Introduction
Modern insulin therapy provides many different treatment modalities for type 1 diabetes (T1D) management in terms of glucose monitoring technology, means of insulin administration, and insulin analogs. Multiple daily insulin injection (MDI) remains the most common form of therapy worldwide but is often insufficient for maintaining acceptable glucose control. Insulin pump therapy (continuous subcutaneous insulin infusion [CSII]) has proven to provide modest improvements in glycemic control when compared with MDI.
1–3 Replacing conventional glucose monitoring (self-monitoring of blood glucose [SMBG]) with continuous glucose monitoring (CGM) in combination with either MDI (MDI-CGM) or CSII (with either an auxiliary CGM system or integrated with the pump, so-called sensor-augmented pump [SAP]) improves glycemic control,
4,5 but it is unclear if insulin pumps retain their superiority.
6 Despite improvements, the observed outcomes still fall short of bringing population-level HbA1c values within the internationally recognized therapeutic target of <53 mmol/mol.
4,7Automated insulin delivery (AID) systems, augmenting SAP systems with algorithm-driven insulin dosing to improve glycemic control, have emerged as the next technological step to push the treatment envelope. Several generations of AID systems have now been evaluated in randomized controlled trials (RCTs).
8 The earlier systems incorporate features to down-regulate the basal delivery on existing (low glucose suspend [LGS]) or expected hypoglycemia (predicted LGS [PLGS]), whereas the modern systems can increase basal insulin during rising glucose and decrease or suspend insulin during falling glucose, with some also delivering automatic correction boluses. Commercially available AID systems include Medtronic MiniMed™ 670G and 780G, Tandem t:slim X2 and Tandem Mobi with Control-IQ technology, Omnipod 5, Medtrum TouchCare System Nano Pump, and CamAPS (in myLife YpsoPump, Dana Diabecare RS, and DANA-I pumps). Pivotal multicenter trials of CamAPS,
9 Medtronic MiniMed 670G and 780G,
10–12 Tandem Control-IQ technology,
13 DBLG1,
14 and Insulet Omnipod 5 systems
15 consistently demonstrate superior glycemic outcomes compared with SAP or MDI with lower hemoglobin (Hb)A1c, higher time in range (TIR, 3.9–10.0 mmol/l), and reduced hypoglycemia (<3.9 mmol/l).
Beyond clinical trials, real-world data (RWD) are essential to evaluate system performance, safety, and sustainability over time. Several large-scale studies have demonstrated that glycemic benefits of AID persist in clinical practice settings. Examples include real-world evaluations of Tandem Control-IQ technology,
16,17 Medtronic MiniMed 780G,
18,19 Diabeloop DBLG1,
20 CamAPS FX,
21 and Insulet Omnipod 5.
22 Collectively, these studies report sustained improvements in HbA1c and increased TIR, without evidence of increased severe hypoglycemia or diabetic ketoacidosis, consistent with the results from RCTs.
These findings have been reinforced by registry data,
23 systematic reviews,
24–26 and meta-analyses,
27,28 also covering additional AID systems, confirming that AID systems outperform conventional therapy across multiple outcome domains, with effect sizes consistent across age groups and trial settings.
Based upon these encouraging results, several consensus guidelines now recommend AID to be considered for all populations with T1D, as it increases the likelihood of reaching recommended glycemic targets.
4,29,30 The British National Institute for Health and Care Excellence recommends use for children and adolescents in gestational diabetes and for adults with HbA1c over 58 mmol/mol.
31,32Despite robust evidence supporting AID systems, several evidence gaps remain. Most RWD studies rely primarily on CGM-derived metrics such as TIR, with only a few reporting HbA1c, which remains the gold standard for assessing long-term risk and for health-economic modeling and allows for comparison to non-CGM-based therapies. Moreover, comparative analyses between treatment modalities or AID systems are scarce, and long-term follow-up beyond 1 year is uncommon. Addressing these gaps is essential to strengthen the evidence base for clinical guideline development and reimbursement decisions.
National registries provide a unique opportunity to evaluate the impact of different treatment modalities in a nationwide population. In Sweden, structured diabetes care is supported by the Swedish National Diabetes Register (NDR), covering >95% of individuals with T1D.
33 CGM adoption became widespread in the mid-2010s, and AID uptake has accelerated since 2017. This study uses NDR data to evaluate the association between insulin delivery/glucose monitoring combinations and HbA1c in Swedish adults with T1D from 2014 to 2024, focusing on AID systems. We hypothesized that adults using AID would achieve significantly better HbA1c compared with those treated with MDI and CGM, consistent with the findings from clinical trials and real-world studies described above.
