Comparative efficacy of diverse therapeutic regimens for small intestinal bacterial overgrowth: a systematic network meta-analysis
Abstract
Background:
Objective:
Design:
Data sources and methods:
Results:
Conclusion:
Trial registration:
Introduction
Methods
Identification of studies and data extraction
Study selection
Data extraction
Subgroup analysis
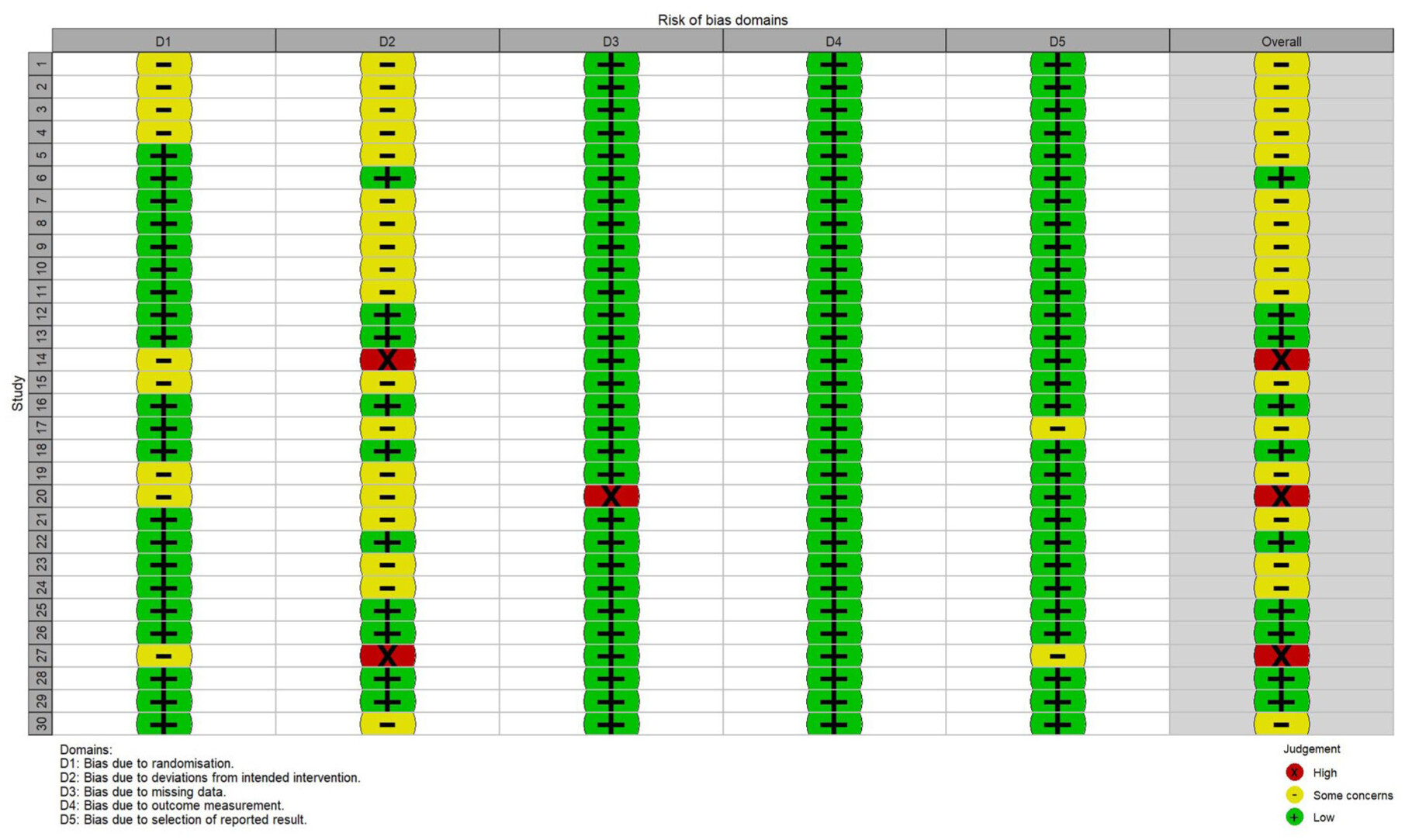
RoB of individual studies
Statistical analysis
Results
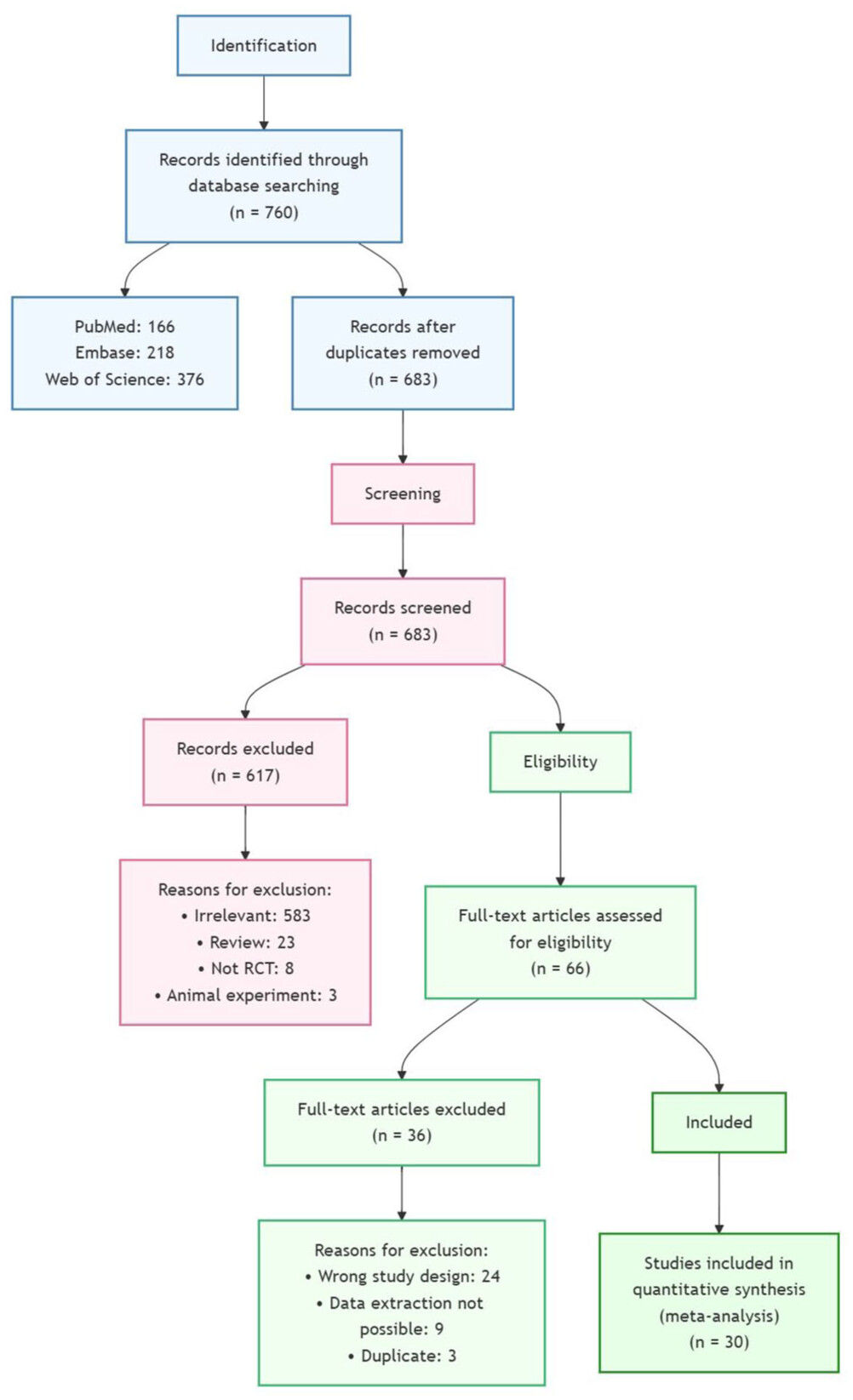
Literature search and quality assessment
| Study | Country | Sample size (n) | Mean age (years) | Male (%) | Population | SIBO diagnosis | Treatment regimens | The duration of follow-up | Adverse reactions |
|---|---|---|---|---|---|---|---|---|---|
| Biancone et al.28 | Italy | 14 | 45 (21–60) | 50.00% | SIBO patients with inactive CD of the ileum | GHBT | Rifaximin 1200 mg/day vs placebo for 1 week | 2 weeks | None |
| Di Stefano et al.29 | Italy | 21 | 56 (23–84) | 47.62% | SIBO patients | GHBT | Rifaximin 1200 mg/day vs chlortetracycline 1000 mg/day for 1 week | 3 days | None |
| Pardo et al.30 | Spain | 9 | 58.6 ± 8.5 | 77.78% | SIBO patients with liver cirrhosis | Quantitative bacterial culture of jejunal fluid | Cisapride 40 mg/day for 1 week vs no treatment | 1 week | None |
