A systematic review and synthesis of global stroke guidelines on behalf of the World Stroke Organization
Abstract
Background:
Aims:
Summary of review:
Conclusion:
Background
Aims
Methods
Participants/population
Collating guidelines, extracting data, and reviewing quality
| Minimal level of resource availability | Essential service level | Advanced stroke services |
|---|---|---|
| Stroke care delivery is based at a local clinic staffed predominantly by non-physicians; laboratory tests and diagnostic studies are scarce; and much of the emphasis is placed on bedside clinical skills, teaching, and prevention | Access to a CT scan, physicians, and the potential for acute thrombolytic therapy; however, stroke expertise may still be difficult to access | Multidisciplinary stroke expertise, multimodal imaging, and comprehensive therapies are available including endovascular thrombectomy |
Stakeholder involvement
Results
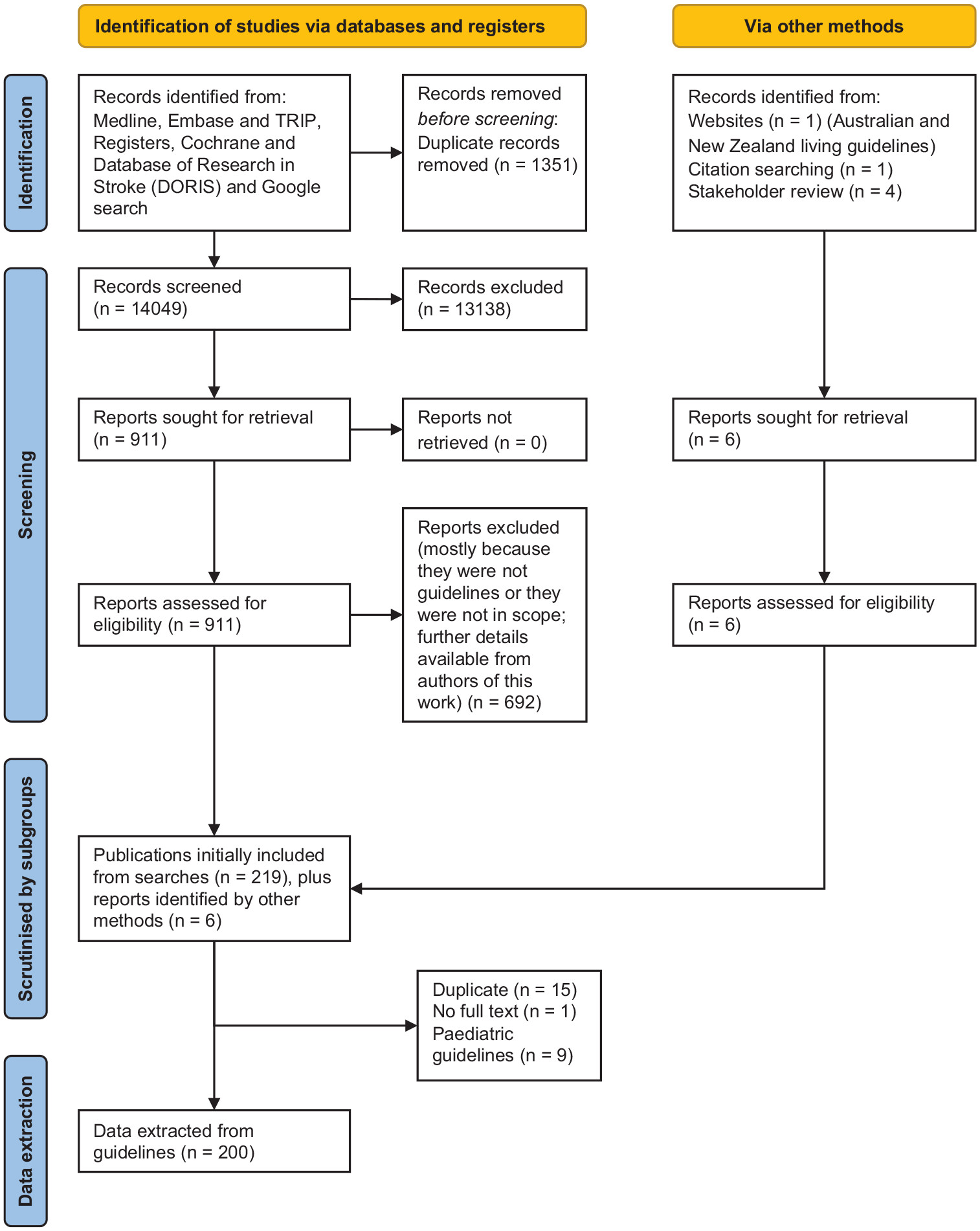
Grading systems used
Quality assessments by AGREE II
| Recommendations | Minimal system | Essential system | Advanced system |
|---|---|---|---|
| A. Early diagnosis and pre-hospital care | |||
| Public education programs about identification of stroke signs and the need to seek emergency care should be designed and implemented in the community | √ | √ | √ |
| Whenever available, emergency transportation systems should be used to reduce time to ED arrival; emergency dispatch systems should have a method to prioritize possible strokes | √ | √ | |
| First responders should (1) rapidly evaluate airway, breathing, and circulation to identify and treat a life-threatening situation; (2) use a validated pre-hospital stroke assessment tool; (3) ascertain the time of onset of stroke symptoms (from the patient or witnesses) | √ | √ | |
| Patients with a possible stroke should be immediately transported to the closest hospital capable of providing emergency stroke care, including IV thrombolysis | √ | √ | |
| Emergency responders should notify the hospital (pre-hospital notification) that a patient with a possible stroke is en-route to prepare the appropriate hospital resources | √ | √ | |
| Hospital caring for patients with acute stroke should establish protocols for emergency inter-hospital transfers | √ | √ | √ |
| Regional systems of stroke care should be developed including two major categories: (1) centers capable of providing initial emergency care, including administration of IV thrombolysis, and (2) centers capable of performing endovascular stroke treatment with comprehensive periprocedural care | √ | ||
| Stroke centers should (1) have a protocol for emergency stroke evaluation and treatment in the ED; (2) have a designated multidisciplinary stroke team (or access to stroke expertise trough telemedicine); (3) implement a strategy to monitor stroke quality metrics; (4) seek certification by an independent external accreditation entity; (5) ensure continuing stroke education | √ | √ | |
| B. Hyperacute hospital care (first hours after stroke) | |||
| Hospitals caring for patients with acute stroke should have an organized protocol for the emergency evaluation of patients with suspected stroke using a validated stroke screening tool | √ | √ | |
| Telemedicine/telestroke resources and systems should be supported by health care institutions and governments to ensure availability of stroke expertise coverage 24/7 wherever it is not available on site | √ | √ | |
| Tracheal intubation is indicated for a compromised airway or insufficient ventilation due to impaired alertness or bulbar dysfunction | √ | √ | |
| Supplemental oxygen should be provided to maintain oxygen saturation ⩾94% | √ | √ | √ |
| Hypotension and hypovolemia should be corrected to maintain systemic perfusion levels necessary to support organ function | √ | √ | √ |
| Emergency treatment of hypertension is indicated if there is concomitant acute myocardial ischemia, aortic dissection, or preeclampsia/eclampsia | √ | √ | √ |
| Capillary blood glucose should be checked immediately in suspected stroke. Hypoglycemia (glucose below 60 mg/dL or 3.3 mmol/L) should be treated with IV dextrose | √ | √ | √ |
| Electrocardiography and other blood tests (complete cell count, serum electrolytes and creatinine, INR and partial thromboplastin time, serum troponin) should be obtained, but should not delay the initiation of reperfusion therapy | √ | √ | √ |
| A stroke severity rating scale (e.g. NIHSS) should be used in the ED | √ | √ | |
| All patients with suspected acute stroke should undergo brain imaging (head CT or brain MRI) without delay upon hospital arrival and before receiving any specific treatment for stroke | √ | √ | |
| Reperfusion therapy for AIS | |||
| Patients eligible for IV thrombolysis should have the treatment initiated as soon as possible | √ | √ | |
| IV alteplase (0.9 mg/kg, maximum dose 90 mg over 60 min with initial 10% of dose given as bolus over 1 min) is recommended for selected patients who can be treated within 4.5 h of ischemic stroke symptom onset or last known well. Contraindications for IV thrombolysis are shown in (Supplemental Appendix 4) | √ | √ | |
| In patients with AIS who awake with stroke symptoms or have unclear time of onset >4.5 h from last known well, IV alteplase administered within 4.5 h of stroke symptom recognition can be beneficial if MRI shows DWI-FLAIR mismatch | √ | ||
| For patients with AIS within 4.5–9 h of symptom onset who have CT or MRI core/perfusion mismatch, and for whom mechanical thrombectomy is either not indicated or not planned, consider intravenous thrombolysis with alteplase | √ | ||
| Only the assessment of blood glucose must precede the initiation of IV alteplase in all patients | √ | √ | |
| Patients with AIS and acute hypertension who are otherwise eligible for IV thrombolysis should have their BP lowered below 185/110 mm Hg before IV thrombolysis is initiated | √ | √ | |
| Eligible patients should receive IV thrombolysis even if mechanical thrombectomy is being considered. Do NOT evaluate response to IV thrombolysis before proceeding with catheter angiography for mechanical thrombectomy | √ | √ | |
| Patients with clinically suspected LVO should have non-invasive angiography (e.g. CTA) | √ | √ | |
| Patients with AIS within 6–24 h of time last known well who have a LVO in the anterior circulation should have advanced imaging (CTP or DW-MRI, with or without MRI perfusion) to determine eligibility for mechanical thrombectomy | √ | ||