Materials and Methods
This was a retrospective, registry-based cohort study using data from the Swedish NDR between January 1, 2014, and December 31, 2024. The NDR is one of the world’s largest national quality registries for diabetes, with >95% coverage of individuals living with T1D in Sweden. It includes comprehensive data on demographics, treatments, biomarkers, comorbidities, and outcomes. All endocrinology clinics, pediatric diabetes units, and primary care centers treating people with diabetes in Sweden contribute to the registry. The NDR is maintained by the Swedish Society for Diabetology on behalf of the Swedish Association of Local Authorities and Regions.
33The study period starts about when CGM adoption became common in Sweden and extended through the most recent available year. The analysis was restricted to adults (≥18 years) with a confirmed diagnosis of T1D and at least one recorded HbA1c value per calendar year. In 2024, a total of 52,570 adults across 90 clinics met the inclusion criteria. Clinics involved ranged from small regional hospital units to large-scale university centers.
The primary outcome was glycated HbA1c (mmol/mol). For each eligible individual, all HbA1c measurements recorded within a calendar year were averaged to generate a single annual value per patient. The analysis was performed on clinic-level aggregated data, meaning that for every clinic, calendar year, and treatment combination, one aggregated data cell was provided by averaging over the annual values calculated for each patient using that treatment combination. To prevent any possibility of reidentification in small strata, standard statistical disclosure control rules were applied prior to data release from the registry: cells with <5 individuals were excluded, and those with <15 individuals were recoded as containing 10 participants.
A treatment combination was defined by the insulin delivery technology and glucose monitoring method registered in the NDR (
Table 1). The specific brand or model of the pump or CGM sensor was not always specified, and best efforts have been made to find as descriptive categories as possible. Treatment combinations with fewer than 100 users per year were excluded. Categories were mutually exclusive except for the common AID and CSII-CGM categories.
Base characteristics regarding age, gender distribution, duration since diabetes diagnosis, smoking, and physical activity (PA) habits were extracted from the registry (
Table 2). PA was measured using patient-reported information collected during clinic visits. Patients were asked to self-estimate their regular PA level. In addition, participants reported the frequency with which they engaged in these activities. Physically inactive corresponds to patients responding that they have less than one instance of 30 min PA per week (session bouts of 10 min may be added together). Reporting was generally high (>90%) for all variables, and missing data were imputed using multivariate imputation by chained equations.
34Statistical modeling
We applied linear mixed-effects regression to estimate adjusted differences in mean HbA1c across treatment combinations, with MDI-CGM as the reference. Fixed effects included the base characteristics (
Table 2), calendar year, and clinic size (small, medium, large, very large, by patient volume) and treatment combination. Clinic size was assessed in initial models but excluded from final estimates when nonsignificant. In addition, interaction between treatment effect and time was investigated (to model difference in gradual improvement between the treatment combinations) but without plausible results.
Random intercepts and random slopes for year were specified at the clinic level to account for baseline differences and heterogeneous temporal trends. Weighted rescaled regression was used to balance unequal numbers of observations per clinic.
35Effect estimates are reported as adjusted mean differences in mmol/mol with corresponding
P-values, with
P < 0.05 considered statistically significant. Analyses were performed in R v4.5.
36Model assumptions were assessed via diagnostic plots, including posterior predictive checks, residual versus fitted plots, homogeneity of variance, variance inflation factors (VIFs), and Q–Q plots for residuals and random effects.
Ethical approval
The study was performed in accordance with the Declaration of Helsinki and involved analyzing aggregated data on clinic level, which does not require any ethics approval according to Swedish law (Lag (2003:460) om etikprövning av forskning som avser människor).
Results
The dataset consists of over 600,000 patient years (
Table 1). More than a third of those correspond to the use of MDI-CGM (
n = 219,245). Due to the lack of device registration, especially in the first years of the dataset, more than 130,000 patient years are attributed to the Unknown combination. Over 100,000 patient years relate to pump use, of which about 30,000 correspond to AID systems.
The covariates were in general not correlated, except age and duration (ρ = 0.35) and age and proportion male (ρ = 0.54). The population has positive health trends, with fewer patients being inactive (
P < 0.001) and fewer daily smokers (
P < 0.03) every year, in line with the trends in the general Swedish population during this time period.
37 The patients are also getting older (
P < 0.03) and have had diabetes for a longer time (
P < 0.001). The body mass index (BMI) increased from 26.0 to 26.9 over the time period (
P < 0.001).
The MDI users were substantially older than the CSII-CGM and AID users (50 vs. 41 years, P < 0.001). Within the AID group, the Medtronic 780G users were slightly older than the Medtronic 670G and the Tandem Control-IQ users (43.3 vs. 41.8 and 40.1 years, P < 0.001).
There were small but significant differences in duration of diabetes between almost every treatment combination, with the longest duration for Medtronic 780 (26.8 years) and the shortest for Unknown (23.6 years) users.