| Madrid et al.31 | Chile | 17 | 58(44–69) | 41.18% | SIBO patients with liver cirrhosis | LHBT | Norfloxacin 800 mg + neomycin 1500 mg/day vs cisapride 30 mg/day vs placebo for 15 days | 3 weeks | None |
| Castiglione et al.32 | Italy | 28 | 39(21–70) | 58.62% | SIBO patients with Crohn’s disease | GHBT + LHBT | Metronidazole 750 mg/day vs ciprofloxacin 1000 mg/day for 10 days | 1 week | None |
| Pimentel et al.33 | United States | 84 | 43.3 ± 1.4 | 54.76% | SIBO patients with irritable bowel syndrome | LHBT | Neomycin 1000 mg/day vs placebo for 10 days | 1 week | None |
| Lauritano et al.34 | Italy | 90 | 32 ± 12 | 26.67% | SIBO patients | GHBT | Rifaximin 600 mg/day vs 800 mg/day vs 1200 mg/day for 1 week | 1 month | Weakness (n = 5), headache (n = 2), constipation (n = 2), dizziness (n = 1), insomnia (n = 1) |
| Scarpellini et al.35 | Italy | 77 | 34 ± 19 | 37.66% | SIBO patients | GHBT | Rifaximin 1600 mg/day vs 1200 mg/day for 1 week | 1 month | Constipation (n = 5), indigestion (n = 3) |
| Parodi et al.36 | Italy | 52 | / | / | SIBO patients with Rosacea | GHBT + LHBT | Rifaximin 1200 mg/day vs placebo for 10 days | 1 month | None |