| Patients should receive mechanical thrombectomy with a stent retriever or with direct aspiration if they meet all the following criteria: (1) age ⩾ 18 years; (2) pre-stroke mRS score of 0–1; (3) causative occlusion of the internal carotid artery or MCA (M1); (4) NIHSS score of ⩾ 6; (5) ASPECTS of ⩾ 6; and (6) treatment can be initiated (groin puncture) within 6 h of symptom onset or last known well | √ | ||
| Mechanical thrombectomy is also recommended between 6 and 24 h in patients who have sizable mismatch between ischemic core (by CTP or MRI-DWI) and either clinical deficits or area of hypoperfusion (by CTP or MRI-PWI) | √ | ||
| Mechanical thrombectomy can be considered in patients with an occlusion or stenosis of the cervical ICA in addition to an intracranial LVO | √ | ||
| The technical goal of mechanical thrombectomy should be reperfusion to a modified Thrombolysis in Cerebral Infarction (mTICI) grade 2b/3 | √ | ||
| Intracerebral hemorrhage | |||
| In patients with lobar spontaneous ICH and age < 70 years, deep/posterior fossa spontaneous ICH and age < 45 years, or deep/ posterior fossa and age 45–70 years without history of hypertension, head CTA plus consideration of venography is recommended to exclude macrovascular causes or cerebral venous thrombosis | √ | √ | |
| In patients with spontaneous IVH and no detectable parenchymal hemorrhage, catheter angiography is recommended to exclude a vascular anomaly | √ | ||
| In patients with spontaneous ICH and non-invasive angiography suggestive of a vascular anomaly, catheter angiography is recommended to confirm and, if possible, treat the vascular anomaly | √ | ||
| In case of IVH and hydrocephalus that is contributing to decreased level of consciousness, external ventricular drainage is recommended | √ | ||
| In patients with spontaneous ICH and hypertension presenting within 6 h of symptom onset, we recommend acute lowering of SBP to a target of 140 mm Hg (strictly avoiding SBP < 110 mm Hg) to reduce the risk of hematoma expansion | √ | √ | √ |
| In patients with anticoagulant-associated spontaneous ICH, anticoagulation should be discontinued immediately, and anticoagulation should be reversed as soon as possible | √ | √ | √ |
| In patients with VKA-associated spontaneous ICH and INR ⩾ 2.0, 4-factor (4F) prothrombin complex concentrate (PCC) is recommended over fresh-frozen plasma (FFP). FFP or 3F-PCC should be used when 4F-PCC is not available. IV vitamin K should be administered shortly after 4F-PCC or FFP to prevent later re-emergence of anticoagulation | √ | √ | |
| For heparin-related ICH, protamine sulfate is recommended | √ | √ | |
| In patients with ICH who were taking a direct oral anticoagulant, idarucizumab is recommended for reversal of dabigatran and andexanet alpha or, if not available, 4F-PCC for reversal of factor Xa inhibitors | √ | ||
| Cerebral venous sinus thrombosis | |||
| A non-invasive venogram (CTV or MRV) should be performed in suspected CVST if the plain CT or MRI are inconclusive | √ | √ | |
| In patients with suspected or confirmed CVST, possible causative infections should be excluded | √ | √ | √ |
| Anticoagulation should be started immediately after the diagnosis of CVST, even if intracranial hemorrhage is present. IV heparin or subcutaneous LMWH can be used | √ | √ | √ |
| C. Acute In-Hospital care (first days after stroke)-medical management (for organization of care, dysphagia, mobilization, skin care, and continence, see rehabilitation Table 3) | |||
| Patients should be admitted to a stroke unit (a specialized, geographically defined hospital unit dedicated to the management of stroke patients) or, if critically ill, to an intensive care unit (see also rehabilitation recommendations) | √ | √ | |
| Cardiac monitoring is recommended to screen for atrial fibrillation and other potentially serious cardiac arrhythmias for at least the first 24 h | √ | √ | |
| In patients with AIS, blood pressure should be maintained below 180/105 mm Hg for at least the first 24 h after acute reperfusion treatment | √ | √ | |
| In patients with AIS, administration of aspirin is recommended within 24–48 h after stroke onset. For those treated with IV thrombolysis, aspirin administration is generally delayed until > 24 h. Patients with aspirin allergy should be given an alternative antiplatelet medication | √ | √ | √ |
| Gradual early mobilization should be encouraged. Patients who have limited mobility should be treated with thigh-high intermittent pneumatic compression devices (IPC), if available | √ | √ | √ |
| Body temperature should be monitored and fever (temperature > 38°C) should be treated. Sources of fever should be investigated and treated | √ | √ | √ |
| Antiseizure medications are only indicated for documented secondary seizures | √ | √ | √ |
| Massive Stroke | |||
| The management of patients with massive stroke should be reached by shared decision making with participation of patient (when possible) and family taking into consideration the anticipated prognosis for functional recovery | √ | √ | √ |
| Patients with massive cerebral or cerebellar infarction or hemorrhage or at risk of malignant swelling should be rapidly transferred to a center with neurosurgical expertise if their condition is deemed survivable | √ | √ | √ |
| In patients with massive strokes, serial physical examinations (and repeat head CT scan when appropriate) should be performed to identify worsening brain swelling | √ | √ | |
| Patients with massive strokes should be immediately intubated if they develop neurological deterioration with respiratory insufficiency | √ | √ | |
| Decompressive hemicraniectomy is indicated within 48 h of symptom onset in patients with massive hemispheric infarction and worsening neurological condition (functional benefit much greater in patients < 60 years) | √ | ||
| Ventriculostomy is recommended in the treatment of symptomatic obstructive hydrocephalus after cerebellar infarction. Concomitant or subsequent decompressive suboccipital craniectomy is indicated if brainstem compression is present | √ | ||
| Patients with spontaneous ICH (with or without IVH) and symptomatic hydrocephalus should be treated with ventricular drainage | √ | ||
