How to do a systematic review
Abstract
Introduction
Why do a systematic review in stroke?

What is a systematic review?
| Type of research question | Type of systematic review | Published examples from the field of stroke | Framework for systematic review questions | Resources for protocol development | Tools for quality assessment of included studies | Reporting guidelines |
|---|---|---|---|---|---|---|
| What is the effectiveness of an intervention (e.g. a treatment, service or policy)? | Intervention review | What is the safety and effectiveness of thrombolytic therapy for the treatment of acute ischaemia stroke?15 What is the effect of stroke unit care, as compared to alternative forms of care for people following a stroke?16 Does fitness training after stroke reduce death, dependence and disability?17 | PICO: Population, intervention, comparison, outcome PICOS: Population, intervention, comparison, outcome, study type PICOT: Population, intervention, comparison, outcome, timeframe18 PICOC: Population, intervention, comparison, outcome, context | Cochrane Handbook for intervention reviews19 Joanna Briggs Institute (JBI) Reviewers’ manual20 Standards for Systematic Reviews of Comparative Effectiveness Research21 Methodological Expectations of Cochrane Intervention reviews (MECIR)22 | Cochrane risk of bias tool23 JBI Critical appraisal tools24 | Preferred reporting items for systematic reviews and meta-analyses (PRISMA)25 Methodological expectations of cochrane intervention reviews (MECIR)22 |
| What is the accuracy (sensitivity or specificity) of a diagnostic test? | Diagnostic test accuracy (DTA) review | What is the accuracy of MRI for the detection of acute hemorrhagic lesions within 12 hours of stroke symptoms?26 What is the accuracy of cognitive diagnosis of multidomain, cognitive impairment/dementia in stroke survivors?27 | PICOT: Population, index test, comparator, outcome, target condition PPIRT: Population, prior tests, index tests, reference standard, target condition | Cochrane Handbook for DTA reviews28 | Quality assessment of diagnostic accuracy studies (QUADAS-2) tool29 Critical Appraisal Skills Program (CASP) diagnostic checklist30 Centre for evidence based medicine (CEBM) diagnostic study appraisal worksheet31 JBI critical appraisal tools24 | PRISMA-DTA: Checklist for reporting of diagnostic test accuracy systematic reviews (in development)32 |
| What is the prognosis / prevalence / predictors of recovery of a condition? | Observational studies review | What is the worldwide incidence of stroke?33 What is the prevalence of pre-stroke dementia and the prevalence and incidence of post-stroke dementia and their associated risk factors?34 What are the predictors of upper limb recovery following stroke?35 | PEO: Population, exposure, outcomes PCO: Population, context, outcome PICo: Population, interest, context | Cochrane Methods: Prognosis (resources and publications36: | CASP cohort study checklist30 CEBM Prognosis appraisal worksheet31 JBI Critical appraisal tools24 | Meta-analysis of observational studies in epidemiology (MOOSE)37 |
| What is the accuracy of an outcome assessment or measurement tool? | Review of measurement instruments | What is the validity and reliability of the Modified Rankin Scale?38 What are the psychometric properties of outcome measures used in stroke self-management interventions?39 | De Vet 2011. Chapter 9: Systematic reviews of measurement properties40 COnsensus-based Standards for the selection of health Measurement Instruments (COSMIN)41 | COSMIN checklist41: Also potentially relevant: OMERACT filter42 | COSMIN41 | |
| What are the views or experiences of people with a condition? | Qualitative review | What are stroke survivors’ experiences of rehabilitation?43 What are carers’ experiences of caring for stroke survivors?44 | SPIDER: Sample, phenomena of interest, design, evaluation, research type45 SPICE: Setting, perspective, intervention, comparison, evaluation46 ECLIPS: Expectations, client group, location, impact, professionals involved, service47 | Cochrane Handbook Chapter 20 (Qualitative research and Cochrane reviews)48 JBI Reviewers’ manual20 | CASP qualitative checklist30 JBI Critical appraisal tools24 | Enhancing transparency in reporting the synthesis of qualitative research: ENTREQ49 Realist and meta-narrative evidence syntheses – evolving standards (RAMESES) publication standards50 |
