Drug-induced interstitial lung disease: a real-world pharmacovigilance study of the FDA Adverse Event Reporting System from 2004 to 2021
Abstract
Background:
Objectives:
Design:
Methods:
Results:
Conclusion:
Plain language summary
Introduction
Design and methods
Data source
Definition of DILD cases
Data processing and analysis
Data standardization and filtering

Stratified analysis
Statistical analysis
| Measures | Calculation formula | Criteria |
|---|---|---|
| ROR | ROR = (a/b)/(c/d) | a ⩾ 3, 95% CI > 1 |
| PRR | PRR = (a/(a + c))/(b/(b + d)) | a ⩾ 3, PRR ⩾ 2, χ2 ⩾ 4 |
| BCPNN | IC = log2a(a + b + c + d)/((a + c) (a + b)) | IC025 > 0 |
Results
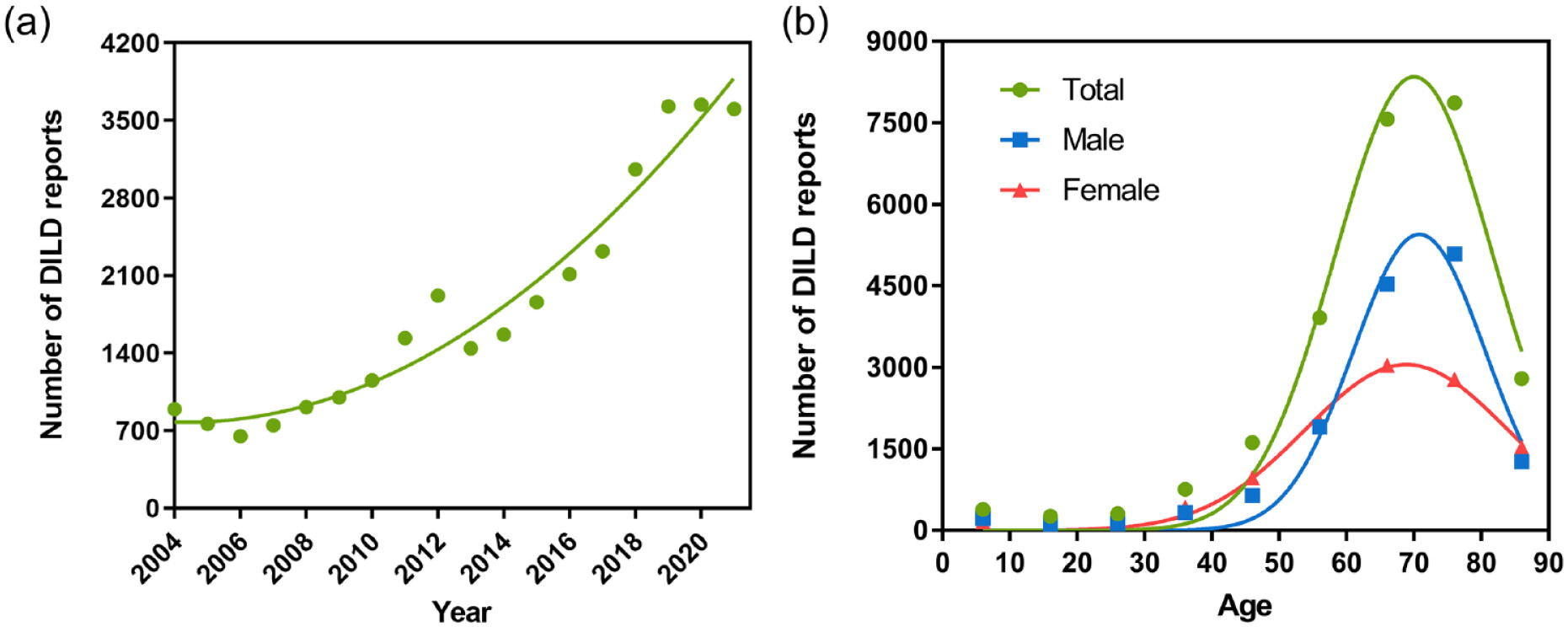
Temporal trends and demographic characteristics
| Group | Number of reports, n (%) |
|---|---|
| Age group (year) | |
| 0–6 | 294 (0.90%) |
| 7–12 | 173 (0.53%) |
| 13–18 | 151 (0.46%) |
| 19–40 | 1207 (3.68%) |
| 41–60 | 5792 (17.65%) |
| 61–80 | 15,865 (48.34%) |
| ⩾81 | 2898 (8.83%) |
| Unknown | 6441 (19.62%) |
| Age mean ± SD | 65.24 ± 15.72 |
| Sex group | |
| Male | 16,037 (48.86%) |
| Female | 13,072 (39.83%) |
| Unknown | 3712 (11.31%) |
| Reporters | |
| Consumer | 4476 (13.64%) |
| Physician | 15,743 (47.97%) |
| Other health professional | 6451 (19.66%) |
| Pharmacist | 2312 (7.04%) |
| Others | 2264 (6.90%) |
| Unknown | 1575 (4.80%) |
| Reporter country | |
| Japan | 14,483 (44.13%) |
| United States | 4424 (13.48%) |
| France | 3799 (11.57%) |
| Others | 8780 (26.75%) |
| Unknown | 1335 (4.07%) |
| Group | Total | Outcome, n (%) | |||||
|---|---|---|---|---|---|---|---|
| DE | DS | LT | HO | OT | Unknown | ||
| Age group (year)a | |||||||
| 0–6 | 294 | 110 (37.41%) | 6 (2.04%) | 46 (15.65%) | 66 (22.45%) | 60 (20.41%) | 6 (2.04%) |
| 7–12 | 173 | 61 (35.26%) | 1 (0.58%) | 19 (10.98%) | 44 (25.43%) | 47 (27.17%) | 1 (0.58%) |
| 13–18 | 151 | 42 (27.81%) | 2 (1.32%) | 23 (15.23%) | 45 (29.80%) | 39 (25.83%) | 0 (0.00%) |