| Patients with cerebellar ICH who develop neurological deterioration, have brainstem compression, and/or have hydrocephalus from ventricular obstruction should be treated with decompressive suboccipital craniectomy (with or without ventricular drainage) | √ | ||
| Recommendations | Minimal | Essential | Advanced |
|---|---|---|---|
| Diagnosis | |||
| Etiological diagnosis | |||
| A diagnostic evaluation of stroke etiology should be started or ideally completed within 48 h of stroke onset | √ | √ | |
| Blood work | |||
| Blood work including complete blood count, prothrombin time, partial thromboplastin time, glucose, HbA1c, creatinine, and fasting or non-fasting lipid profile, is recommended in patients with ischemic stroke or TIA | √ | √ | √ |
| Cerebral imaging | |||
| In patients with a suspicion of an ischemic cerebrovascular event (stroke or TIA), neuroimaging with CT or MRI is recommended | √ | √ | |
| Vascular imaging | |||
| Vascular imaging of the extracranial cervical arteries is recommended to identity patients with severe internal carotid artery stenosis who may benefit from urgent carotid endarterectomy or stenting. Imaging can be performed with carotid Doppler ultrasound, CTA, or MRA | √ | √ | |
| Electrocardiographic monitoring | |||
| Patients with an ischemic stroke or TIA should have an ECG to screen for atrial fibrillation | √ | √ | √ |
| Patients with an ischemic stroke or TIA should have at least 24 h of cardiac monitoring to screen for atrial fibrillation | √ | ||
| Patients with an embolic ischemic stroke or TIA without atrial fibrillation on initial short-term ECG should have longer-term monitoring, for at least 14 days to screen for atrial fibrillation | √ | ||
| Hypertension | |||
| Need for blood pressure management | |||
| Blood pressure should be assessed and treated in all patients with ischemic and hemorrhagic stroke and TIA | √ | √ | √ |
| Timing of blood pressure treatment | |||
| Blood pressure treatment should be initiated as soon as possible after a stroke or TIA, or at least before discharge | √ | √ | √ |
| Selection of antihypertensive drugs | |||
| Angiotensin-converting enzyme-inhibitors combined with a thiazide diuretic can reduce the risk of stroke in ischemic stroke or TIA patients with and without a diagnosis of hypertension and are therefore favored in patients with ischemic stroke or TIA | √ | √ | √ |
| In patients with ischemic stroke or TIA and hypertension, any of the following antihypertensive drugs can be used: thiazide diuretic, angiotensin-converting enzyme inhibitor, or angiotensin II receptor. Beta-blockers may be used in patients with ischemic heart disease | √ | √ | √ |
| In patients with ischemic stroke or TIA and hypertension, an individualized approach to the selection of antihypertensive medications based on comorbidities is recommended | √ | √ | √ |
| Blood pressure targets | |||
| The target blood pressure for patients with stroke or TIA is < 130/80 mm Hg | √ | √ | √ |
| Weight | |||
| In patients with ischemic stroke or TIA, it is recommended to estimate the BMI at the time of the event and during long-term follow-up | √ | √ | √ |
| In patients with ischemic stroke or TIA who are overweight or obese, weight loss is recommended | √ | √ | √ |
| In patients with ischemic stroke or TIA who are obese, referral to a multidisciplinary lifestyle modification program is recommended | √ | ||
| Diabetes | |||
| In patients with ischemic stroke or TIA and diabetes, a target of HbA1c ⩽ 7% is recommended | √ | √ | √ |
| In patients with ischemic stroke or TIA and diabetes, the treatment of diabetes should include glucose-lowering agents with demonstrated efficacy for reducing vascular outcomes | √ | √ | √ |
| In patients with ischemic stroke or TIA and diabetes, a transdisciplinary team approach is recommended | √ | ||
| Lipids | |||
| Ischemic stroke and TIA patients without a proven cardioembolic mechanism and an LDL-cholesterol level > 2.5 mmol/L (> 100 mg/dL) should receive atorvastatin 80 mg to reduce stroke recurrence | √ | √ | √ |
| The target LDL-cholesterol level in patients with ischemic stroke and TIA should be < 1.8 mmol/L (70 mg/dL) | √ | √ | √ |
| The target LDL-cholesterol level in patients with ischemic stroke and TIA and atherosclerotic disease of the extracranial or intracranial arteries should be 1.8 mmol/L (70 mg/dL). Ezetimibe can be added to Atorvastatin to reach this goal | √ | √ | √ |
| In patients with ischemic stroke or TIA in whom a target LDL-cholesterol level is not achievable, a consideration should be made to refer to an expert in lipid management for adding a PCSK9 inhibitor | √ | ||
| In patients with ischemic stroke or TIA on lipid-lowering medications, lipid levels should be monitored 1–3 months after treatment initiation, followed by regular assessments and dose adjustments every 3–12 months thereafter | √ | √ | √ |
| Lifestyle modification | |||
| Physical activity | |||
| In patients with ischemic stroke or TIA who can engage in physical activity, low/moderate-intensity aerobic activity for 10 min 4 days/week, or vigorous aerobic activity for 20 min twice a week is recommended | √ | √ | √ |
| Smoking cessation | |||
| In patients with ischemic stroke or TIA, smoking status should be evaluated and documented in all health care encounters | √ | √ | √ |
| In patients with ischemic stroke or TIA who are active smokers, counseling with or without pharmacological therapy with nicotine replacement, bupropion, or varenicline is recommended | √ | √ | √ |
| In patients with ischemic stroke or TIA who are active smokers, a referral to a smoking cessation clinic is recommended if available | √ | ||
| Alcohol consumption | |||
| In patients with ischemic stroke or TIA who drink > 2 alcoholic drinks daily for men or > 1 alcoholic drink daily for women, counseling for alcohol intake reduction is recommended. Specialized services are also recommended | √ | √ | √ |
| In patients with ischemic stroke or TIA with alcohol used disorder, specialized services are recommended | √ | ||
| Recreational drugs | |||
| In patients with ischemic stroke or TIA who use stimulant recreational drugs, counseling is recommended | √ | √ | √ |
| In patients with ischemic stroke or TIA who use recreational drugs, specialized services are recommended | √ | ||
| Salt consumption | |||
| In patients with ischemic stroke or TIA, salt intake should be reduced, at least < 2000 mg daily | √ | √ | √ |
| Atrial fibrillation | |||
| Oral anticoagulation | |||
