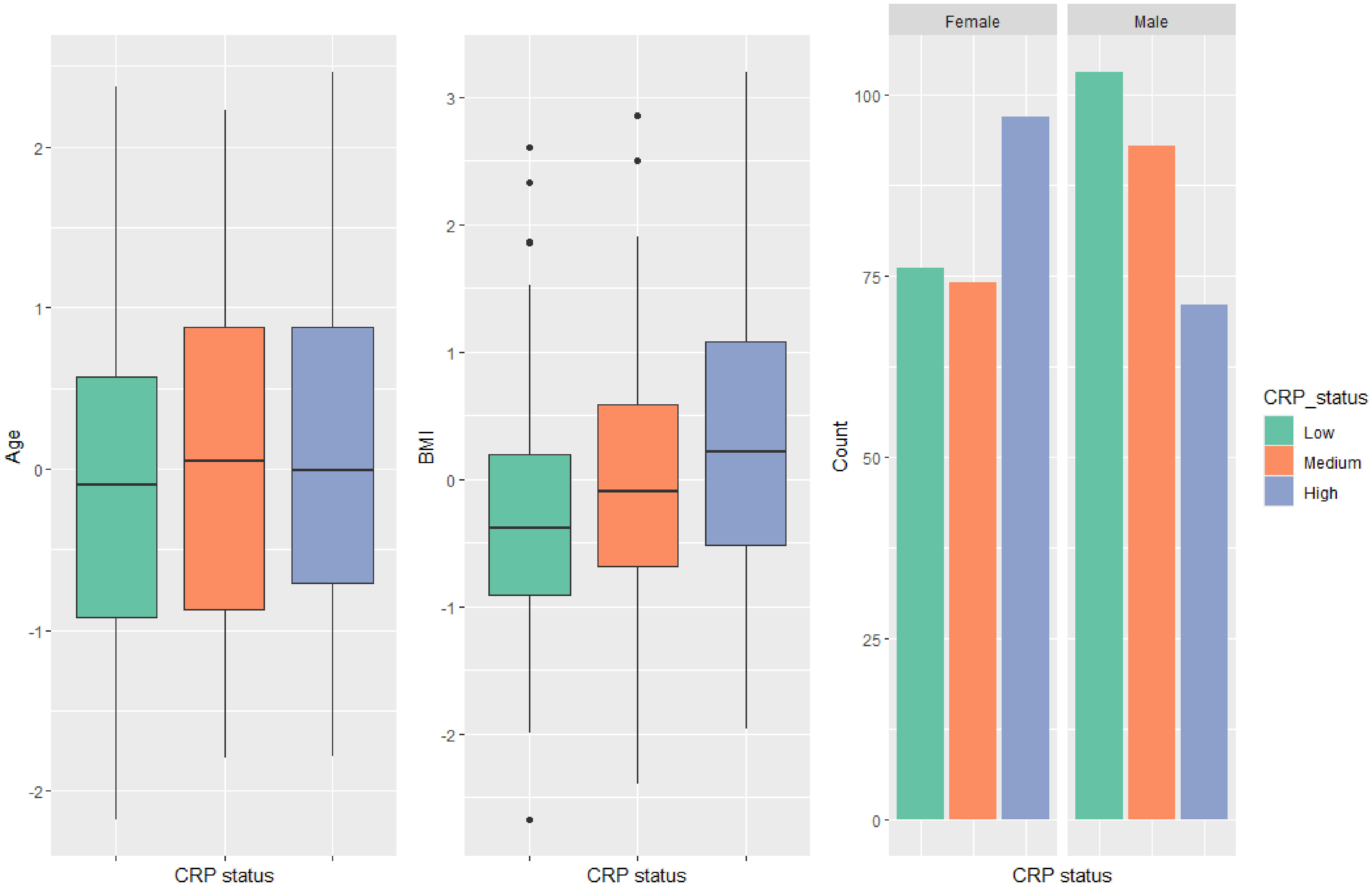
In agreement with previous studies, differences were also observed in the analyses investigating sex differences, whereby more female participants had increased levels of CRP than males. Differences in aging between male and female participants have been widely investigated in the literature.
57 In general, female participants tend to have higher total life expectancy than males, but female participants have an increased risk to live an unhealthy longevity characterized by functional impairment, which has been dubbed the female–male health–survival paradox.