| Lauritano et al.37 | Italy | 137 | / | / | SIBO patients | GHBT | Rifaximin 1200 mg/day vs metronidazole 750 mg/day for 1 week | 1 month | Exist, unknown |
| Furnari et al.38 | Italy | 76 | 54 (26–84) | 19.48% | SIBO patients | GHBT | Rifaximin 1200 mg/day vs Rifaximin 1200 mg/day + partially hydrolyzed guar gum 5 g/day for 10 days | 1 month | None |
| Chang et al.39 | Colombia | 24 | 42.7 (20–75) | 33.33% | SIBO patients with celiac disease | LHBT | Rifaximin 1200 mg/day vs placebo for 10 days | 2 weeks | None |
| Collins and Lin40 | United States | 63 | 12.6 ± 3 | 14.29% | SIBO patients with chronic abdominal pain | LHBT | Rifaximin 1650 mg/day vs placebo for 10 days | 2 weeks | None |
| Jun et al.41 | South Korea | 14 | / | / | SIBO patients with chronic liver disease | LHBT | Probiotics vs placebo for 4 weeks | 1 month | / |
| Khalighi et al.42 | Iran | 30 | 38.73 ± 14.35 | 50.00% | SIBO patients with chronic abdominal pain or diarrhea | HBT | Following 3 weeks of aggressive therapy with broad-spectrum antibiotics, a monthly maintenance therapeutic regimen (15 days of minocycline 200 mg/day + lactol probiotic) vs a monthly maintenance therapeutic regimen (15 days of antibiotic therapy with minocycline 200 mg/day) for 6 months | 6 months | None |
