Review article: do stimulant laxatives damage the gut? A critical analysis of current knowledge
Abstract
Plain language summary
Introduction
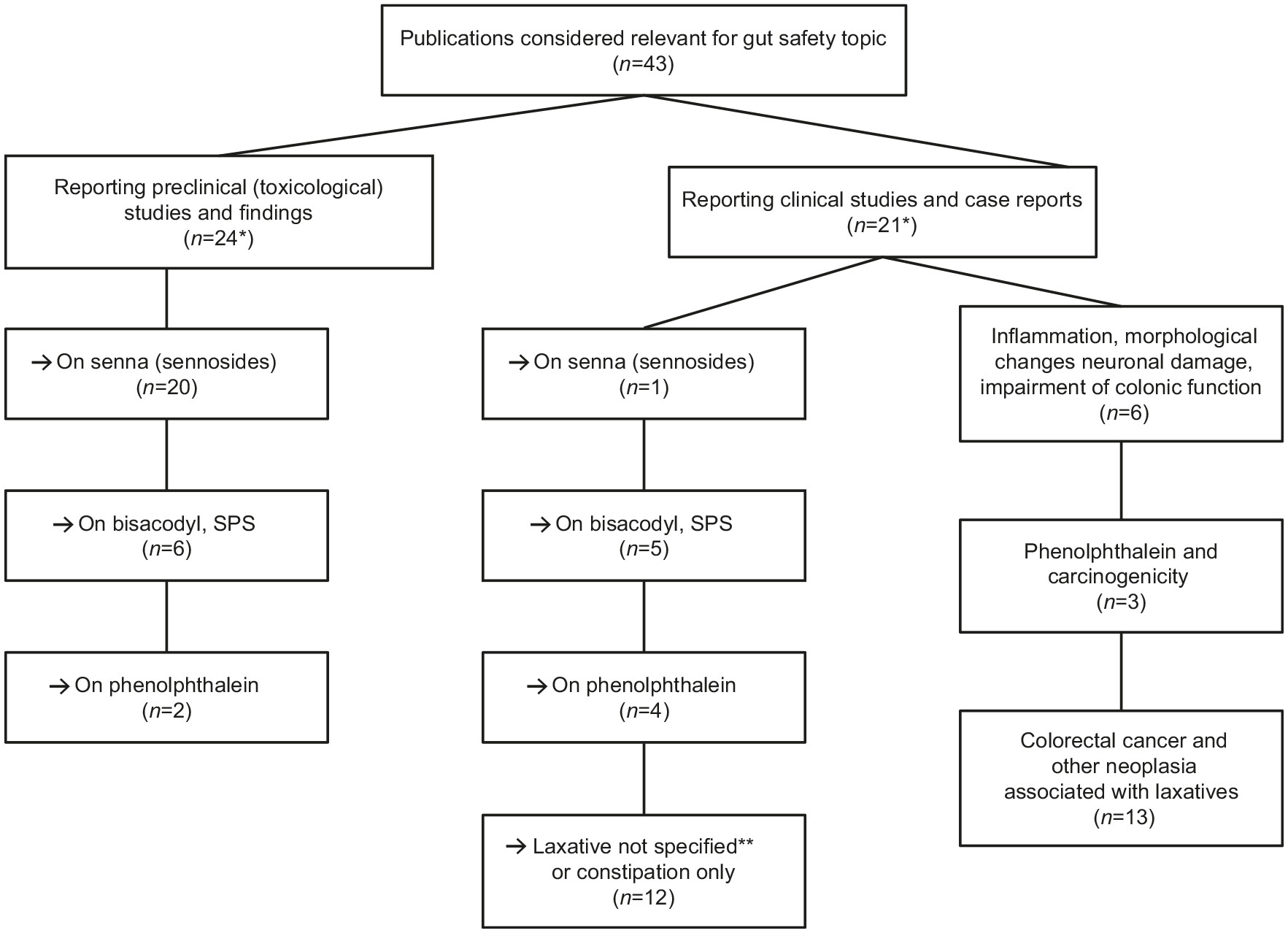
Methods
Results
| Citation | Study design | Duration | Key endpoints | Key findings | Conclusion |
|---|---|---|---|---|---|
| Genotoxicity/mutagenicity studies | |||||
| Heidemann et al., 199334 | Genotoxicity tests in vitro and in vivo on crude senna, senna extract, sennosides, rhein, and aloe-emodin | Not applicable | In vitro tests: mouse spot test; CA in vivo; Ames/Salmonella sp.; Ames/E. coli; HGPRT; CA/CHO; MLA In vivo tests: CA/rat; MNT/rat; MNT/mouse; mouse spot test Ex vivo tests: UDS/rat | Fructus sennae: negative (mouse spot; CA rat; MNT rat) Senna extract: positive (SAL; CHO); negative/equivocal (HGPRT) Sennosides: negative (SAL and E. coli/CHO/ML); negative (MNT mice) Rhein: negative (SAL; CHO); negative/equivocal (ML); negative in mouse MNT Aloe-emodin: positive (SAL; CHO), one positive and two negative tests (HGPRT); negative (mouse spot; CA/rat; MNT/mouse; rat UDS) | Senna extracts and aloe-emodin were genotoxic in some in vitro tests; no genotoxic effect in in vivo tests Crude senna, sennosides, and rhein* were not genotoxic in in vitro or in vivo conditions |
| Mengs, 198835 | Mutagenicity testing of sennosides | Not applicable | Ames test (Salmonella typhimurium; E. coli); MLA; CHO; MNT | No effects (max concentration or dose) in the Ames test (5000 µg/plate), MLA (5000 µg/ml nonactivation; 1000 µg/ml with activation); CHO (5000 µg/ml nonactivation; 4000 µg/ml with activation); MNT (2500 mg/kg) | The sennosides tested were not mutagenic in the test systems employed |
| Brusick and Mengs, 199736 | Review article of genotoxic risk of senna [senna extract; sennosides (A, B, C, D)]; rhein; crude senna; aloe-emodin | Not applicable | In vitro tests: Ames/Salmonella sp.; Ames/E. coli; MLA; HGPRT assay; CA/CHO; rat hepatocyte UDS; cell transformation (C3H/M2 cells) In vivo tests: MNT/rat; CA/rat bone marrow; somatic cell mutation/mouse; UDS/rat hepatocyte; MNT/mouse; CA/rat bone marrow metaphase | Sennosides: negative (five tests) Rhein: negative (four tests); one test was equivocal (MLA) Senna extract: positive (Ames; CA); negative (HGPRT) Crude senna: negative (three in vivo tests) Aloe-emodin: positive in all in vitro tests (except in one HGPRT experiment); negative (four in vivo tests) | Therapeutic doses of senna laxative produced in healthy human volunteers: aloe-emodin not detected in plasma (lower limit of quantification 0.5 mg/ml), using same assumptions for effects in rodents results in human safety margins for somatic genotoxicity of ~20,000 |
| NTP, 200137 | Genotoxicity studies on emodin | Not applicable | In vitro tests: Ames/Salmonella sp; CA/CHO In vivo tests: MNT/rat bone marrow erythrocytes; MNT/mouse bone marrow erythrocytes; MNT/mouse peripheral blood erythrocytes | Emodin was mutagenic in S. typhimurium strain TA100 with S9 activation (not in strain TA98 ± S9) CA was seen in CHO cells ± S9 Negative tests: rat bone marrow MNT; mouse bone marrow, and peripheral blood erythrocytes | Mutagenicity potential did not result in carcinogenesis (see NTP, 2001 below) |