58,59 It has been suggested that this paradox may be caused by hormonal and genetic sex-related differences. For instance, age at natural menopause is considered a marker of biological aging and is increasingly recognized as a marker for chronic disease risk in later life and age-related morbidity and mortality.
60,61 We did not find the same results in the IL-6 analysis. The reason for this discrepancy is unknown but may be related to inherent differences between IL-6 and CRP. IL-6 is a multifunctional cytokine, which are small proteins produced by a variety of cells and tissue throughout the body. In turn, CRP is a larger protein produced by the liver, induced by cytokines such as IL-6, IL-1, and TNF-alpha. It is possible that differences in liver function or other hormonal or genetic characteristics might differently affect CRP production in female participants. Further studies should investigate these hypotheses.
Our data suggest that obesity (elevated BMI) is a major driver of inflammation in aging, considering that significant differences were found both in IL-6 and CRP levels, with higher OR than other clinical variables investigated (OR 1.66 in the IL-6 analysis, OR 1.72 in the CRP analysis). In our sample, 200 participants (39.4%) were overweight (BMI range: 24.9–29.9) and 114 (22.1%) were obese (BMI: 30-48).
The relationship between BMI and inflammation was the most robust association in our sample, alongside aging. This suggests that obesity may be a key driver of peripheral inflammation in the elderly population. Adipose tissue is one of the main sources of IL-6 and aging-related increase in adiposity has been suggested as one of them main probable triggers of inflammaging.
18 Adipose tissue-derived IL-6 may have an effect on metabolism through several mechanisms, including adipose tissue-specific gene expression, triglyceride release, lipoprotein lipase downregulation, and insulin sensitivity.
62 Obesity is a complex condition that can be influenced by many factors like genetics, diet, physical activity levels, environmental, socioeconomic, and behavioral factors. It is also associated with an increased risk of developing aging related conditions like diabetes, atherosclerosis, cardiovascular disease, and other chronic conditions. Obesity seems to be at the crossroads of several conditions associated with aging, inflammation, and possibly contributing to neurodegeneration and dementia. Future studies should investigate the effect of obesity in cognitive impairment taking into consideration the possible direct or indirect routes through which this condition can affect brain health.
Decreased sleep and less brain protective diets were also factors, but weaker ones. Nutrition scores were found to be related to IL-6 levels whereby worse nutrition was linked to increase in IL-6. It is known that specific foods and nutrients can help modulate acute and chronic inflammation status,
63,64 and the benefits of a healthy diet for successful aging are widely known. However, the impact of dietary intervention trials to prevent dementia has proven less efficacious than expected.
65 Further studies should investigate the relationship between BMI, nutrition, aging, and cognitive impairment to assess if specific nutrition plans focusing on BMI reduction and/or anti-inflammatory diets can increase successful aging.
A relationship between cognitive impairment, sleep disturbances and increased risk of dementia has been widely explored.
66,67 Sleep disturbances are a common feature of dementia, and nearly half of all adults older than 60 years of age report sleep disturbances.
68 Sleep disturbances are linked to inflammation, and inflammation has been linked to increased Aβ and vascular burden.
68 Since both sleep disturbance and inflammation are modifiable risk factors for dementia, understanding of the mechanisms linking sleep disturbance and dementia could facilitate the identification of targets for prevention.
The mechanisms underlying neurodegeneration and dementia are complex and multifaceted. A combination of several environmental and genetic factors are related to its etiology and progression. As such, a combination of lifestyle changes including weight management, anti-inflammatory diet, and better sleep habits, would likely be effective interventions strategies for reducing dementia risk.