In terms of BMI, the Medtronic 780G and 670G users had significantly higher BMI (27.5 kg/m2) than all other treatment combinations, and the AID systems, including Tandem Control-IQ, had significantly higher BMI (27.2 kg/m2) than the CSII-CGM and MDI groups (both about 26.5 kg/m2, P < 0.001).
The level of physical inactivity was highest for the MDI and Unknown groups (15%–19%) and significantly lower for the CSII-CGM group (14%) and yet lower for the AID combinations (11%). Within the AID group, there was a significant difference between the Tandem Control-IQ and Medtronic 670G users on one hand and the Medtronic 780G users (11, 10 vs. 13%, P < 0.001).
The gender distribution also differs between the treatment groups, with a male-dominated population for the MDI (58% male), whereas the CSII-CGM users are leaning more female (47% male), with Tandem t:slim X2, Unknown, and OmniPod having relatively most females (45% male). Wedged in-between are the AID systems with close to equal proportions (49% male).
Finally, daily smoking was twice as common in the MDI group (8%) compared with the AID users (4%, P < 0.001). The CSII-CGM group lands in-between (6%). Fewest smokers were found in the Medtronic 670G group (3%), which had a significantly smaller proportion than the other AID systems (P < 0.001).
Trends in glycemic control
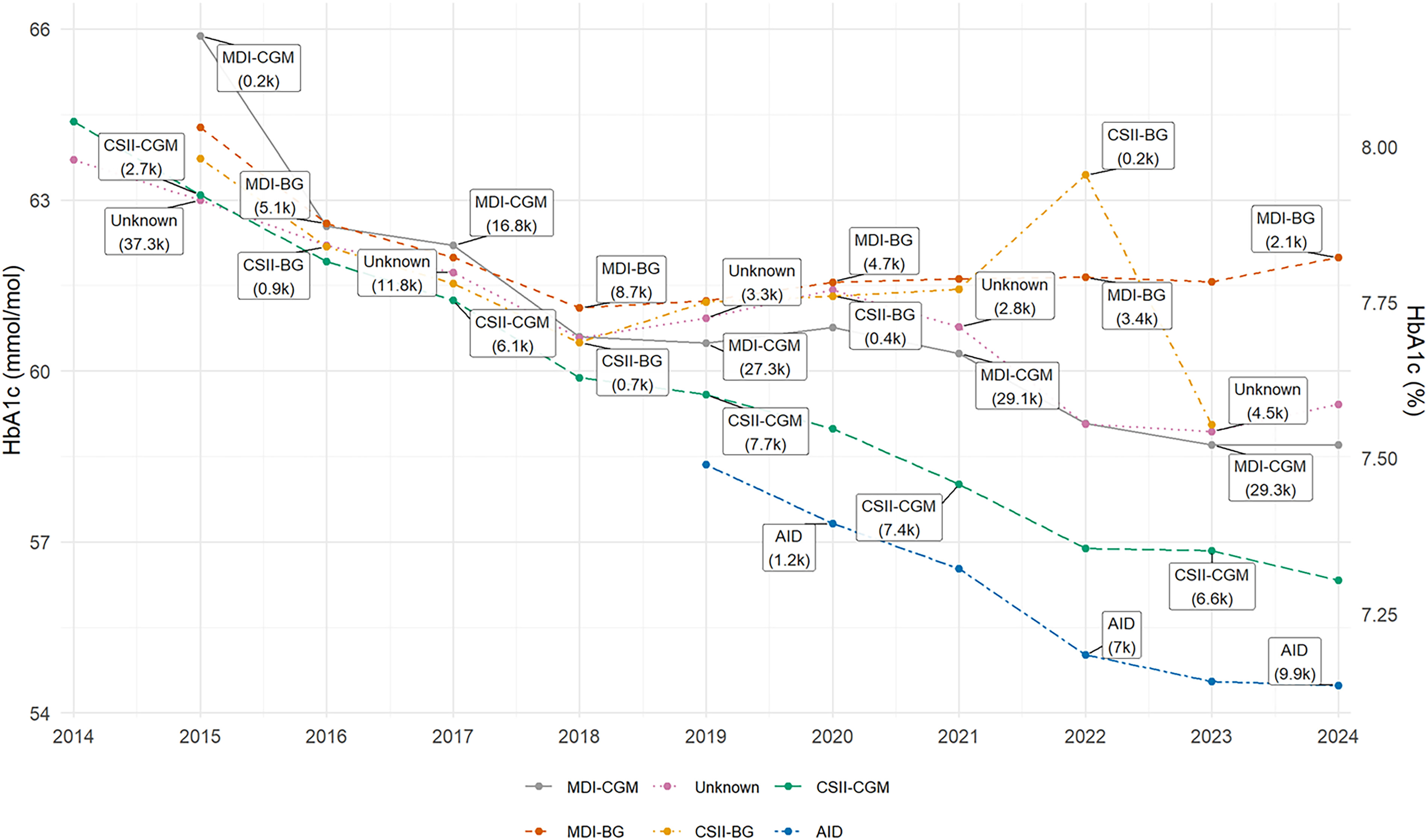
Between 2014 and 2024, mean HbA1c among adults with T1D in Sweden decreased steadily from approximately 65 mmol/mol to 57 mmol/mol. The decline was gradual but consistent, corresponding to a mean national improvement of ∼0.5 mmol/mol per year (
P < 0.001). When stratifying by treatment modality, the overall decline was not uniform (
Fig. 1). Patients treated with MDI-BG showed the slowest improvement, with mean HbA1c stabilizing around 63 to 64 mmol/mol after 2020. By contrast, patients using CGM in combination with either MDI or insulin pumps demonstrated sustained reductions. The most pronounced improvements were seen among users of AID systems, who reached mean HbA1c levels below 55 mmol/mol by 2024.