| 19–40 | 1207 | 242 (20.05%) | 29 (2.40%) | 124 (10.27%) | 482 (39.93%) | 318 (26.35%) | 12 (0.99%) |
| 41–60 | 5792 | 1285 (22.19%) | 94 (1.62%) | 462 (7.98%) | 2136 (36.88%) | 1751 (30.23%) | 64 (1.10%) |
| 61–80 | 15,865 | 4991 (31.46%) | 351 (2.21%) | 1253 (7.90%) | 5449 (34.35%) | 3657 (23.05%) | 164 (1.03%) |
| ⩾81 | 2898 | 993 (34.27%) | 44 (1.52%) | 205 (7.07%) | 972 (33.54%) | 654 (22.57%) | 30 (1.04%) |
| Unknown | 6441 | 1524 (23.66%) | 136 (2.11%) | 302 (4.69%) | 1359 (21.10%) | 3000 (46.58%) | 120 (1.86%) |
| Sex groupa | |||||||
| Male | 16,037 | 5519 (34.41%) | 339 (2.11%) | 1352 (8.43%) | 5232 (32.62%) | 3428 (21.38%) | 167 (1.04%) |
| Female | 13,072 | 2973 (22.74%) | 287 (2.20%) | 947 (7.24%) | 4725 (36.15%) | 3946 (30.19%) | 194 (1.48%) |
| Unknown | 3712 | 756 (20.37%) | 37 (1.00%) | 135 (3.64%) | 596 (16.06%) | 2152 (57.97%) | 36 (0.97%) |
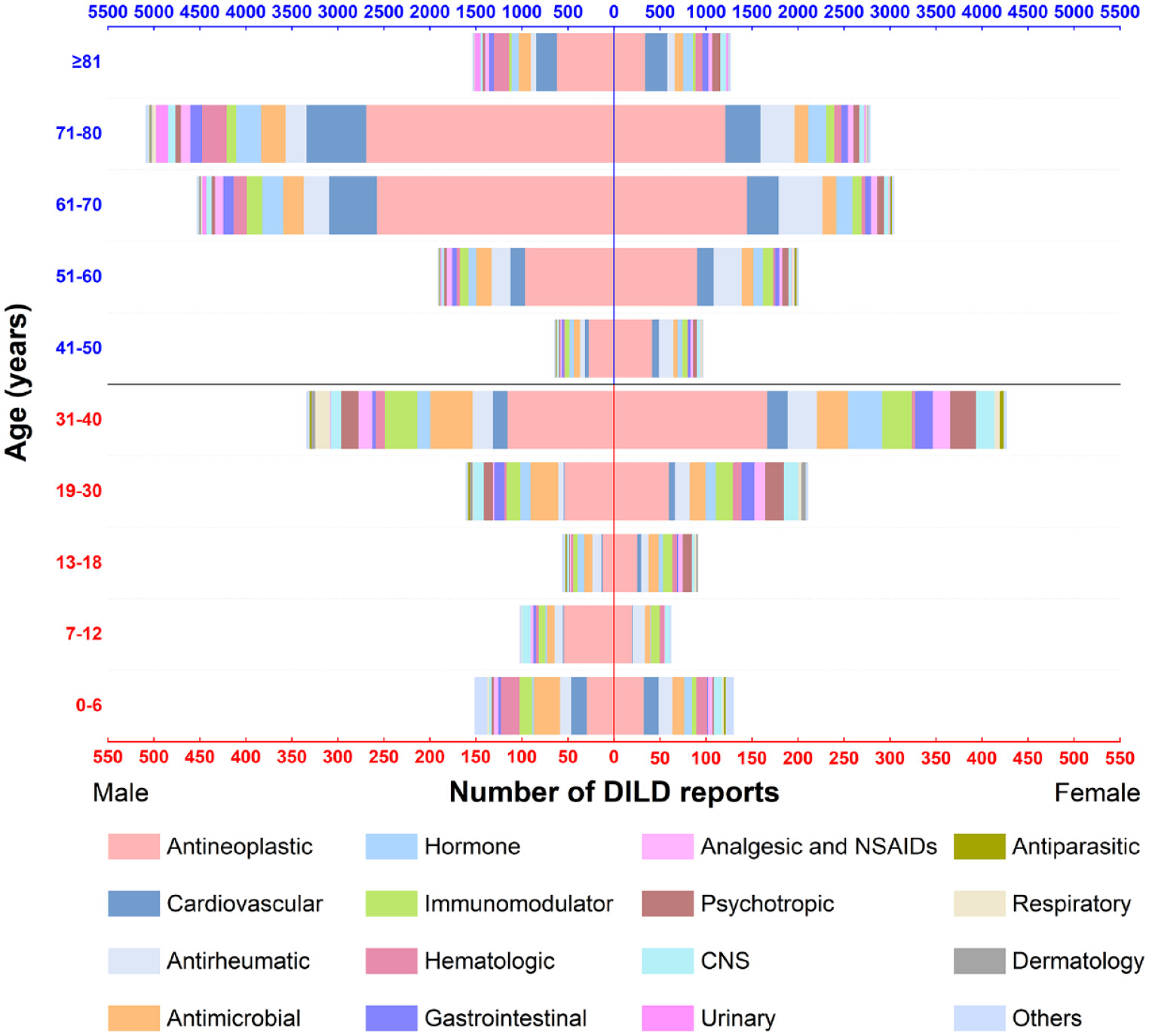
Classification of drugs associated with DILD

| Drug class | DILD reports (%) | Japan DILD reports (%, n = 31,486)a | Maximum DILD reports in 1 year (year) | Male (%, n = 29,279)a | Median age [IQR] (n = 26,380a) | Annual average % change (95% CI), p valueb |
|---|---|---|---|---|---|---|
| Overall database from 2004 to 2021 | 32,821 (100.00%) | 14,483 (46.00%) | 3645 (2020) | 16,037 (54.77%) | 68 [59] | / |
| Antineoplastic | 15,652 (47.69%) | 8034 (53.24%) | 1961 (2019) | 8061 (59.63%) | 68 [60] | 0.64 (0.49–0.79), p < 0.001 |
| Cardiovascular | 3668 (11.18%) | 904 (25.91%) | 367 (2019) | 1871 (55.77%) | 71 [63] | 0.41 (0.30–0.52), p < 0.001 |
| Antirheumatic | 3276 (9.98%) | 1177 (37.28%) | 385 (2020) | 1048 (34.62%) | 65 [56] | 0.59 (0.48–0.70), p < 0.001 |
| Antimicrobial | 1922 (5.86%) | 888 (50.03%) | 141 (2020) | 1045 (58.97%) | 66 [55] | 0.16 (0.08–0.23), p < 0.001 |
| Hormone | 1857 (5.66%) | 812 (45.49%) | 175 (2019) | 864 (51.22%) | 69 [60] | 0.50 (0.38–0.62), p < 0.001 |
| Immunomodulator | 1257 (3.83%) | 524 (42.43%) | 174 (2020) | 584 (52.19%) | 61 [50] | 0.67 (0.50–0.83), p < 0.001 |