| Stoll et al., 200631 | Mutagenicity testing of bisacodyl | Not applicable | Micronucleus test: SHE test | Micronucleus test: no effect on percent PCEs SHE: No change in transformation frequency at any dose level | Bisacodyl has no mutagenic potential |
| Inflammation, morphology, nerve damage | |||||
| Saunders et al., 197738 | Impact of bisacodyl on upper jejunum sections from humans, and jejunal, ileal, colonic areas in rats | Not applicable | Net water transport LM and EM of colon samples | Bisacodyl induced net water secretion (p < 0.01) in human intestinal segments In rats: concentration-related inhibition of water absorption in all regions by bisacodyl; LM – focal alterations in surface absorptive cells; EM: mucosal damage | Laxative effect of bisacodyl may be related to water absorption inhibition Morphological changes may be related to how bisacodyl decreases water transport |
| Carcinogenicity studies | |||||
| Lydén-Sokolowski et al., 199339 | Rat carcinogenicity study with a purified senna extract (sennosides) in drinking water | 2 years Daily doses consumed: 0, 5, 15, and 25 mg/kg | Tumors – specifically GI tract, liver, kidneys, and adrenals | No treatment-related effects in macroscopic findings No differences in microscopic findings (high-dose group versus controls) No treatment-related increase in neoplasms | There was no relationship between long-term administration of purified senna extract and tumor development of the GI tract, liver, kidneys, and adrenals of rats |
| Siegers et al, 199340 | To evaluate the tumorigenic potential of sennosides and aloin alone and their tumor-promoting potential in an established murine model of colorectal tumorigenesis (DMH) | 20 weeks DMH 20 mg/kg SC for 10 weeks. Sennosides 0.03% in diet (equivalent to 100 mg/kg/day – mild laxative effect) Aloin 0.03% in diet | Colorectal tumors Hepatic and nephrotoxicity | No. of tumor-bearing animals: senna only 0/20; senna + DMH 7/19; DMH alone 10/19; aloin alone 1/20; aloin + DMH 7/20 Incidence and growth of colorectal tumors were not affected by senna or aloin DMH-induced hepatotoxic and nephrotoxicity effects, which were enhanced by aloin but not senna | DMH-induced colorectal tumors were not impacted by aloin or sennoside-enriched diets Aloin- and sennoside-fed mice had no significant changes in hepato- or nephrotoxicity |
| NTP, 200137 | Rat and mouse carcinogenicity studies with emodin via the die | 2 years Rat study: 0, 280, 830, or 2500 ppm emodin Mouse study: 0, 160, 312, or 625 ppm emodin in males and 0, 312, 625, or 1250 ppm emodin in females | Tumors | Rat study: Zymbal gland carcinoma (three females, 2500 ppm), which exceeded current historical controls (equivocal finding); negative trends in mononuclear cell leukemia Mouse study: low incidences of renal tubule adenoma and carcinoma in male mice (one of each in the 312 and 625 ppm groups; rare tumor in male mice, possible association with emodin) | No evidence of carcinogenic activity of emodin in male rats and female mice; equivocal evidence of carcinogenic activity in female rats and male mice |
| Borrelli et al., 200541 | Carcinogenicity study of senna pod extract in rats | 2 years Positive control AOM Senna pod extract (by gavage): 30 or 60 mg/kg on 6 days/week | Tumors | Colon ACF and colon tumors: not present in controls or either dose of senna; present in all groups dosed with AOM; significantly decreased in AOM + senna pod extract versus AOM alone | Senna is devoid of carcinogenic potential in rats; senna may act as an anti-tumoral agent against colon carcinogenesis |