Uptake of AID systems
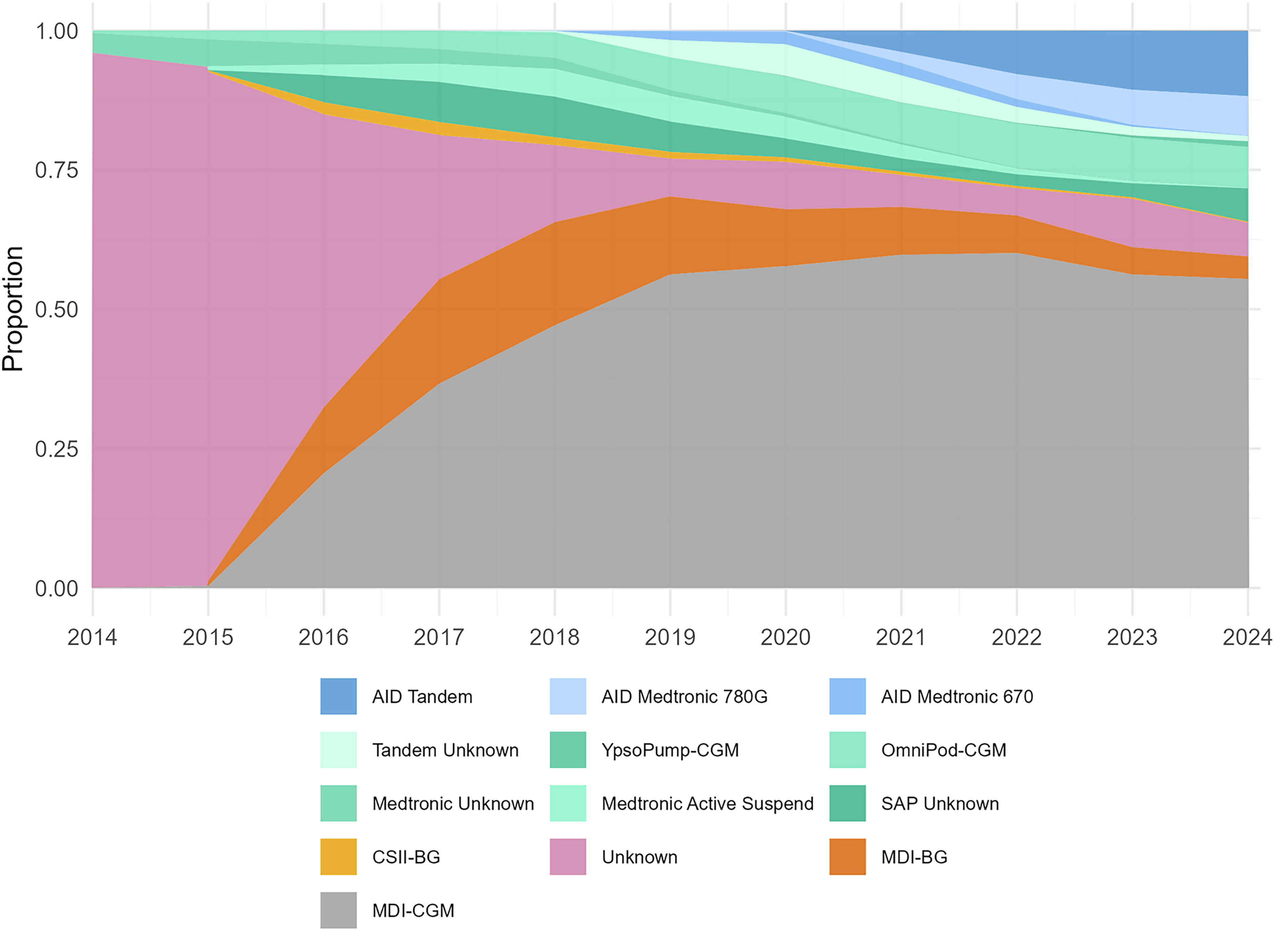
Marked shifts in treatment patterns were observed during the study period (
Fig. 2). The first years had a low rate of reporting of treatment, but, in 2017, about 80% of adults used MDI, and all pump users relied on non-AID therapy. Between 2015 and 2018, the use of CGM expanded rapidly, surpassing BG monitoring as the predominant glucose monitoring method by 2018.