Systematic reviews in stroke
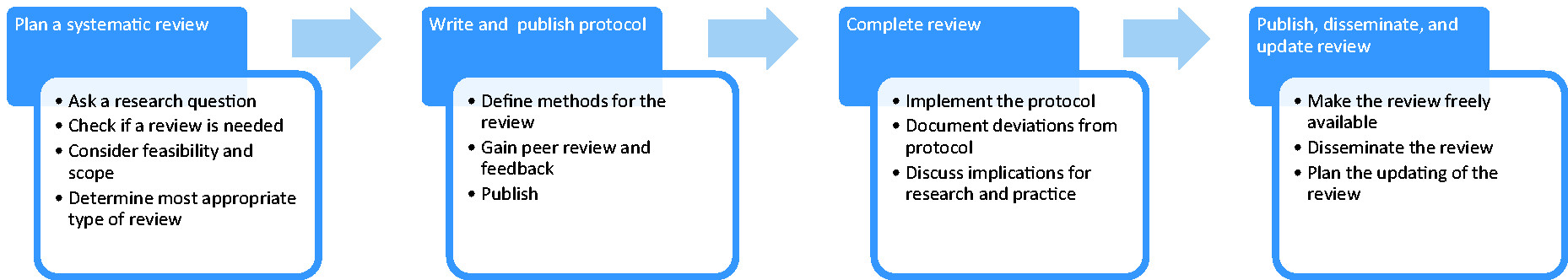
Planning a systematic review
What is the research question?
| Background and question: My patient has recently had a stroke, and can only walk with assistance. Many physiotherapists have a preference for a specific approach to rehabilitation.71,72 These approaches include the Bobath approach73,74 and the motor learning approach.75 What specific physiotherapy approach should I use in order to best improve the walking of my patient? |
| Forming the PICO question: • Patient: Patients with acute stroke (less than six weeks) with reduced mobility. • Intervention: Any specific approach to physiotherapy. • Control: No physiotherapy. • Outcome: Independence in activities of daily living; ability to walk independently. |
| PICO question: In patients with a recent acute stroke (less than 6 weeks) with reduced mobility, is any specific physiotherapy approach method more beneficial than no physiotherapy at improving independence in activities of daily living and gait speed? |
Is a systematic review needed?
Feasibility and scope of the systematic review
| PICO question: In patients with a recent acute stroke (less than six weeks) with reduced mobility, is any specific physiotherapy approach more beneficial than no physiotherapy at improving independence in activities of daily living and gait speed? |
| Arguments in favor of a broad review: • Limiting the review to patients who had a stroke during the last six weeks will arguably result in a fairly “narrow” review, and potentially large volumes of evidence arising from other patients would be excluded. A broader review would result in a review of a greater volume of evidence. • Assessing the effects of different physiotherapy approaches (not only the Bobath approach) will be clinically relevant to clinicians, who have to consider all available approaches when reaching a treatment decision. Limiting the review to only one specific approach (e.g. the Bobath approach) does not answer the clinical question relating to the relative effects of different approaches. • Considering control groups other than just a “no physiotherapy” control group will reflect the choice faced by many clinicians, who have to choose between two or more different approaches, rather than between one approach or no physiotherapy. • A broader review will have more data from additional studies, making it possible to perform meaningful subgroup analyses. Example of a broader review question: In patients with stroke with reduced mobility, is any specific approach to physiotherapy more beneficial than no physiotherapy or any other physiotherapy approach at improving independence in activities of daily living and gait speed? |
| Arguments in favor of a narrow review: • The broad review would be more work (more articles to screen, more data to extract, more analyses to be done, more results to discuss). • There would be a need to consider the generalizability of results arising from this broad population to the sub-population of primary interest for this review (patients with stroke during the last six weeks). • A review focused on just one physiotherapy approach (e.g. the Bobath approach) will be more concise and of greater interest for readers interested in this specific approach. Example of a more narrow review question: In patients with a recent acute stroke (less than six weeks) with reduced mobility, is the Bobath approach more beneficial than the motor learning approach at improving independence in activities of daily living and gait speed? |
What sort of systematic review best suits the research question?