As a limitation, an investigation of the relationship between increased BMI and increased body fat content, levels of exercise, and activities of daily living were not part of the scope of this study. The inclusion of these factors in future studies might help describe the relationship between BMI, nutrition and inflammation. A relationship between inflammation and comorbidities was not found when the tertiles approach was used, but a correlation did emerge when IL-6 and comorbidities were examined as continuous variables. This is consistent with a large literature demonstrating that various disease states (cardiovascular disease, diabetes, hypertension, hyperlipidemia, for instance) can increase the degree of peripheral inflammation. Note that we did not investigate the relationship between specific comorbidities and peripheral inflammation but rather considered the number of comorbidities as a factor. It is possible that some of the comorbidities we identified may be more significantly related to peripheral inflammation than others. For instance, diabetes type II (DT2) is a risk factor for cognitive dysfunction and is associated with elevated levels of IL-6 and CRP, as well as elevated BMI. In this study, 53 individuals reported having been diagnosed with DT2 (10.3% of the cohort). Within the CRP status groups, the distribution of individuals with diabetes was essentially uniform, with 19 individuals in Group 0 (10.6% of the group); 16 individuals in Group 1 (9.5%); and 18 individuals in group 2 (10.7%). In the IL-6 status groups we found 12 individuals in Group 0 (6.8%); 15 individuals in Group 1 (8.9%); and 26 individuals in Group 2 (15.1%). The mean MoCA score was identical between individuals with and without a diagnosis of DT2 (M = 22), but mean BMI was higher in individuals with DT2 (M = 28) in comparison with individuals without DT2 (M = 26). Future studies should further investigate these relationships.
Further, although the MoCA is a validated tool for detecting cognitive impairment, it is not a full neuropsychological evaluation. Future studies will investigate the relationship between inflammation levels and a complete neuropsychological battery.
We did not find a relationship between inflammation and smoking, but very few of our participants reported a history of smoking (17 out of 514, or 3.3%). A comparison within a group of aging participants with a larger presence of smokers could lead to different results. Finally, the investigation would be considerably helped by longitudinal data, which is currently being collected on most of the cohort.
Clearly, peripheral low grade inflammation is common; if 36% of cognitively unimpaired older individuals display elevated IL-6, and roughly 70% of those with cognitive impairment also do so (such individuals make up roughly 20% of the aging population, according to the Alzheimer Society of Canada
69), then roughly 42% of older individuals show peripheral inflammation. Moreover, there is a clear split—about half of such individuals would show some form of cognitive impairment, while the other half do not. This in itself is illuminating—clearly the presence of peripheral inflammation, while predisposing towards development of cognitive impairment, is not incompatible with the presence of normal cognition. Ongoing longitudinal follow-up of this Canadian cohort should allow us to better understand the prognosis of low grade peripheral inflammation in those who are cognitively unimpaired.
We trust that a better understanding of the mechanisms related to the effects of peripheral inflammation, neuroinflammation, and cognitive impairment may contribute to the prevention and treatment of dementia. Future studies in this cohort will investigate the association between peripheral inflammation and neuro-inflammation in relation to other biomarkers of cognitive impairment and dementia.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Pedro Rosa-Neto has received personal compensation in the form of consulting fees from Novonordist, Eisai, and Eli Lilly; spends 20% effort on relevant clinical procedures for McGill University Research Centre for Studies in Aging—Lumbar Punctures—biomarkers studies; and receives the following research support: Operating Funding—Weston Brain Institute, Neuroinflammation Research; Operating funds—Canadian Institutes of Health Research (CIHR) (FRN, 152985), Tau propagation Research; and Infrastructure Funds—Fonds de Recherche du Québec – Santé (FRQS; (MOP-11-51-31), Aging cohort.
Dr Howard Chertkow has been supported by a Foundation Grant from the CIHR (Canadian Institutes for Health Research), along with operating funds from the Weston Foundation, Weston Brain Institute, and the National Institute of Health (USA) (Aging Research)-Phase II clinical trial of transcranial direct current stimulation in the treatment of primary progressive aphasia-1R01AG075111-01A1. Dr Chertkow directs the Bank Centre for Clinical Trials Research at Baycrest, which carries out pharmaceutical clinical trial activities sponsored by pharma companies. Any profits from these are returned to the research institute at Baycrest. Dr Chertkow, via the Bank Centre for Clinical Trials Research, has participated as a site PI in the past five years for pharmaceutical clinical trial activities sponsored by the following companies: Hoffmann-La Roche Limited, Eli Lilly Corp., Anavex Life Sciences, Alector LLC, Eisai, Bristol-Myers-Squibb (BMS), AriBio, Biogen MA Inc., IntelGenX Corp., and Immunocal. Dr Chertkow has received honoraria for sitting on advisory boards for Eisai, Biogen, and Lilly Inc. in Canada.
The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.