Reporting quality improved markedly after 2019, coinciding with automation of registry data transfer from local electronic health records, resulting in missing reported treatment stabilizing at 10% since then. In this period (2019–2024), the proportion of adult patients using insulin pumps has increased from 25% to 36%. SAP and CSII-CGM systems have gradually been replaced by AID systems, with the exception of OmniPod Dash, whose AID counterpart OmniPod 5, was introduced in the beginning of 2025. By 2024, the majority of pump-treated adults were using a device with AID capabilities (55%), with Tandem Control-IQ technology emerging as the most widely used system nationwide and the Medtronic 670G replaced by the next-generation Medtronic 780G. The MyLife YpsoPump and the Dana IIS, both compatible with the CamAPS AID algorithm, together with Medtrum TouchCare Nano, are also present with modest market shares.
Mixed-model results
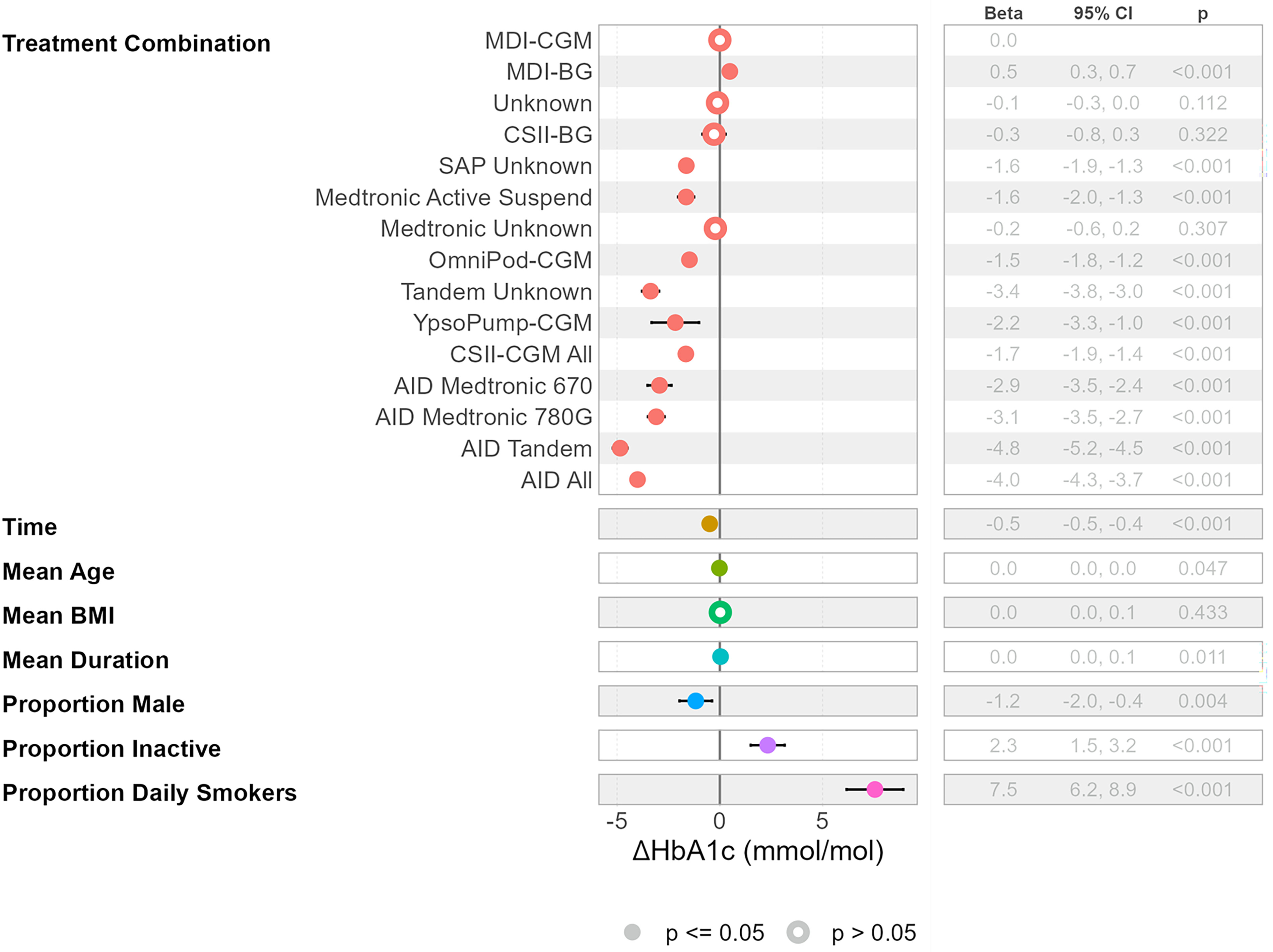
Using mixed-model regression, adjusted for temporal trend, effects of covariates, and differences in offset and trends between clinics and the treatment effects for the various treatment modalities in comparison with the reference method MDI-CGM were derived (
Fig. 3). The effects of age and duration of diabetes were both significant but very small (−0.02 and 0.04 mmol/mol per year). BMI did not have any effect.