Write and publish a protocol

Clarify review aims and objectives
Find relevant research
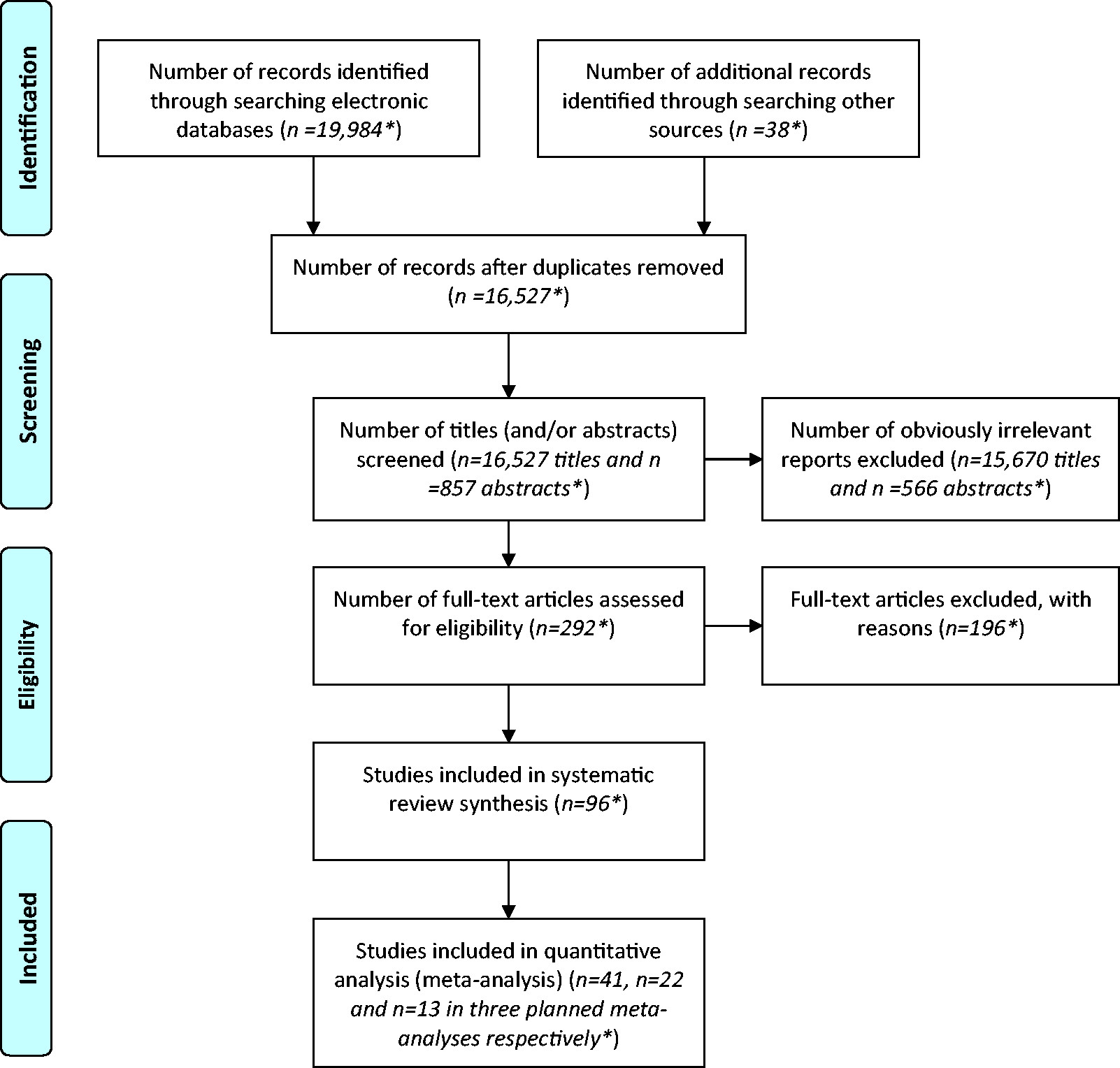
Collect data
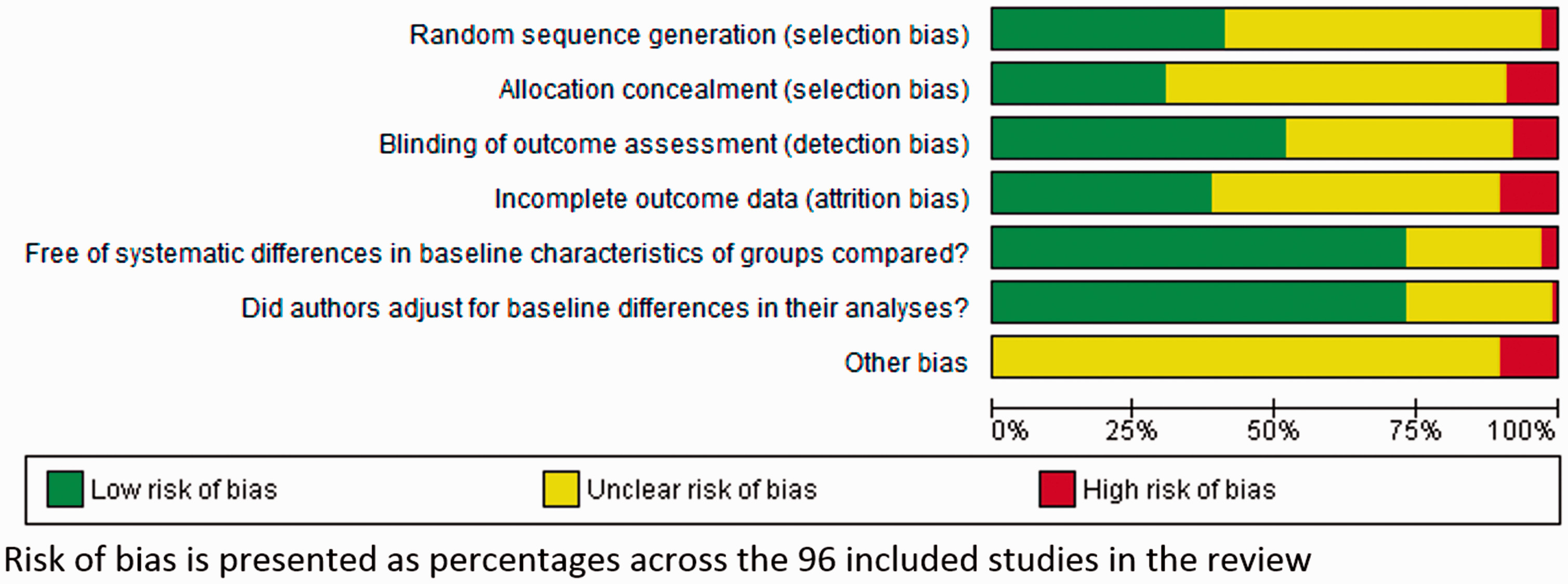
Assess quality of included studies
| Common sources of bias | Description of bias23 | Methods to avoid introduction of bias | Examples from review of physical rehabilitation approaches70 |
|---|---|---|---|
| Selection bias | The groups of participants who are being compared have differences at baseline, due to the way that participants have been allocated to groups. | Randomization (allocation of participants to groups based on a random process, or sequence, with the order of allocation concealed from all people involved in the study) | Zhu 2006a allocated participants to groups “according to time of hospital admission.” This method introduced a risk of selection bias as the characteristics of participants could vary according to time, and the researchers could potentially influence the allocation of a participant to a specific treatment group. |
| Performance bias | The groups of participants receive differences in care, other than differences in the intervention which is being tested. | Masking (blinding) of participants and personnel (concealment) to the study treatment being delivered | Dean 2000, Chan 2006 and Gelber 1995a did not use masking of person delivering the intervention, who could therefore be more enthusiastic and encouraging towards patients in the intervention group than the control group. |
| Detection bias | The way outcomes are measured in the groups of participants differs. | Masking (blinding) of outcome assessor to the study treatment being delivered | Salbach 2004a un-masked outcome assessors, introducing a high risk of detection bias. |