| Mitchell et al., 200642 | Rat carcinogenicity study with senna | 2 years Senna at 0, 25, 100, and 300 mg/kg/day (oral gavage) | Tumors | Increased tubular basophilia and epithelial hypertrophy in the kidneys No alterations in the colonic nervous plexus or increase of cell proliferation in the large intestine No treatment-related neoplastic changes were observed in any organ examined | Senna is not considered to be carcinogenic after daily administration for 2 years at doses up to 300 mg/kg/day in rats |
| Surh et al., 201343 | To assess carcinogenicity of senna in a haplo-insufficient mouse model | 40 weeks Senna fed in the diet at 0, 100, 300, 3000, 10,000 ppm | Tumors | Significant increases in epithelial hyperplasia (males, females): cecum 10,000 ppm 22/25 and 19/25; colon 3000 ppm 3/24 (not significant) and 7/25, 10,000 ppm 25/25 and 25/25; rectum 10,000 ppm 1/25 and 1/25 (not significant) | Large intestine is the major target of senna-induced toxicity in wild-type and p53+/− mouse models No neoplastic changes seen |
| Borrelli et al, 200144 | Subchronic carcinogenic study in rats – to assess ability of bisacodyl and cascara in inducing ACF and tumors in the colon. | 13 weeks Positive control AOM Bisacodyl: 4.3 or 43 mg/kg/day on 6 days/week ± AOM Cascara: 140 or 420 mg/kg/day on 6 days/week ± AOM | ACF and tumors in the colon | Both doses bisacodyl + AOM did not modify ACF appearance but increased number of crypts/focus versus AOM alone Cascara (both doses) + AOM – no increase in ACF or crypt/focus versus AOM alone AOM alone induced colon tumors as expected Neither bisacodyl nor cascara at either dose (absence of AOM) induced tumors Compared with AOM alone, low-dose bisacodyl and both cascara doses + AOM had no effect on tumors Compared with AOM alone, high-dose bisacodyl + AOM significantly increased number of tumors ~10-fold (of the nine rats in this group, 78% had tumors) | When bisacodyl, at either dose, was given without AOM initiating treatment, there was no evidence of tumor development Bisacodyl has a possible promoting effect on colon rat carcinogenesis, particularly at diarrheal dose Cascara had no promoting or initiating activity at a laxative and diarrheagenic dose |
| Citation | Study design and aims | Study population | Findings | Conclusions |
|---|---|---|---|---|
| Inflammation, morphology, nerve damage | ||||
| Smith, 196832 | Case report | A 57-year-old female with history of plant-based stimulant laxative abuse for 40 years | Terminal 5 cm ileum with colon from caecum to rectum examined Ileocaecal valve was stenosed and incompetent Colon had loss of interhaustral folds Submucosa contained adipose tissues Atrophy of muscle layers Plexus had abnormal neurological structures | Patient with cathartic syndrome had myenteric plexus damage |
| Joo et al., 199823 | To investigate colonic anatomy changes with chronic use of stimulant laxatives (diphenylmethanes, anthraquinones) | Part 1: mean 7.9 years (1–30) of laxative use Part 2: mean 5.2 years (2–19) of laxative use | Colon changes (haustral fold loss) | Part 1: chronic versus nonusers (% patients): colonic redundancy 35% versus 19%; colonic dilatation 45% versus 23%; loss of haustral marking 28% versus 0% p < 0.005 Part 2: 39% had loss of haustral folds Over both parts, 32% of chronic users had haustral fold loss; no difference in duration/type of laxative use between patients with haustral fold loss versus those without |