| Hematologic | 1151 (3.51%) | 731 (64.41%) | 140 (2015) | 700 (70.21%) | 75 [67] | 0.61 (0.38–0.84), p < 0.001 |
| Gastrointestinal | 830 (2.53%) | 337 (43.82%) | 91 (2019) | 414 (54.83%) | 69 [59] | 0.36 (0.19–0.54), p < 0.001 |
| Analgesic and NSAIDs | 780 (2.38%) | 382 (51.76%%) | 80 (2018) | 367 (52.13%) | 68 [57] | 0.45 (0.32–0.58), p < 0.001 |
| Psychotropic | 717 (2.18%) | 99 (14.33%) | 126 (2018) | 225 (34.88%) | 67 [49.5] | 0.62 (0.34–0.90), p < 0.001 |
| CNS | 660 (2.01%) | 275 (44.57%) | 89 (2020) | 265 (44.09%) | 66 [48] | 0.37 (0.18–0.55), p < 0.001 |
| Urinary | 343 (1.05%) | 104 (31.80%) | 109 (2020) | 264 (83.81%) | 78 [72] | 0.84 (0.23–1.46), p = 0.009 |
| Respiratory | 254 (0.77%) | 62 (25.83%) | 50 (2021) | 123 (52.56%) | 65 [46.25] | 0.71 (0.43–0.98), p < 0.001 |
| Others | 228 (0.69%) | 82 (37.44%) | 25 (2021) | 112 (52.83%) | 66 [48] | 0.22 (0.00–0.45), p = 0.049 |
| Dermatology | 114 (0.35%) | 60 (55.05%) | 41 (2021) | 70 (67.96%) | 63 [51.5] | 0.99 (0.29–1.69), p = 0.009 |
| Antiparasitic | 112 (0.34%) | 12 (10.91%) | 26 (2018) | 24 (26.97%) | 54 [44] | 0.89 (0.41–1.36), p = 0.001 |

Drugs associated with DILD
| Drug name | Total adverse event reports | DILD reports (%) | Japan DILD reports (%, n = 31,486a) | Maximum DILD reports in 1 year (year) | Male (%) (n = 29,279a) | Median age [IQR] (n = 26,380a) |
|---|---|---|---|---|---|---|
| Overall database from 2004 to 2021 | 43,046,990 | 32,821 (0.08%) | 14,483 (46.00%) | 3645 (2020) | 16,037 (54.77%) | 68 [59] |
| Methotrexate | 343,703 | 891 (0.26%) | 39 (4.53%) | 197 (2019) | 261 (29.97%) | 64 [56] |
| Doxorubicin | 81,024 | 844 (1.04%) | 138 (16.57%) | 293 (2020) | 142 (41.76%) | 64 [53] |
| Pembrolizumab | 70,054 | 783 (1.12%) | 673 (85.95%) | 229 (2018) | 657 (85.88%) | 71 [66] |
| Nivolumab | 130,296 | 708 (0.54%) | 549 (77.54%) | 160 (2021) | 539 (78.69%) | 69 [62] |
| Amiodarone | 53,830 | 634 (1.18%) | 43 (7.08%) | 105 (2019) | 407 (68.52%) | 76 [69] |