| In patients with ischemic stroke or TIA and valvular (mechanical valve replacement or moderate/severe mitral stenosis) atrial fibrillation, oral anticoagulation is recommended | √ | √ | √ |
| In patients with ischemic stroke or TIA with nonvalvular atrial fibrillation or flutter, either paroxysmal, persistent, or permanent, oral anticoagulation is recommended | √ | √ | √ |
| In patients with ischemic stroke or TIA with nonvalvular atrial fibrillation, direct oral anticoagulants are preferred over vitamin-K antagonists | √ | √ | √ |
| In patients with ischemic stroke or TIA with nonvalvular atrial fibrillation, who are receiving vitamin-K antagonists and cannot achieve a consistent INR level, direct oral anticoagulants are preferred over vitamin-K antagonists | √ | √ | |
| Antiplatelet therapy | |||
| Patients who are suitable for anticoagulation should not receive antiplatelets for secondary stroke prevention | √ | √ | √ |
| Valvular heart disease | |||
| In patients with a mechanical mitral valve and a previous ischemic stroke or TIA before the valve replacement, add aspirin 75–100 mg daily to warfarin targeting an INR range of 2.5–3.5 | √ | √ | √ |
| In patients with ischemic stroke or TIA who have native aortic or nonrheumatic mitral valve disease and no AF or any other indication for anticoagulation, antiplatelet agents are recommended | √ | √ | √ |
| In patients with ischemic stroke or TIA who have a bioprosthetic aortic or mitral valve replacement without any other indication for anticoagulation, long-term antiplatelet therapy is recommended beyond the 3- to 6-month window after the procedure | √ | √ | √ |
| Left ventricular and atrial thrombus | |||
| In patients with ischemic stroke or TIA and a left ventricular thrombus, anticoagulation at least for 3 months is recommended | √ | √ | √ |
| In patients with ischemic stroke or TIA and left atrial or left atrial appendage thrombus in the context of ischemic, nonischemic, or restrictive cardiomyopathy and LV dysfunction, oral anticoagulation with vitamin-K antagonists is recommended for at least 3 months | √ | √ | √ |
| Patent foramen ovale | |||
| Neurocardiology assessment | |||
| In patients with a cryptogenic stroke and a PFO, a team-based approach by a cardiologist and a neurologist is recommended to identify the causal role of the PFO and to define the best therapeutic approach | √ | ||
| PFO closure | |||
| Patients with non-lacunar ischemic stroke and a PFO should undergo PFO closure if they are aged between 18 and 60 years, if PFO is considered causal | √ | ||
| Atherosclerotic extracranial large vessel disease | |||
| Urgent evaluation | |||
| Patients with an acute ischemic stroke or TIA and ipsilateral internal carotid artery stenosis of 50–99% should be evaluated urgently by an expert team to decide carotid revascularization | √ | ||
| Carotid endarterectomy | |||
| Patients with an acute ischemic stroke or TIA in the past 6 months and ipsilateral extracranial internal carotid artery stenosis of 70–99% should be offered a carotid endarterectomy if the morbidity/mortality risk of the surgical team is < 6% | √ | ||
| Patients with an acute ischemic stroke or TIA in the past 6 months and ipsilateral extracranial internal carotid artery stenosis of 50–69% could be offered a carotid endarterectomy depending on individual characteristics, including age, sex, and comorbidities if the morbidity/mortality risk of the surgical team is < 6%. The benefit of carotid endarterectomy in patients with 50–69% stenosis is substantially lower than in those with ⩾ 70%. In patients with 50–69% stenosis, carotid endarterectomy is associated with a higher risk of poor outcomes in the first 2 post-procedural years. However, there is a significant benefit from surgery for any stroke or operative death at 5 years.203 As such, an estimated life expectancy > 5 years is required in this group. In addition, there was no clear benefit of carotid endarterectomy in the NASCET trial for women with 50–69% stenosis204 | √ | ||
| Carotid endarterectomy if indicated, should be performed as early as possible if the patient is clinically stable, ideally within 14 days after symptoms onset | √ | ||
| Carotid endarterectomy is preferred over carotid stenting | √ | ||
| Carotid stenting | |||
| Carotid stenting may be considered for patients who are NOT candidates for a carotid endarterectomy (e.g. technical, anatomic, or medical reasons) | √ | ||
| Medical therapy | |||
| In patients with ischemic stroke or TIA and carotid or vertebral artery stenosis, intensive medical therapy (e.g. antiplatelet agents, lipid-lowering medications, blood pressure management, and diabetes control) is recommended, regardless of whether a revascularization procedure is done, in addition to diet, exercise, and smoking cessation | √ | √ | √ |
| In patients with ischemic stroke or TIA and aortic arch atheroma, antiplatelet therapy is recommended | √ | √ | √ |
| In patients with ischemic stroke or TIA and aortic arch atheroma, a target LDL-cholesterol of 1.8 mmol/L (70 mg/dL) should be pursued with high-dose statin therapy | √ | √ | √ |
| Intracranial atherosclerotic disease | |||
| Anticoagulation | |||
| In patients with acute ischemic stroke or TIA due to high-grade intracranial atherosclerotic disease, the use of anticoagulants is not recommended unless there is another indication for anticoagulation (e.g. atrial fibrillation) | √ | √ | √ |
| Single antiplatelet therapy | |||
| In patients with ischemic stroke or TIA caused by moderate to high-grade intracranial atherosclerotic stenosis (50–99%), aspirin 325 mg daily is recommended over oral anticoagulation. There are no strong recommendations supporting the use of dual antiplatelet therapy (DAPT) over single antiplatelet therapy (SAPT) in this population. While the Stenting vs Aggressive Medical Therapy for Intracranial Arterial Stenosis (SAMMPRIS) trial showed that DAPT is better than stenting, it did not prove that DAPT is better than SAPT.205 A post hoc analysis of the Clopidogrel in High-Risk Patients with Acute Non-disabling Cerebrovascular Events (CHANCE) trial showed no differences in the beneficial effect of DAPT vs SAPT in minor stroke patients with vs without intracranial atherosclerotic disease (ICAD).206 In the Clopidogrel Plus Aspirin Versus Aspirin Alone for Reducing Embolization in Patients With Acute Symptomatic Cerebral or Carotid Artery Stenosis (CLAIR) Trial, 93 of 100 patients had symptomatic ICAD. DAPT use was associated with a 54.4% (16.4–75.1) relative risk reduction on microembolic signals on transcranial Doppler ultrasound.207 | √ | √ | √ |