| Attrition bias | There are differences in retention / withdrawals between the groups of participants. | Complete data collection in both groups The reasons for missing data must be reported for each treatment group, so that any differences between groups can be explored. | Fang 2003a had more drop-outs from one group than the other. |
| Reporting bias (including publication bias, and selective outcome reporting) | There are systematic differences between reported and unreported findings. | Comprehensive searching for all eligible studies (regardless of publication status) can help avoid publication bias. Pre-specification of outcome measures within a published protocol can help avoid selective outcome reporting. Statistical methods can be used to aid detection of reporting biases (funnel plots and sensitivity analyses). | Trials published in non-English language and in Chinese journals may not all have been identified70 |
Synthesize evidence
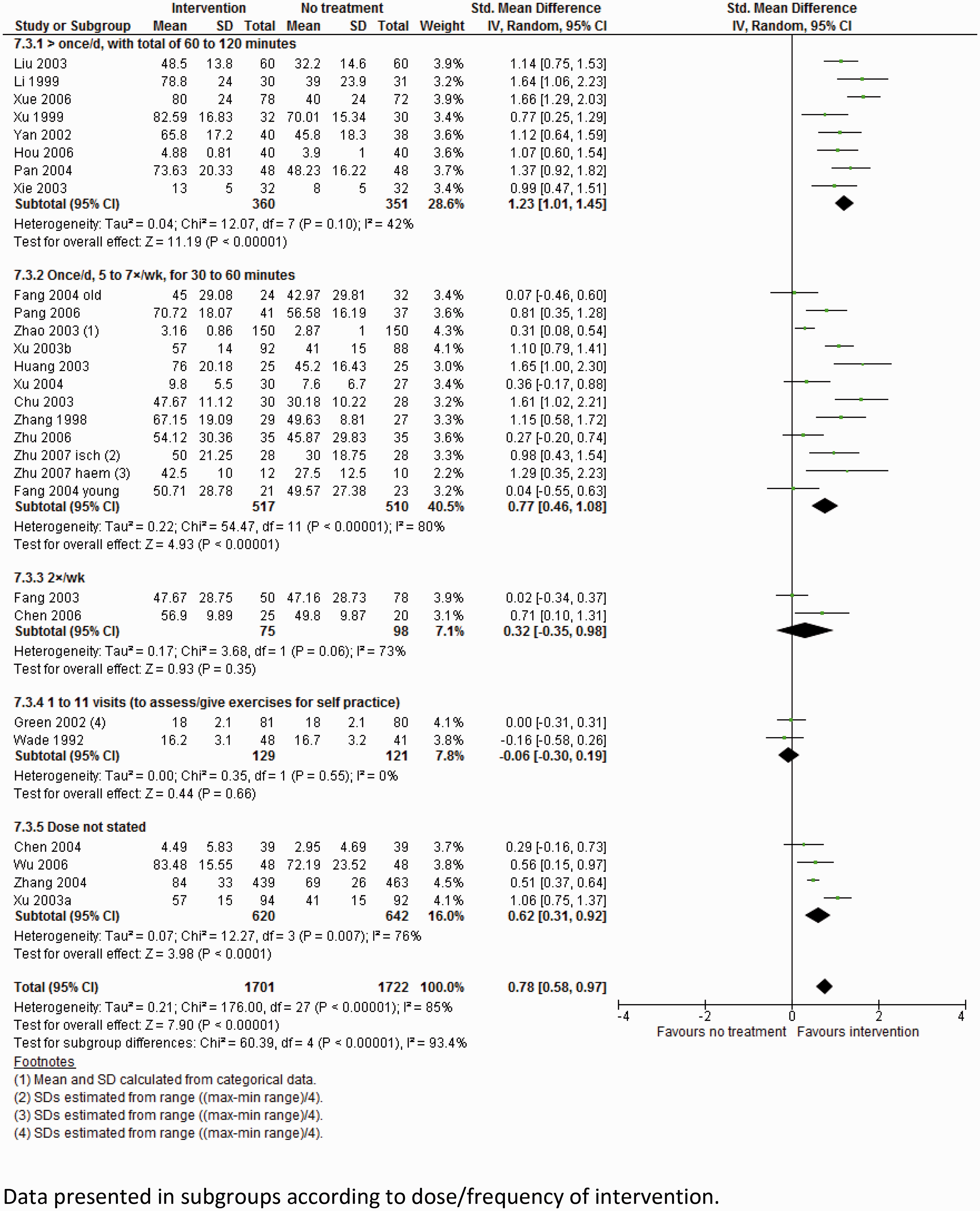
| PICO question: In patients with a recent acute stroke (less than six weeks) with reduced mobility, is any specific physiotherapy approach more beneficial than no physiotherapy at improving independence in activities of daily living and gait speed? |
| Relevant subgroup analyses to consider: • Effects of therapy given at different times after stroke (<1 week, 1–3 weeks, or 3–6 weeks). • Effects of therapy in different parts of the world (Europe, Australasia, America, Asia). • Effects of therapy at different doses/intensities (> 45 min/day, 30–45 min/day, 15–30 min/day, <5 sessions/week, <2 sessions/week). • Effects of therapy delivered by different professions (physiotherapist, nurse, assistant therapist, carer/family member). • Effect of different specific therapy approaches (e.g. Bobath approach, motor learning approach, orthopedic methods). |