In line with the trend seen in the data, a reduction of 0.5 mmol/mol per year was found. The effect of sex (male) was −1.2 mmol/mol. Inactivity was estimated to have an increasing effect of 2.3 mmol/mol. Smoking was the factor with the overall largest impact, with an effect size of 7.5 mmol/mol.
Treatment Combinations
The treatment effect of the AID systems as a group was consistently associated with a larger reduction of HbA1c compared with the reference MDI-CGM (−4.0,
P < 0.001). The largest effect within this group was attained by Tandem Control-IQ technology (−4.8 mmol/mol,
P < 0.001), followed by Medtronic 780G (−3.1 mmol/mol,
P < 0.001) and Medtronic 670G (−2.9 mmol/mol,
P < 0.001). The differences between these systems in turn were also significant (
Table 3).
The treatment effect in the CSII-CGM group was larger than the MDI-CGM reference but lower than the AID group. CSII had similar effect as the reference. MDI-BG was the only option with inferior effect compared with MDI-CGM.
Model performance and diagnostics
Model diagnostics (see supplementary material for plots) indicated adequate fit and no violation of assumptions. Posterior predictive checks confirmed close alignment of predicted and observed HbA1c distributions. Residual plots demonstrated linearity and homoscedasticity. VIFs were elevated for treatment category and age. Estimating a simplified model without age reduces the variance influence factor for the treatment category below 5, indicating no collinearity concerns. Q–Q plots of residuals and random effects supported approximate normality. No influential outliers were identified.
System Comparisons
Direct comparisons between Medtronic 670G and 780G in the FLAIR crossover trial demonstrated a mean HbA1c reduction of –5 mmol/mol for the 780G users versus –3 mmol/mol with 670G.
47 Real-world evidence from a pediatric cohort corroborates these findings with 6.5 mmol/L lower HbA1c 6 months after AID onboarding for the users of the newer system than corresponding follow-up data for the 670G system.
50 However, patients already using 670G and transferring at onset to 780G saw no improvement. No significant difference in terms of HbA1c could be found in our results.
Our study reports superior HbA1c outcomes for Control-IQ technology users compared with Medtronic 780G users. In contrast, some real-world studies have also reported higher TIR outcomes with Medtronic 780G compared with Control-IQ technology.
51–54 We selected HbA1c as the outcome metric due to the ability to compare outcomes across individuals using different proprietary technologies. Concerns about using TIR to compare the efficacy of AID systems utilizing different sensor systems have been raised,
55 with head-to-head comparisons between sensor systems indicating systematic differences,
56,57 and international consensus guidelines caution against comparison of CGM metrics across sensor types.
58 Results with HbA1c as outcome are mixed, with two studies comparing Control-IQ technology and 780G reporting similar HbA1c improvements from baseline (−6 to −8 mmol/mol),
53,59 whereas other studies confirm similar trends as this study with lower HbA1c with Control-IQ technology.
49,54,60 Given that the present analysis is not a head-to-head comparison and prior evidence remains inconsistent, the observed superiority of Control-IQ in this study should be interpreted with appropriate caution.
A possible explanation for the overall superior results for the AID systems compared with other modalities could be a selection bias where patients with better glycemic control are more likely to transition to AID therapy. In 2018, when AID was introduced into clinical practice, this group had a lower HbA1c than the alternatives (
Fig. 3). However, it should be recalled that the values represent averages over the year and that several studies have shown that the glycemic improvement after switching to AID is substantial already after 3 months with both Tandem and Medtronic AID systems.
61,62 Further studies are needed to investigate the effect of treatment switching and if the improvement effects are homogeneous over the population.