| Blood pressure management | |||
| In patients with ischemic stroke or TIA caused by moderate to high-grade intracranial atherosclerotic stenosis (50–99%), a systolic blood pressure target of < 140 mm Hg is recommended | √ | √ | √ |
| Lipid-lowering agents | |||
| In patients with ischemic stroke or TIA caused by moderate to high-grade intracranial atherosclerotic stenosis (50–99%), high-dose statin therapy is recommended | √ | √ | √ |
| Physical activity | |||
| In patients with ischemic stroke or TIA caused by moderate to high-grade intracranial atherosclerotic stenosis (50–99%), at least moderate physical activity is recommended | √ | √ | √ |
| Angioplasty and stenting | |||
| In patients with ischemic stroke or TIA and moderate to high-grade intracranial atherosclerotic stenosis (50–99%), angioplasty and stenting is not recommended. Dual antiplatelets is an appropriate medical therapy | √ | √ | √ |
| Antithrombotic management in non-cardioembolic stroke | |||
| Indication for antiplatelet therapy | |||
| In patients with non-cardioembolic ischemic stroke or TIA who do not require anticoagulation, long-term antiplatelet therapy is indicated | √ | √ | |
| Selection of single antiplatelet agents | |||
| Antiplatelet agents are recommended for secondary stroke prevention in patients with non-cardioembolic ischemic events who do not require oral anticoagulation, including aspirin 81–325 mg daily, clopidogrel 75 mg daily, or aspirin + dipyridamole 25/200 mg daily | √ | √ | √ |
| In patients with an acute ischemic stroke or TIA who were not on an antiplatelet agent, a single loading dose of 160 mg should be administered after an intracranial hemorrhage is ruled out on neuroimaging studies | √ | √ | |
| In patients with an acute ischemic stroke or TIA in whom swallowing mechanisms are impaired, rectal aspirin 325 mg daily, or aspirin 81 mg daily or clopidogrel 75 mg daily administered via enteral tube are reasonable alternatives to oral intake | √ | √ | |
| Minor ischemic stroke or TIA | |||
| In patients with a minor ischemic stroke (NIHSS ⩽ 3) or high-risk TIA (ABCD2 ⩾ 4), DAPT with aspirin 81 mg daily and clopidogrel 75 mg daily should be initiated as early as possible, ideally within 12–24 h of symptoms onset, after an intracranial hemorrhage is excluded on neuroimaging studies. A single loading dose of aspirin (160–325 mg) and clopidogrel (300 mg as per the CHANCE trial or 600 mg as per the POINT trial) should be used at the beginning of DAPT therapy. DAPT is indicated for 21 days and should be followed by long-term single antiplatelet therapy with aspirin 81 mg daily or clopidogrel 75 mg daily | √ | √ | |
| In patients with mild-moderate ischemic stroke (NIHSS ⩽ 5) or high-risk TIA (ABCD2 ⩾ 4) DAPT with aspirin 75–100 mg daily and ticagrelor 90 mg twice daily should be initiated as early as possible, ideally within 24 h of symptoms onset, after an intracranial hemorrhage is excluded on neuroimaging studies. A single loading dose of aspirin (300–325 mg) and ticagrelor (180 mg) should be used at the beginning of DAPT therapy. DAPT is indicated for 30 days and should be followed by long-term single antiplatelet therapy | √ | √ | |
| Embolic stroke of undetermined source (ESUS) | |||
| Patients with ESUS should not receive oral anticoagulants. The recommended antithrombotic regimen for secondary stroke prevention in ESUS patients is antiplatelet therapy | √ | √ | |
| Extracranial artery dissection | |||
| In patients with ischemic stroke or TIA and extracranial carotid or vertebral artery dissection, either antiplatelet therapy or oral anticoagulants are recommended for at least 3 months | √ | √ | |
| Carotid web | |||
| In patients with ischemic stroke or TIA and a carotid web in the ipsilateral vascular territory, antiplatelet therapy is recommended | √ | √ | |
| Fibromuscular dysplasia | |||
| In patients with ischemic stroke or TIA and fibromuscular dysplasia, antiplatelet therapy and lifestyle modification are recommended | √ | √ | |
| Positive anti-phospholipid antibodies | |||
| In patients with ischemic stroke or TIA and positive anti-phospholipid who do not fulfill criteria for anti-phospholipid syndrome, antiplatelet therapy is recommended. There are no strong recommendations for patients with confirmed anti-phospholipid syndrome regarding antithrombotic therapy | √ | √ | |
| Sickle cell disease | |||
| In patients with ischemic stroke or TIA and sickle cell disease, chronic blood transfusions are recommended to reduce hemoglobin S to < 30% of total hemoglobin | √ | √ | |
| Vasculitis | |||
| In patients with ischemic stroke or TIA and symptoms of giant cell arteritis, immediate treatment with high-dose steroids should be initiated | √ | √ | √ |
| In patients with ischemic stroke or TIA and infectious vasculitis (VZV, bacterial, or other infectious agents), the underlying infection should be treated | √ | √ | √ |
| Spontaneous intracerebral hemorrhage | |||
| Aggressive long-term blood pressure monitoring, treatment, and control are recommended | √ | √ | √ |
| Cerebral venous sinus thrombosis | |||
| Anticoagulation with warfarin (or dabigatran) is recommended for a minimum of 3–6 months | √ | √ | √ |
| Recommendation | Minimal | Essential | Advanced |
|---|---|---|---|
| Organization of hospital care and principles of rehabilitation | |||
| Acute stroke patients admitted to hospital should have an initial assessment by rehabilitation professionals as soon as possible after admission | √ | √ | |
| Stroke patients should be treated on a specialized, geographically defined stroke rehabilitation unit, with coordinated care, staffed by an interdisciplinary rehabilitation team (physicians, nurses, physiotherapists, occupational therapists, speech-language therapists, and social workers and dieticians) with expertise/training in stroke rehabilitation, recovery, and return to work | √ | √ | |
| Patient, family, and caregiver education should be provided formally and informally | √ | √ | √ |
| Person-centered, collaborative, goal setting with patients and their families is recommended, clearly communicated and documented, regularly reviewed, including around transitions of care | √ | √ | √ |