| Kwak et al.43 | South Korea | 13 | 53.85 ± 9.1 | 69.23% | SIBO patients with chronic liver disease | LHBT | Probiotics vs placebo for 4 weeks | 1 month | / |
| Lunia et al.44 | India | 59 | 48.9 ± 10.9 | 59.32% | SIBO patients with liver cirrhosis | GHBT + LHBT | Probiotics for 3 months vs no treatment | 3 months | None |
| Ghoshal et al.45 | India | 8 | 41 (18–54) | 87.50% | SIBO patients with irritable bowel syndrome | GHBT | Norfloxacin 800 mg/day vs placebo for 10 days | 1 month | None |
| Liang et al.46 | China | 126 | 50.7 ± 11.2 | 57.94% | SIBO patients with gastric or colorectal cancer | GHBT | Bifidobacterium triple viable capsule 1500 mg/day vs placebo for 4 weeks | 1 month | None |
| Vera et al.47 | Chile | 81 | / | 11.11% | SIBO patients with FDIGs | LHBT | Rifaximin 800 mg/day vs ciprofloxacin 1000 mg/day vs metronidazole 1500 mg/day for 10 days | 2 weeks | Overall side effects: metronidazole 50%, ciprofloxacin 39%, rifaximin 11% |
| Furnari et al.48 | Italy | 20 | 19.6(9.2–36.9) | 45.00% | SIBO patients with cystic fibrosis | GHBT | Rifaximin 1200 mg/day for 2 weeks vs no treatment | 1 month | Nausea (n = 1) |