| Riemann et al., 198022 | To evaluate ultrastructure of submucosal nerve tissue in laxative abuse Anthraquinones (cascara sagrada, aloe, folia sennae, cortex frangulae) or bisacodyl. Average daily dose ~180 mg (70–260 mg) | Mean abuse = 13.5 years; 8–21 years | Ultrastructure of submucosal nerve tissue | Dose- and time-related severe damage of submucosal nerve fibers Main pathological features: axon ballooning, nerve-specific cell organelle reduction, lysosomal activity, and increased melanin-loaded macrophages Significant decrease in neurosecretory granules in nerve endings versus normal tissue |
| Saunders et al., 199045 | Impact of bisacodyl enemas on rectal mucosa in humans | Single biopsies collected at 3, 6, 24, 30, and 48 h post-enema | Histology (LM) | Injury of superficial epithelium and upper third of the crypt within 30 min after bisacodyl Mild inflammation seen histologically for up to 30 h after bisacodyl enema |
| Carcinogenicity | ||||
| Sonnenberg and Müller, 199326 | Meta-analysis of 14 case-control studies to evaluate if constipation or laxative use was associated with an increased CRC risk Various laxatives used for different dose frequencies | Laxative-users (n): patients with CRC (8–165) and non-CRC controls (6–2225) No-laxative-use (n): CRC patients (64–626) and non-CRC controls (94–9135) | In all but one study, CRC occurred more frequently in constipated patients than non-constipated patients; summary OR (95% CI): 1.48 (1.32–1.66) Use of laxatives also associated with CRC: OR (95% CI): 1.46 (1.33–1.61) The CRC risk was small but significant for both constipation and laxative use | Increased CRC risk may reflect confounding influence of diet rather than a primary influence of colonic inertia |
| Siegers et al, 199346 | A retrospective and a prospective epidemiological study To assess copresence of PMC and colorectal diseases | Patients undergoing endoscopy N: retrospective, 3049; prospective, 1095 31/33 patients with adenoma or CRC in prospective study abused anthranoid laxatives for 10–30 years | Retrospective study: no abnormality 3.13% of patients had PMC; adenoma 8.64% (p < 0.01); CRC 3.94% Prospective study: no abnormality 6.9% of patients had PMC; adenoma 9.8%; CRC 18.6% (p < 0.0008) Prospective study: adenoma and CRC were linear functions of age, sex, and PMC; adenomas age (factor 2.47), sex (factor 0.7), PMC (factor 4.57); CRC age (factor 6.14), neither sex nor PMC were significant factors Prospective study: RR: 3.04 (95% CI: 1.18–4.90) for CRC as a result of anthranoid-laxative abuse | Current data are contradictory as CC per se, together with dietary factors (e.g. low fiber and high fat intake), increase CRC risk in men |
| Kune, 199347 | Case-control arm of the Melbourne CRC study To evaluate the possible mutagenicity of anthraquinones | Histologically confirmed new patients with colorectal adenocarcinoma from April 1980 to April 1981 (685 cases) Community controls were age/sex-matched – randomly selected (723 controls) | RR of CRC for any anthraquinone use: 1.01 (not significant) RR of CRC for any phenolphthalein use: 1.35 (not significant) | Use of anthraquinone-stimulant laxatives or phenolphthalein-containing laxatives was not associated with any increased risk of CRC |