| Rehabilitation should include as much scheduled task-specific therapy as possible, to meet optimal recovery and tolerability | √ | √ | |
| Commence mobilization within 48 h of stroke onset unless otherwise contraindicated but do not start intensive out-of-bed activities within 24 h of stroke onset | √ | √ | |
| Stroke patients and their families/carers should have access to specialist palliative care teams and care consistent with the principles and philosophies of palliative care | √ | √ | |
| Skin care | |||
| Regular skin assessments are recommended with objective scales of risk, e.g. Braden scale | √ | √ | √ |
| Minimize skin friction and skin pressure, provide appropriate support surfaces, avoid excessive moisture, maintain adequate nutrition and hydration to prevent skin breakdown | √ | √ | √ |
| Regular turning, good skin hygiene, and use of specialized mattresses, wheelchair cushions, and seating are recommended until mobility returns | √ | √ | √ |
| Management of dysphagia and provision of food and fluids | |||
| Health care professionals should regularly monitor and reassess patients with dysphagia who need modified food and liquid | √ | √ | √ |
| Ensure good oral and dental (including dentures) hygiene, particularly for those with dysphagia, through assistance and/or education | √ | √ | √ |
| For stroke survivors with swallowing difficulties, behavioral approaches such as swallowing exercises (e.g. shaker exercises, or chin tuck against resistance), environmental modifications, safe swallowing advice, and appropriate dietary modifications should be used early | √ | √ | √ |
| Offer swallowing therapy, e.g. compensatory strategies, exercises and postural advice, at least 3 times a week to people with dysphagia after stroke who are able to participate, for as long as they make functional gains. | √ | √ | |
| For patients whose nutrition status is poor or deteriorating, nutrition supplementation should be offered. Enteral diet should be started if required within 7 days of admission (preferably within 24–48 h) | √ | √ | |
| Patients who require tube feeding should receive percutaneous endoscopic gastrostomy rather than nasogastric tube in the post-acute phase | √ | √ | |
| Aerobic training | |||
| Individually tailored aerobic training involving large muscle groups (with monitoring of heart rate and blood pressure) should be incorporated into a comprehensive stroke rehabilitation program to enhance cardiovascular endurance and cognitive function | √ | √ | √ |
| Exercise is needed at least 3 times weekly for a minimum of 8 weeks, progressing as tolerated to 20 min or more per session, exclusive of warm-up and cool-down | √ | √ | √ |
| Use strategies to address specific barriers to physical activity related to patients, health care providers, family, and/or the environment | √ | √ | √ |
| Group circuit class therapy should be used to increase scheduled therapy time | √ | √ | |
| Offer early supported discharge services for those with mild to moderate disability | √ | √ | |
| Provide advice on prescribed medications for people after stroke | √ | √ | √ |
| Support and educate people after stroke and their families and carers, in relation to emotional adjustment to stroke, recognizing that psychological needs may change over time and in different settings | √ | √ | √ |
| Specific impairments: upper and lower limb | |||
| Stroke survivors with difficulty walking should undertake tailored repetitive practice of walking (or components of walking) using circuit class therapy (with a focus on overground walking practice and/or Treadmill training with or without body weight support) | √ | √ | |
| Rhythmic auditory stimulation (RAS) could be considered for improving gait parameters, including gait velocity, cadence, stride length, and gait symmetry | √ | ||
| For reduced strength in arms or legs, provide progressive resistance training which is meaningful, engaging, repetitive, progressively adapted, task-specific, and goal-oriented | √ | √ | |
| Non-invasive brain stimulation, including repetitive transcranial magnetic stimulation (rTMS) could be considered as an adjunct to upper extremity therapy | √ | ||
| For stroke survivors with some active wrist and finger extension, traditional or modified intensive constraint-induced movement therapy should be provided | √ | √ | |
| For stroke survivors who have difficulty sitting, practising reaching beyond arm’s length while sitting with supervision/ assistance should be undertaken | √ | √ | √ |
| For stroke survivors who have difficulty in standing up from a chair, practice of standing up should be undertaken | √ | √ | √ |
| For stroke survivors who have difficulty with standing, activities that challenge balance should be provided | √ | √ | √ |
| Force platform biofeedback should be used for people with difficulty standing | √ | √ | |
| Consider mirror therapy for patients with very severe paresis | √ | √ | |
| Functional Electrical Stimulation (FES) of the wrist and forearm muscles should be considered to reduce motor impairment and improve function and for people with gait disturbance | √ | ||
| Virtual reality, including both immersive technologies such as head mounted or robotic interfaces and non-immersive technologies such as gaming devices can be used as adjunct tools to other rehabilitation therapies, to provide additional opportunities for engagement, feedback, repetition, intensity, and task-oriented training | √ | ||
| Chemo-denervation using botulinum toxin can be used to increase range of motion and decrease pain for patients with focal symptomatically distressing spasticity (upper and lower limbs) | √ | ||
| Ankle-foot orthoses should be used on selected patients with foot drop following proper assessment and with follow-up to verify effectiveness | √ | √ | √ |
| Mental Practice should be considered for upper and low limb motor re-training | √ | √ | |
| Vision | |||
| Screen for visual problems | √ | √ | |
| For visual neglect, use interventions focused on functional tasks | √ | √ | |
| Refer people with double vision for formal orthoptic assessment | √ | √ | |
| Aphasia | |||
| Provide speech and language therapy, within 4 weeks post-stroke, to improve functional communication, communication aids, reading comprehension, general expressive language, written language and supported conversation techniques for potential communication partners | √ | √ | |