| Etanercept | 1,301,974 | 607 (0.05%) | 218 (36.64%) | 64 (2018) | 178 (31.06%) | 64 [56] |
| Adalimumab | 1,528,994 | 565 (0.04%) | 87 (15.88%) | 56 (2012) | 211 (38.29%) | 65 [55.25] |
| Everolimus | 121,289 | 524 (0.43%) | 286 (54.68%) | 64 (2011) | 242 (50.21%) | 66 [58] |
| Tocilizumab | 165,928 | 494 (0.30%) | 265 (53.64%) | 72 (2020) | 184 (40.17%) | 65 [57] |
| Rituximab | 256,867 | 487 (0.19%) | 33 (6.86%) | 148 (2021) | 127 (51.63%) | 62 [52] |
| Bevacizumab | 163,636 | 470 (0.29%) | 342 (73.08%) | 55 (2012) | 282 (66.67%) | 69 [62] |
| Docetaxel | 159,218 | 448 (0.28%) | 241 (58.92%) | 55 (2012) | 289 (68.65%) | 69 [62] |
| Infliximab | 402,189 | 411 (0.10%) | 133 (37.15%) | 43 (2020) | 138 (37.10%) | 63 [52] |
| Oxaliplatin | 81,014 | 366 (0.45%) | 190 (52.20%) | 57 (2021) | 283 (82.51%) | 70 [63] |
| Abatacept | 179,763 | 357 (0.20%) | 164 (45.94%) | 72 (2021) | 99 (31.43%) | 68 [58] |
| Osimertinib | 23,759 | 343 (1.44%) | 241 (70.26%) | 115 (2019) | 122 (38.98%) | 74 [66.43] |
| Paclitaxel | 75,462 | 341 (0.45%) | 204 (60.00%) | 51 (2019) | 159 (51.62%) | 68 [61] |
| Atorvastatin | 203,754 | 323 (0.16%) | 74 (24.18%) | 44 (2021) | 169 (61.68%) | 70 [65] |
| Erlotinib hydrochloride | 111,517 | 299 (0.27%) | 133 (45.08%) | 41 (2010) | 173 (64.07%) | 67 [60] |
| Cyclosporine | 109,612 | 291 (0.27%) | 198 (70.46%) | 39 (2014) | 155 (57.41%) | 59 [47] |

| Drug name | Total adverse event reports | DILD reports (%) | Japan DILD reports (%, n = 31,486a) | Maximum DILD reports in 1 year (year) | Male (%) (n = 29,279a) | Median age [IQR]a (n = 26,380a) | ROR (95% CI) | PRR (χ2) | IC (IC025) | Labelb |
|---|---|---|---|---|---|---|---|---|---|---|
| Fam-trastuzumab deruxtecan-nxki | 2392 | 96 (4.01%) | 15 (15.63%) | 84 (2021) | 7 (10.45%) | 60 [50] | 56.50 (46.01–69.39) | 54.28 (4957.35) | 5.12 (4.44) | Yes |
| Ramucirumab | 6614 | 170 (2.57%) | 164 (96.47%) | 30 (2018) | 108 (87.80%) | 70.5 [65] | 36.28 (31.14–42.27) | 35.37 (5637.04) | 4.87 (4.36) | Yes |