Interpret findings
| PICO question: In patients with a recent acute stroke (less than six weeks) with reduced mobility is any specific physiotherapy method more beneficial than no physiotherapy at improving independence in activities of daily living and gait speed? | ||||
|---|---|---|---|---|
| Selected outcomes | Standardized mean difference (95% CI) | No. of participants (studies) | Quality of the evidence (GRADE) | Comments |
| Independence in ADL scales | 0.58 (0.11 to 1.04) | 9 studies 540 participants | ⊕⊕⊕ ⊖moderate | Quality of evidence downgraded as there was substantial statistical heterogeneity in results (I2 = 85%) |
| Gait velocity | −0.06 (−0.29 to 0.18) | 3 studies 271 participants | ⊕⊕⊖⊖low | Quality of evidence downgraded twice as dose of physiotherapy varied substantially between studies, and 1/3 studies were carried out in China (and a significant subgroup effect relating to geographical location of the study was identified) |
Complete the systematic review
After review completion: Publication, dissemination and up-dating
Conclusions
Acknowledgements
Declaration of conflicting interests
Funding
Permissions
References
Cite
Cite
Cite
Download to reference manager
If you have citation software installed, you can download citation data to the citation manager of your choice
Information, rights and permissions
Information
Published In
Keywords
Authors
Metrics and citations
Metrics
Journals metrics
This article was published in International Journal of Stroke.
View All Journal MetricsPublication usage*
Total views and downloads: 179102
*Publication usage tracking started in December 2016
Altmetric
See the impact this article is making through the number of times it’s been read, and the Altmetric Score.
Learn more about the Altmetric Scores