Potential confounders
The registry-based design limits the possibility to fully address potential confounders. The use of other glucose-lowering medications besides insulin could influence the result if prescription and use differ among the treatment groups. A double-blinded study on 72 adults with T1D and BMI of 30 or higher on AID in combination with off-label use of glucagon-like peptide-1 receptor agonist achieved an HbA1c reduction of 3 mmol/mol.
63 Several studies have been undertaken to investigate the combination of SGLT2 inhibitors and AID systems, with some promising outcomes in terms of TIR but without results for HbA1c.
64 The NDR registry has registration of these drugs, but the reporting has been insufficient, making the data unreliable. For that reason, these variables were not included in the analysis, and future studies need to control for these confounders as data quality improves. Other unknown factors are comorbidities that may impact the glycemic control, directly or indirectly. No data on comorbidities were used (data on thyroid disease were available in the registry and assessed but found to be of insufficient reporting quality).
The treatment groups had large differences in age, but the estimated effect difference was <0.2 mmol/mol when comparing the oldest and youngest cohorts, and this factor was ignored in favor of a simpler model. The effects of differences in duration of diabetes were also negligible.
Large differences in gender composition were found between MDI and the pump-based treatment options, and a relatively large gender difference effect in favor of men was estimated in the model. Results from previous studies on differences in glycemic control between the sexes have also reported worse HbA1c outcomes for women.
65,66 Differences in erythrocyte properties between the sexes may produce a bias with lower HbA1c values for men,
67 providing one potential physiological explanation among others.
68Another variable with large differences between the treatment modalities was the proportion of physical inactivity. The effect of PA on HbA1c has been studied in T1D with varying conclusions. Most studies report a small but significant improvement associated with PA. A large RWD study covering more than 18,000 patients reported a 3 mmol/mol higher HbA1c for the inactive,
69 and a meta-analysis, covering both observational and interventional trials, also found the effect to be of this magnitude.
70 These results are close to the 2.5 mmol/mol effect size found in our study.
Smoking has been linked to increased HbA1c levels in the diabetes populations, with magnitudes in the same range as found in this study.
71 It is likely that part of the effect can be explained by highly correlated socioeconomic factors, which in turn are linked to lower health outcomes. Smoking and nicotine use are in Sweden strongly associated with socioeconomic factors such as education, income level, employment status, and country of origin.
72 Further analysis is needed to investigate this effect and if it can be divided into further explanatory variables.
We found no clear relationship between BMI and HbA1c in our dataset. The association between these variables in T1D has been investigated across diverse populations with inconsistent results. Several RWD studies have reported a weak inverse relationship between BMI and HbA1c after adjustment for key covariates, with effect sizes around
β ≈ –0.1 (
P < 0.01).
73–75 These findings suggest that higher BMI may be associated with slightly lower HbA1c, potentially reflecting greater insulin doses per kilogram of body weight. In contrast, a 5-year longitudinal registry study showed that adults who gained ≥20 lb had higher mean HbA1c at follow-up compared with those with stable weight.
76 Moreover, a recent study of adults with T1D treated with insulin pumps reported a U-shaped association between BMI and HbA1c, with both underweight and obese individuals exhibiting suboptimal glycemic control.
77 Collectively, these findings underscore the heterogeneity and generally weak nature of the BMI–HbA1c relationship, consistent with our own observations.
National trends and context
Temporal trends in our data revealed a steady improvement in HbA1c across all modalities except the BG-based therapies (−0.5 mmol/mol per year from 2014 to 2024). While CGM adoption and improved sensor technology with increased accuracy and consistency may explain some of this improvement,
78 the publication of revised national and international guidelines during the time period may also have an impact.
Strengths and limitations
Strengths of our study include near-complete national coverage (>95% of adults with T1D), standardized HbA1c measurement, real-life data preventing conventional study-induced biases (e.g., Hawthorne effect and selection bias), long follow-up period (10 years), and robust analytic methods accounting for and controlling for many important covariates. In addition, the reimbursement system applied in Sweden potentially reduces the socioeconomic bias in the data by allowing the participants in the study to use the available treatment options without personal financial impact, which otherwise likely would distort the distribution of treatments over the population.
Lack of device registration implied that a large number of data points were classified into the Unknown treatment group. This issue was most pronounced in the early years and stabilized at a low level by 2019. Since HbA1c outcomes for the Unknown group generally fell between those of the MDI-CGM and MDI/CSII-BG groups, and considering the multitude of data from the SAP and AID systems prompting registration for these treatment modalities, it is likely that the Unknown group mainly consists of MDI users. Whether they used CGM or not is more unclear, which may partly obscure the comparison between the MDI-CGM and MDI-BG groups. However, a comparison using the limited dataset 2021–2024, where underreporting was low, upheld the significant difference in HbA1c outcome between CGM and BG-based MDI therapy.