| Eribulin | 6252 | 137 (2.19%) | 131 (95.62%) | 26 (2018) | 12 (9.09%) | 66 [57] | 30.57 (25.79–36.22) | 29.92 (3812.85) | 4.62 (4.06) | Yes |
| Osimertinib | 23,759 | 343 (1.44%) | 241 (70.26%) | 115 (2019) | 122 (38.98%) | 74 [66.43] | 19.69 (17.68–21.92) | 19.42 (5887.47) | 4.18 (3.82) | Yes |
| Riluzole | 2010 | 40 (1.99%) | 26 (70.27%) | 13 (2020) | 22 (61.11%) | 72 [66.5] | 26.71 (19.53–36.53) | 26.20 (968.99) | 4.02 (2.99) | Yes |
| Durvalumab | 11157 | 155 (1.39%) | 118 (76.13%) | 64 (2019) | 116 (78.38%) | 70 [65.98] | 18.07 (15.41–21.19) | 17.83 (2439.19) | 4.00 (3.47) | Yes |
| Amiodarone | 53,830 | 634 (1.18%) | 43 (7.08%) | 105 (2019) | 407 (68.52%) | 76 [69] | 15.95 (14.74–17.26) | 15.77 (8608.73) | 3.92 (3.66) | No |
| Linagliptin/metformin hydrochloride | 2706 | 44 (1.63%) | 42 (95.45%) | 9 (2019) | 28 (71.79%) | 73 [67.5] | 22.65 (16.81–30.52) | 22.30 (894.08) | 3.92 (2.94) | No |
| Temsirolimus | 8750 | 113 (1.29%) | 52 (46.02%) | 22 (2012) | 92 (84.40%) | 68 [59] | 17.54 (14.56–21.12) | 17.33 (1733.29) | 3.92 (3.30) | No |
| Pembrolizumab | 70,054 | 783 (1.12%) | 673 (85.95% | 229 (2018) | 657 (85.88%) | 71 [66] | 15.98 (14.87–17.16) | 15.81 (10,463.82) | 3.91 (3.67) | Yes |
| Atezolizumab | 26,163 | 285 (1.09%) | 235 (82.46%) | 178 (2021) | 149 (62.87%) | 70 [65] | 14.54 (12.93–16.36) | 14.40 (3498.46) | 3.76 (3.37) | Yes |
| Doxorubicin | 81,024 | 844 (1.04%) | 138 (16.57%) | 293 (2020) | 142 (41.76%) | 64 [53] | 14.17 (13.23–15.18) | 14.03 (9960.03) | 3.75 (3.53) | No |
| Ursodiol | 4598 | 59 (1.28%) | 56 (98.25%) | 7 (2016) | 34 (65.38%) | 69.5 [60.75] | 17.11 (13.23–22.12) | 16.90 (881.73) | 3.74 (2.89) | No |
| Panitumumab | 24,553 | 256 (1.04%) | 231 (90.23%) | 82 (2012) | 203 (82.52%) | 70 [65] | 14.15 (12.50–16.01) | 14.01 (3069.96) | 3.73 (3.32) | Yes |
| Alogliptin benzoate | 2692 | 35 (1.30%) | 35 (100.00%) | 8 (2018) | 21 (65.63%) | 73 [70] | 18.28 (13.09–25.52) | 18.05 (563.29) | 3.61 (2.52) | No |