Publications citing this one
Receive email alerts when this publication is cited
Web of Science: 295 view articles Opens in new tab
Crossref: 373
- Toward shared situational awareness in human-AI hybrid intelligence: A survey in high-risk aviation environments
- Thermal inactivation of Cronobacter sakazakii in reconstituted powdered infant formula: Systematic review and meta-analysis
- Local content development and the resource curse in Nigeria: Comparative lessons and policy reform pathways
- User-centric dimensions of trust and acceptance in blockchain adoption for land administration systems: A systematic literature review
- Major determinants of sustainable agriculture practices adoption: A systematic review
- What we count shapes what we learn: A systematic review of age-friendly urban environments and later-life well-being
- Automated compliance checking across the building lifecycle: Systematic and semantic review integrating PRISMA and deep search
- Concept analysis: a methodological means to clarify key concepts in health professions education
- The rapid rise of soft robotics in surgical operations: Trends, challenges, and future directions
- Information Systems
- View More
Figures and tables
Figures & Media
Tables
View Options
View options
PDF/EPUB
View PDF/EPUBAccess options
If you have access to journal content via a personal subscription, university, library, employer or society, select from the options below:
loading institutional access options
World Stroke Organization members can access journal content using society membership credentials.
Alternatively, view purchase options below:
Purchase 24 hour online access to view and download content.
Access journal content via a DeepDyve subscription or find out more about this option.