| Adapt all written material for people with aphasia | √ | √ | |
| Help and enable people with communication problems to communicate everyday needs and wishes and to support decision making | √ | √ | √ |
| Prevention and treatment of shoulder subluxation | |||
| Health care staff, patients, and family should be educated to correctly protect, position, and handle the involved arm | √ | √ | √ |
| Taping of the affected shoulder can reduce pain | √ | √ | √ |
| Mood disorders | |||
| Patients with acute stroke should be screened for depression using a structured depression assessment tool | √ | √ | √ |
| Treatment for post-stroke depression may include psychotherapy and/or antidepressants | √ | √ | |
| For severe, persistent, or troublesome tearfulness, emotional incontinence or lability, consider a trial of antidepressants | √ | √ | |
| Assessment and management of cognitive functions | |||
| Treat hypertension to reduce the risk of cognitive decline | √ | √ | √ |
| Use interventions for visual neglect after stroke that focus on the relevant functional tasks | √ | √ | |
| Continence | |||
| For urinary incontinence, identify and manage its cause (e.g. infection, constipation, urinary retention, overactive bladder) | √ | √ | √ |
| For fecal incontinence, identify and manage its cause (e.g. infection, fecal impaction, and overflow) | √ | √ | √ |
| Prolonged use of indwelling bladder catheters should be avoided as much as possible to reduce risk of urinary tract infection | √ | √ | √ |
| A bowel management program should be implemented for persistent constipation or bowel incontinence | √ | √ | √ |
| Neuropathic pain | |||
| Offer a choice of amitriptyline, duloxetine, gabapentin, or pregabalin as initial treatment for neuropathic pain; if the initial treatment is not effective or is not tolerated, offer one of the remaining three drugs, and consider switching again if the second and third drugs tried are also not effective or not tolerated. Consider capsaicin cream for people with localized neuropathic pain who wish to avoid, or who cannot tolerate, oral treatments | √ | √ | |
| Discharge from hospital and ongoing care | |||
| Before discharge, agree a health and social care plan with the patient and family/carer; establish they have a safe and enabling home environment, check carers have the support they need | √ | √ | √ |
| People with disabilities after stroke should be followed up within 72 h by the specialist stroke rehabilitation teams | √ | √ | |
| People with ongoing rehabilitation goals and those with difficulty performing activities of daily living should have ongoing access to specialized stroke services after leaving hospital | √ | √ | |
| A planned transition from structured aerobic exercise (in hospital) to more self-directed physical activity at home or in the community should be implemented | √ | √ | √ |
| Encourage participation in evidence-based community exercise programs | √ | √ | |
| Review health and social care needs of people after stroke and their caregivers at 6 months and annually thereafter. This includes people in care homes | √ | √ | |
| Hospitals should maintain a data collection system to monitor performance metrics. Participation in an external stroke data repository is also recommended | √ | √ | |
| Performance metrics |
|---|
| Acute Management |
| Time from stroke onset to assessment by health care professional (in min/h) |
| Proportion of stroke and TIA evaluated with stroke severity rating scale |
| Proportion of stroke and TIA patients receiving a CT scan within 25 min of hospital arrival |
| Door-to-needle time for ischemic stroke patients who receive IV thrombolysis |
| Proportion of ischemic stroke patients who are treated with IV thrombolysis |
| Proportion of eligible ischemic stroke patients who receive mechanical thrombectomy |
| Proportion of eligible ischemic stroke patients who receive mechanical thrombectomy in the late time window (>6 h) |
| Proportion of (mTICI) grade 2b/3 after mechanical thrombectomy |
| Time from hospital arrival to start of antihypertensive therapy in patients with ICH and hypertension |
| Proportion of patients with ICH achieving adequate control of hypertension in the first 24 h |
| Time from hospital arrival to administration of adequate anticoagulation reversal treatment in patients with anticoagulation-related ICH |
| Proportion of patients with CVST who are started on therapeutic anticoagulation within 24 h of the diagnosis |
| Proportion of stroke patients who are admitted to an acute stroke unit |
| Proportion of stroke patients who are screened or assessed for swallowing deficits |
| Proportion of patients with ischemic stroke who receive aspirin within the first 48 h |
| Proportion of stroke patients who receive DVT prophylaxis |
| Time from stroke onset until first mobilization |
| Proportion of eligible patients with large hemispheric infarction who are treated with hemicraniectomy |
| Proportion of stroke patients who receive education about stroke |
| Distribution of discharge locations for stroke patients |
| Secondary Prevention |
| Proportion of stroke or TIA patients with diagnostic evaluation of stroke etiology started within 48 h of stroke onset |
| Proportion of stroke or TIA patients with complete brain and extracranial/intracranial vascular imaging |
| Proportion of stroke or TIA patients with at least 24 h of cardiac monitoring |
| Proportion of stroke or TIA patients with their blood pressure assessed and treated |
| Proportion of stroke or TIA patients with their BMI estimated at the time of the event |
| Proportion of stroke or TIA patients with diabetes who achieve an HbA1c ⩽ 7% |
| Proportion of stroke or TIA patients with who achieve an LDL-cholesterol < 1.8 mmol/L or 70 mg/dL |
| Proportion of stroke or TIA patients who engage in low/moderate-intensity aerobic activity for 10 min 4 days/week |
| Proportion of stroke or TIA patients who are active smokers that receive smoking cessation counseling |
| Proportion of stroke or TIA patients who drink > 2 alcoholic drinks/d daily for men or > 1 alcoholic that receive counseling for alcohol intake reduction |
| Proportion of stroke or TIA patients with salt intake < 2000 mg daily |
| Proportion of stroke or TIA patients with atrial fibrillation who receive oral anticoagulants |
| Proportion of cryptogenic stroke or TIA patients with a PFO who are assessed by a Neurocardiology team or a cardiologist and a neurologist |