While the retrospective design omits a balanced and randomized trial setup between the evaluated treatment modalities, the control for major potential confounders allows for deriving comparable estimates of treatment effects. Limitations in data access concern the lack of covariates, such as socioeconomic status, ethnicity, digital proficiency, comedication, and device use metrics and settings (e.g., percentage of time in closed-loop mode, alarm, basal, and bolus settings), and residual confounding from unmeasured factors cannot be excluded.
Recent registry data illustrate how device features can influence glycemic outcomes. In a Danish registry study examining CGM-based treatment modalities with differing alarm functionalities, significant HbA1c advantages were observed for both SAP (–7 mmol/mol; 95% confidence interval [CI] –9 to –4) and AID (–10 mmol/mol; 95% CI –11 to –8) systems incorporating alarm features compared with MDI–CGM treatments lacking such capabilities.
79 These findings underscore that specific device settings and behavioral interactions may substantially mediate treatment effectiveness and highlight the importance of designing studies and registries that capture relevant data to allow for such revelations.
A further limitation relates to the aggregation of data at the clinic level rather than the individual level. This approach precludes evaluation of treatment transition effects between treatment modalities, such as changes in glycemic outcomes immediately before and after switching from SAP to AID. Moreover, clinic-level aggregation inherently masks individual characteristics, for example, age, sex, diabetes duration, and PA. As a result, the influence of the covariates is attenuated and likely contributes to that the associations found in this study were weaker than what be observed in a person-level dataset.
Related to this, a further concern is modality switching in relation to HbA1c assessment. Since HbA1c reflects glycemia over the preceding 8–12 weeks, patients who initiated a new treatment late in the calendar year may have HbA1c values partially influenced by their prior treatment modality. As individual-level switching data were not available, these dynamics could not be estimated. For treatment modalities with balanced switching in both directions, the effect can be expected to even out. However, switching related to the novel therapies is not symmetric, with a clear net influx from MDI and older pump modalities toward SAP and especially AID. Reverse transitions (e.g., from SAP to MDI, or from AID back to SAP or MDI) are expected to be very infrequent. Consequently, first-year HbA1c values for SAP and particularly AID may, to some extent, reflect pre-switch glycemia and therefore slightly underestimate the true treatment effect of these modalities. The overall impact of this bias is expected to be limited, as the attenuation of treatment effects is diluted by the large proportion of patients whose HbA1c values reflect stable, long-term use of their respective modalities.
Clinical and policy implications
Our results indicate that, in routine practice, AID systems provide clinically meaningful HbA1c reductions compared with MDI-CGM and CSII-CGM. The magnitude of reduction (3–5 mmol/mol) is sufficient to impact the long-term risk of microvascular and cardiovascular complications.
80,81 These findings reinforce contemporary guideline recommendations advocating AID for eligible adults and support efforts to ensure equitable access.
Conclusions
This nationwide, registry-based analysis provides real-world evidence on glycemic outcomes associated with different insulin delivery and glucose monitoring technologies in Swedish adults with T1D over a 10-year period.
There has been a substantial improvement in average HbA1c at the national level, with an approximately 8 mmol/mol reduction between 2014 and 2024 paired with a dramatic increase in CGM usage from virtually zero to over 90% of the population.
Use of AID technology was consistently associated with lower HbA1c compared with both MDI-CGM and CSII-CGM, with the most treatment impact from Control-IQ technology. The AID group results corroborate previous results from RCTs and RWD studies that AID systems are associated with significantly better HbA1c outcomes than non-AID modalities, supporting the broader adoption of AID in routine care, and underscore the value of registry-based surveillance in monitoring long-term outcomes.
Further analyses are necessary to assess potential differences among subpopulations, evaluate the dynamics of glycemic improvement from onset onward to determine its stability, identify potential confounders and transition effects associated with switching treatment modalities, and delineate causal factors related to settings, algorithms, and training. Such investigations are essential to optimize interventions and enhance glycemic control within the diabetes community.
Author Disclosure Statement
F.S. is Senior Manager for Analytics and Modeling at SmartStep Nordics AB, a market access and real-world data consulting firm operating in the Nordics. J.H. has received advisory board fees from Abbott, Sanofi, Novo Nordisk, Eli Lilly, Nordic Infucare, Medtronic, and Boehringer Ingelheim and has received lecture fees from Sanofi, Novo Nordisk, Eli Lilly, Amgen, Astra Zeneca, Boehringer Ingelheim, Nordic Infucare, and Rubin Medical. C.E. is Medical Liaison Manager at Rubin Medical AB, the distributor of Tandem products in Sweden, Norway, Denmark, and Finland.