| Jacobs and White, 199848 | Case-control study To examine associations of colon cancer with constipation and use of commercial laxatives | Patients with colon cancer (all with invasive adenocarcinoma of the colon = 424). Control patients from same area in Seattle (random-digit dialing) (n = 414) Various durations of constipation and laxative use prior to interview | Constipation was associated with substantially increased risk of colon cancer: adjusted RR: 2.0 (95% CI: 1.2–3.6) for constipation 12–51×/year, and 4.4 (95% CI: 2.1–8.9) for constipation ⩾52×/year Frequent use of commercial laxatives increased colon cancer risk: RR: 2.5 (95% CI: 1.3–4.7) for ⩾350 lifetime uses Adjusting for constipation and commercial-laxative use, cancer risk associated with laxative use disappeared, and strong association with constipation seen | Results suggest that frequent constipation may be an important risk factor for colon cancer in middle-aged adults |
| Dukas et al., 200049 | A clinical prospective (case-control) study (Nurses’ health study living in the USA) To assess association between bowel-movement frequency, laxative use, and the risk of CRC | Registered nurses (all female; n = 84,577), 30–55 years at enrollment; follow-up for 12 years | 611 cases of CRC RRs (95% CIs): bowel movements every third day versus daily movements 0.94 (0.69–1.28) for CRC, 0.88 (0.62–1.26) for colon cancer, 1.18 (0.63–2.20) for rectal cancer Compared with women never using laxatives, multivariate RRs (95% CI) associated with weekly to daily laxative use: 1.00 (0.72–1.40) for CRC, 1.09 (0.76–1.57) for colon cancer, 0.68 (0.29–1.57) for rectal cancer | Findings did not support an association between infrequent bowel movement, laxative use, and CRC |
| Nascimbeni et al., 200250 | Prospective case-control study To assess the risk of colon cancer by constipation, anthranoid-laxative use, and PMC | Patients undergoing resection for SC (n = 55) or DD (n = 41) and matched controls (n = 96) | OR (95% CI) versus controls: 1.9 (0.9–4.1, SC) and 2.8 (1.2–6.3, DD) for constipation; 5.3 (2.1–13, SC) and 4.0 (1.5–11, DD) for anthranoid-laxative use All patients: OR: 3.4 (1.3–8.5) –significant association between PMC and anthranoid-laxative use No association between ACF characteristics and history of constipation, or history of anthranoid, laxative use, or PMC | Study demonstrates association between ACF frequency and colon cancer; it does not support a cause–effect relationship between CRC with constipation, use of anthranoid laxatives, or PMC |
| Guerin et al., 201451 | Retrospective, matched cohort design (CC matched 1:3 with non-CC controls) To evaluate the association of CC and CC severity with CRC | Participants in US claims database with CC (n = 28,854) and matched controls (n = 86,562) | 1-year CRC prevalence 2.7% (CC patients) and 1.7% (non-CC patients) p < 0.001 Risk of developing CRC (IRR, 95% CI) versus CC free: mild CC: 0.83 (0.62–1.10), severe CC: 1.40 (1.18–1.66), p < 0.05; very severe CC: 2.26 (1.94–2.64), p < 0.05Most indicators of constipation severity were associated with a significant incremental risk of CRC (except for patients with laxative prescriptions) | CC is associated with significantly higher prevalence and incidence of CRC and benign colorectal neoplasm versus matched CC-free patients. Risks increase with the severity of CC |