| Tuteja et al.49 | United States | 26 | 52 (33–77) | 84.62% | SIBO patients | LHBT | Rifaximin 1100 mg/day vs placebo for 2 weeks | 2 weeks | None |
| García-Collinot et al.50 | Mexico | 40 | 53.2 ± 9.3 | 10.00% | SIBO patients with systemic sclerosis | LHBT | Metronidazole 1000 mg/day vs Saccharomyces boulardii 400 mg/day vs metronidazole 1000 mg + S. boulardii 400 mg/day for 1 week | 1 month | Metronidazole (heartburn n = 6, constipation n = 3), S. boulardii (flatulence n = 2, diarrhea n = 4) |
| Kim et al.51 | South Korea | 19 | 49.4 ± 12 | 57.89% | SIBO patients with functional dyspepsia | LHBT | Ursodeoxycholic acid 300 mg/day for 2 months vs no treatment | 2 months | None |
| Shi et al.52 | China | 28 | 41.9 ± 11.6 | 30.00% | SIBO patients with functional bowel disease | LHBT | Combined live Bacillus subtilis and Enterococcus faecium enteric-coated capsules 1500 mg/day for 4 weeks vs no treatment | 1 month | None |
| Kim et al.53 | South Korea | 83 | 50.9 ± 14.7 | 31.33% | SIBO patients with functional dyspepsia | GHBT | Mosapride 15 mg/day vs rifaximin 1200 mg/day vs mosapride 15 mg + rifaximin 1200 mg/day for 2 weeks | 1 month | None |
| Duan et al.54 | China | 46 | / | / | SIBO patients | HMBT | Berberine 800 mg/day vs rifaximin 800 mg/day for 2 weeks | 1 month | |