| Proportion of cryptogenic stroke patients with PFO, aged 18–60 years, who receive a PFO closure |
| Proportion of stroke or TIA patients with severe ipsilateral internal carotid artery stenosis who receive an endarterectomy within 14 days after symptoms onset |
| Proportion of minor stroke (NIHSS ⩽ 3 for clopidogrel or NIHSS ⩽ 5 for ticagrelor) or high-risk TIA (ABCD2 ⩾ 4) patients who receive dual antiplatelet therapy within 24 h of symptoms onset |
| Proportion of stroke or TIA patients with no indication for anticoagulation or dual antiplatelet therapy who receive a single antiplatelet agent for secondary stroke prevention |
| Rehabilitation |
| Proportion of patients assessed by a multidisciplinary team of stroke specialists |
| Proportion of patients/family receiving education about stroke |
| Proportion of patients/family receiving person centered, collaborative goal setting |
| Amount of task-specific therapy (minutes per day) |
| Proportion commencing mobilization between 24 and 48 h |
| Proportion of patients predicted to die who have access to specialist palliative care teams |
| Proportion of patients (especially those who are immobile) who have regular assessments of skin care and then measures taken to reduce the risk of skin breakdown |
| For patients unable to meet nutritional requirements, proportion receiving nutritional support (oral or with tube feeding), and interval until its institution |
| Proportion of patients having aerobic training at least three times weekly |
| Proportion of people with mild to moderate disability receiving early supported discharge |
| Proportion of people who receive advice on medication |
| Proportion of patients who receive emotional support |
| Proportion of patients who receive repetitive task training for their respective impairments (see individual impairments) |
| Proportion of patients with focal symptomatic distressing spasticity (upper and lower limbs) who receive chemo-denervation using botulinum toxin |
| Proportion of patients with foot drop getting an ankle-foot orthoses |
| Proportion of people with double vision referred for formal orthoptic assessment |
| Proportion of patients with aphasia evaluated by speech and language therapy |
| Proportion of all patients receiving appropriate handling to avoid shoulder subluxation including correct position of the hemiparetic arm and avoidance of overhead pulleys |
| Proportion of patients with acute stroke who are screened for depression using a structured depression assessment tool |
| For patients with a diagnosis of depression, proportion receiving psychotherapy and/or antidepressants |
| Proportion of patients who have their cognitive function assessed |
| Proportion of patients who have access to force platform biofeedback and mirror therapy |
| Proportion of patients receiving, and being discharged with, indwelling bladder catheters (lower proportion is better) |
| For people with constipation or bowel incontinence, the proportion with a bowel management program |
| Proportion of people with ongoing rehabilitation goals and those with difficulty performing activities of daily living who have access to specialized stroke services after leaving hospital |
| Proportion participating long-term in community-based exercise programs |
| Proportion of patients receiving a 6-month review |
| Proportion of hospitals within a region which maintains a data collection system to monitor performance metrics |
Stakeholder assessment
Discussion
Main findings of our searches
Main recommendations for acute stroke
Main recommendations for secondary prevention
Main recommendations for rehabilitation
Quality of guidelines
Strengths of our approach
Weaknesses of our approach
How to use this information
Implications for future guideline development and research
WSO guidelines committee membership
Acknowledgments
Declaration of conflicting interests
Funding
ORCID iDs
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
Cite
Cite
Cite
Download to reference manager
If you have citation software installed, you can download citation data to the citation manager of your choice
Information, rights and permissions
Information
Published In
Keywords
Authors
Metrics and citations
Metrics
Journals metrics
This article was published in International Journal of Stroke.
View All Journal MetricsPublication usage*
Total views and downloads: 51288
*Publication usage tracking started in December 2016
Altmetric
See the impact this article is making through the number of times it’s been read, and the Altmetric Score.
Learn more about the Altmetric Scores
Publications citing this one
Receive email alerts when this publication is cited
Web of Science: 163 view articles Opens in new tab
Crossref: 182
- Availability and content analysis of post-stroke rehabilitation guidelines in Latin America: A systematic review
- Intracranial Pressure Monitoring, Heart Rate Variability, Baroreflex Sensitivity, and Signal Complexity During Neurointensive Care after Decompressive Craniectomy in Malignant Middle Cerebral Artery Infarction
- Tenecteplase in ischemic stroke in the Russian Federation: first clinical experience beyond the therapeutic window
- Can immunization prevent stroke?
- Advancing Stroke Equity Globally: From Recognition to Strategic Action
- Antithrombotic Strategies for the Secondary Prevention of Stroke: A Review
- A non-randomised controlled study of the missing link person-centred care transition support intervention after stroke or TIA
- Post-stroke rehabilitation with a focus on motor and cognitive recovery
- Overlooked Burden of Poststroke Pain: Retrospective Analysis of a Real‐World Registry of Survivors of Chronic Stroke
- Vagus nerve stimulation to improve post-stroke motor function and activity
- View More
Figures and tables
Figures & Media
Tables
View Options
View options
PDF/EPUB
View PDF/EPUBAccess options
If you have access to journal content via a personal subscription, university, library, employer or society, select from the options below:
loading institutional access options
World Stroke Organization members can access journal content using society membership credentials.
Alternatively, view purchase options below:
Purchase 24 hour online access to view and download content.
Access journal content via a DeepDyve subscription or find out more about this option.