| Roberts, 200352 | Population-based, case-control study To determine if bowel-movement frequency, laxative use, and type were associated with colon cancer risk in white and black men and women | Patients with pathologically confirmed invasive adenocarcinoma of the colon 643 cases (349 white, 294 black) and 1048 controls (611 white, 437 black) | Constipation was associated with risk of colon cancer (OR, CI): 2.36; 1.41–3.93 adjusted for age, race, sex, and relevant confounders The association between constipation and cancer was greater for women: 2.69; 1.46–4.94) than for men: 1.73; 0.61–4.88; and stronger in blacks: 2.90; 1.54–5.46 than whites: 2.02; 1.04–3.91 No association with laxative use and colon cancer: OR: 0.88 (0.69–1.11) Fiber commercial laxatives appeared to exert a protective effect: OR: 0.58 (0.32–1.05) | Evidence supports a positive association between constipation and increased risk of colon cancer. No apparent association of any laxative use with colon cancer risk. Commercial-fiber laxatives may lower the potential risk of colon cancer |
| Watanabe et al., 200453 | Population-based prospective case-control study in Japan To investigate the association between laxative use and CRC risk | Healthy subjects (n = 41,670) 7 years follow-up | RR (95% CI) of CRC for constipated subjects versus those daily bowel movements: 1.35 (0.99–1.84), p = 0.006 RR for CRC for laxative-users versus nonusers: 1.31 (0.88–1.95), p = 0.72; and for frequent users (⩾2×/week) 2.75 (1.48–5.09), p = 0.02 RR (95% CI) of colon cancer (constipated versus non-constipated): 1.47 (1.00–2.17), p < 0.05 RR for colon cancer for laxative-users versus nonusers: 1.48 (0.91–2.40), p = 0.11 RR (95% CI) of rectal cancer (constipated versus non-constipated): 1.16 (0.69–1.95), p = 0.59 RR for rectal cancer for laxative-users versus nonusers: 1.04 (0.52–2.10), p = 0.91 | Supports hypothesis that constipation or laxative use increases the risk of colon cancer |
| Citronberg et al, 201427 | Prospective, questionnaire-based study To assess CRC risk with non-fiber and fiber laxatives | Participants in VITAL study (n = 75,214) followed for 10 years | Non-fiber laxatives: HRs associated with low and high use were 1.49 (95% CI: 1.04–2.14) and 1.43 (95% CI: 0.82–2.28), respectively, versus those who used them <once/year; ptrend = 0.05 Use of non-fiber laxatives was higher than fiber laxatives: OR: 6.04 (95% CI: 5.52–6.60) versus 1.24 (95% CI: 1.10–1.41) in patients with low bowel-movement frequency HRs for CRC were significantly decreased and lowest in individuals who reported using fiber laxatives often (4+ days/week for 4+ years) versus those who reported no use (HR: 0.44, 95% CI: 0.21–0.95); ptrend = 0.19 No statistically significant associations between bowel-movement frequency or constipation and CRC risk | Risk of CRC increases with non-fiber laxative use and decreases with fiber laxative use. Further studies needed to clarify relationships |