| Jo et al.55 | South Korea | 80 | 53.9 ± 14 | 53.75% | SIBO patients with functional bloating and no constipation | GHBT | Rifaximin 1200 mg/day + trimebutine maleate 600 mg/day vs Rifaximin 1200 mg/day + placebo for 2 weeks | 2 weeks | Rifaximin + trimebutine maleate (acid reflux n = 1, abdominal discomfort n = 1), Rifaximin (diarrhea n = 1, constipation n = 1, bloating n = 1, acid reflux n = 1, abdominal discomfort n = 2) |
| Ohkubo et al.56 | Japan | 8 | / | / | SIBO patients with chronic intestinal pseudo-obstruction | GHBT | Rifaximin 1200 mg/day vs placebo for 4 weeks | 1 month | / |
| Redondo-Cuevas et al.57 | Spain | 179 | 45.7 ± 16.2 | 17.32% | SIBO patients | LHBT | Rifaximin + herbal preparations + berberine + neomycin + probiotic + prebiotics vs rifaximin | 3 months | / |
Pairwise meta-analysis
NMA results and meta-regression
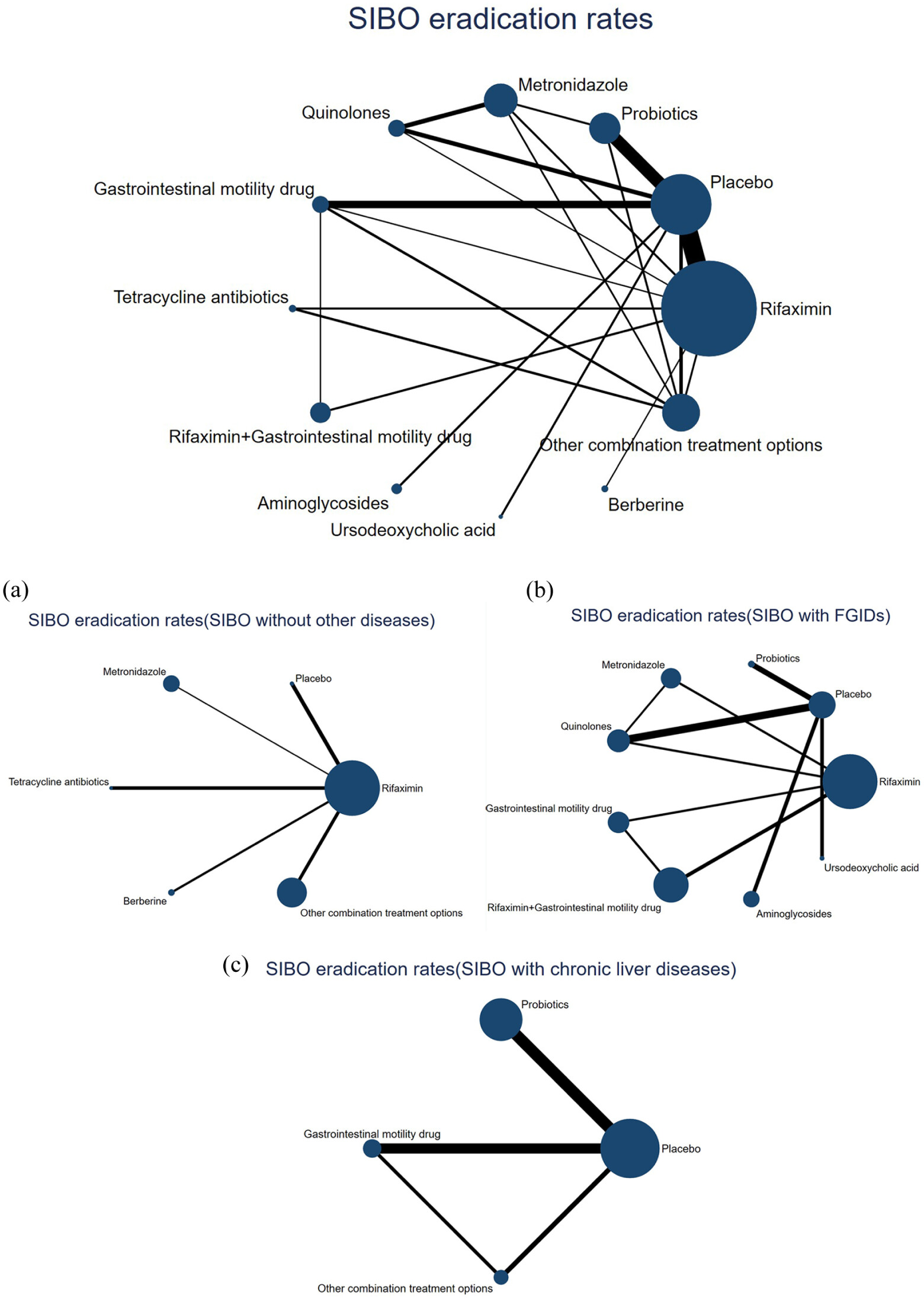
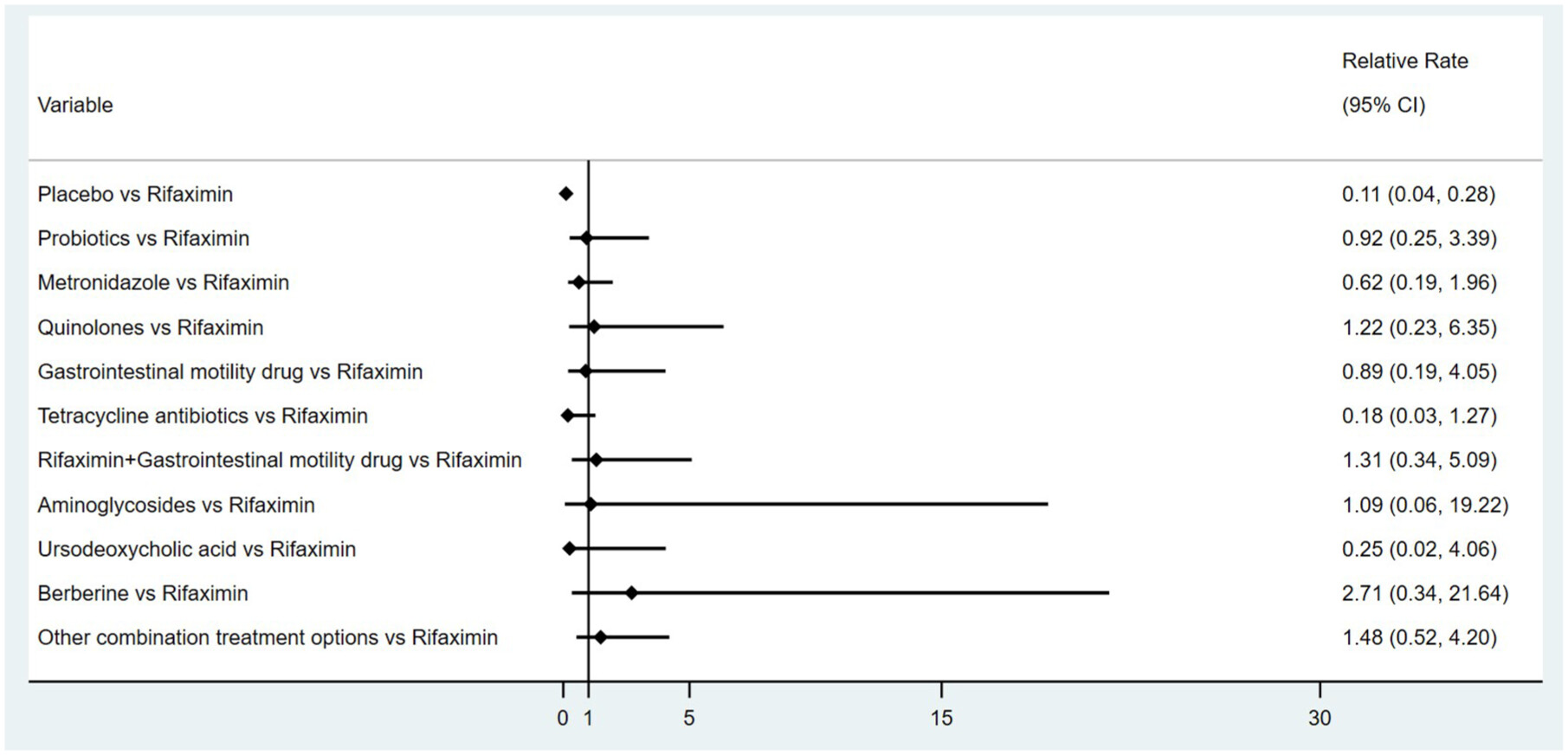
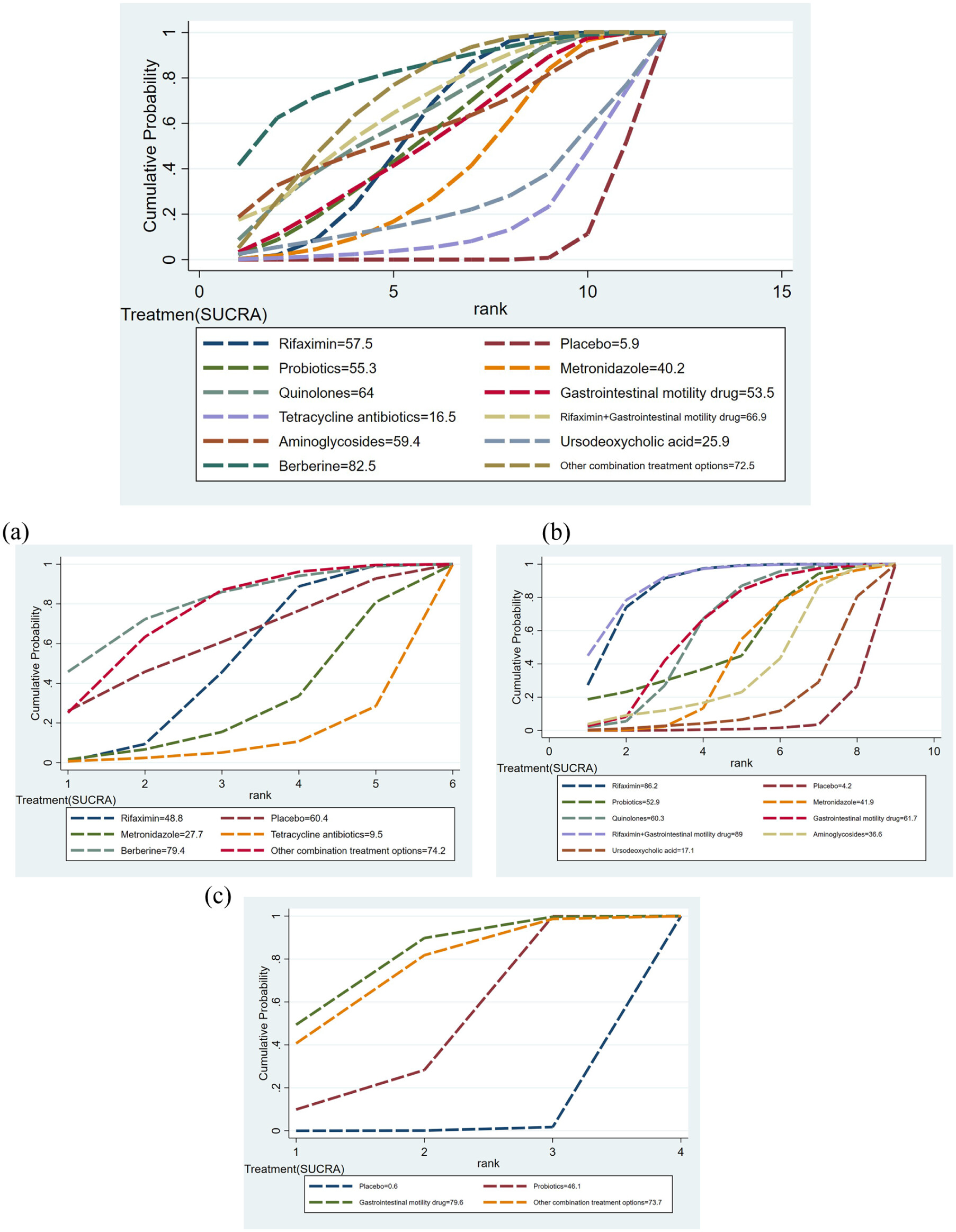

Results of subgroup analyses
SIBO without other diseases
SIBO with FGIDs
SIBO with chronic liver diseases
Discussion
Acknowledgments
ORCID iD
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
Cite
Cite
Cite
Download to reference manager
If you have citation software installed, you can download citation data to the citation manager of your choice
Information, rights and permissions
Information
Published In
Keywords
Authors
Metrics and citations
Metrics
Journals metrics
This article was published in Therapeutic Advances in Gastroenterology.
View All Journal MetricsPublication usage*
Total views and downloads: 2530
*Publication usage tracking started in December 2016
Altmetric
See the impact this article is making through the number of times it’s been read, and the Altmetric Score.
Learn more about the Altmetric Scores
Publications citing this one
Receive email alerts when this publication is cited
Web of Science: 0
Crossref:
There are no citing articles to show.
Figures and tables
Figures & Media
Tables
View Options
View options
PDF/EPUB
View PDF/EPUBAccess options
If you have access to journal content via a personal subscription, university, library, employer or society, select from the options below:
loading institutional access options
Alternatively, view purchase options below:
Purchase 24 hour online access to view and download content.
Access journal content via a DeepDyve subscription or find out more about this option.