| Citronberg et al., 201828 | Retrospective study To examine the association between non-fiber-based laxative use, fiber-based laxative use, and CRC risk | Participants in CCFC registry (n = 4930 with primary invasive CRC) and controls from the general population (n = 4025) | Non-fiber-based laxatives significantly increased CRC risk versus no use; OR: 2.17, 95% CI: 1.47–3.19 No risk of CRC with fiber-based laxative use: OR: 0.99, 95% CI: 0.80–1.22 | Risk of CRC increased with non-fiber-based laxative use, while CRC risk was not significantly associated with fiber-based laxative use, compared with nonusers |
Preclinical toxicology studies
Genotoxicity studies
Inflammation, morphological changes, and neuronal damage, including impairment of colonic function
Carcinogenicity studies
Clinical studies and case reports
Inflammation, morphological changes, and neuronal damage, including impairment of colonic function (cathartic colon)
Carcinogenicity associated with phenolphthalein
Carcinogenicity associated with sennosides, bisacodyl, and SPS or with constipation
Discussion
Structural changes, nerve damage, and functional impairment of the intestine
Colorectal cancer
Association of phenolphthalein with CRC
Association of anthranoid laxatives with CRC
Conclusion
Acknowledgments
ORCID iDs
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
Cite
Cite
Cite
Download to reference manager
If you have citation software installed, you can download citation data to the citation manager of your choice
Information, rights and permissions
Information
Published In
Keywords
Authors
Metrics and citations
Metrics
Journals metrics
This article was published in Therapeutic Advances in Gastroenterology.
View All Journal MetricsPublication usage*
Total views and downloads: 8794
*Publication usage tracking started in December 2016
Altmetric
See the impact this article is making through the number of times it’s been read, and the Altmetric Score.
Learn more about the Altmetric Scores
Publications citing this one
Receive email alerts when this publication is cited
Web of Science: 9 view articles Opens in new tab
Crossref: 11
- Pharmacological management of childhood constipation: Bridging today’s gaps with tomorrow’s therapies
- Impact of antipsychotics, antiparkinsonian drugs, and anthraquinone stimulant laxatives on severe constipation in patients with schizophrenia
- Laxative logic: when lifestyle is not enough
- Assessment of administration pattern of laxatives in pharmacies in Nasiriya City, Iraq
- Pediatric Constipation
- Chronic constipation: From syndrome to diagnosis and effective therapy
- Isolation and identification of a novel bisacodyl analogue, bisacodyl dicycloproyl carboxylate, as a potential illegal additive in a dried plum product advertised for weight loss
- A clinical decision support alert to promote timely laxative use in in-patients prescribed clozapine
- Pharmacological treatment for children with constipation: present and future
- Post-marketing safety assessment of constipation drugs: a real-world pharmacovigilance study based on FAERS database
- View More
Figures and tables
Figures & Media
Tables
View Options
View options
PDF/EPUB
View PDF/EPUBAccess options
If you have access to journal content via a personal subscription, university, library, employer or society, select from the options below:
loading institutional access options
Alternatively, view purchase options below:
Purchase 24 hour online access to view and download content.
Access journal content via a DeepDyve subscription or find out more about this option.