| Bleomycin sulfate | 3505 | 43 (1.23%) | 15 (36.59%) | 5 (2019) | 30 (69.77%) | 50 [28] | 16.34 (12.09–22.08) | 16.15 (610.83) | 3.59 (2.60) | No |
| Pertuzumab | 12,910 | 120 (0.93%) | 84 (70.00%) | 19 (2015) | 1 (0.85%) | 65.5 [54] | 12.93 (10.80–15.48) | 12.82 (1302.07) | 3.54 (2.94) | No |
| Defibrotide sodium | 4905 | 53 (1.08%) | 2 (3.77%) | 48 (2019) | 27 (55.10%) | 6 [3] | 14.25 (10.87–18.69) | 14.11 (643.95) | 3.50 (2.61) | No |
| Bicalutamide | 9447 | 86 (0.91%) | 46 (61.33%) | 10 (2004) | 83 (98.81%) | 79 [75] | 12.10 (9.78–14.97) | 12.00 (865.46) | 3.41 (2.71) | Yes |
| Ampicillin sodium/sulbactam sodium | 1774 | 21 (1.18%) | 19 (100.00%) | 4 (2013) | 16 (88.89%) | 76 [66] | 15.75 (10.24–24.22) | 15.57 (286.45) | 3.23 (1.83) | No |
Data comparison of reporting countries
| Drug name | DILD reports (%) | Drug name | DILD reports (%) |
|---|---|---|---|
| Japan | United States | ||
| Total country ILD reports | 14,483 (100%) | Total country ILD reports | 4424 (100%) |
| Pembrolizumab | 673 (4.65%) | Amiodarone | 295 (6.67%) |
| Nivolumab | 549 (3.79%) | Etanercept | 177 (4.00%) |
| Bevacizumab | 342 (2.36%) | Methotrexate | 171 (3.87%) |
| Everolimus | 287 (1.98%) | Ambrisentan | 136 (3.07%) |
| Paclitaxel | 283 (1.95%) | Adalimumab | 131 (2.96%) |
| Tocilizumab | 265 (1.83%) | Macitentan | 95 (2.15%) |
| Osimertinib | 242 (1.67%) | Infliximab | 85 (1.92%) |
| Docetaxel | 241 (1.66%) | Treprostinil | 83 (1.88%) |
| Atezolizumab | 235 (1.62%) | Bosentan | 81 (1.83%) |
| Panitumumab | 231 (1.59%) | Fam-trastuzumab deruxtecan-nxki | 78 (1.76%) |
| France | Canada | ||
| Total country ILD reports | 3799 (100%) | Total country ILD reports | 2485 (100%) |
| Amiodarone | 275 (7.24%) | Doxorubicin | 524 (21.09%) |
| Atorvastatin | 138 (3.63%) | Methotrexate | 391 (15.73%) |
| Nivolumab | 81 (2.13%) | Rituximab | 218 (8.77%) |
| Everolimus | 78 (2.05%) | Leflunomide | 138 (5.55%) |
| Atenolol | 66 (1.74%) | Tocilizumab | 100 (4.02%) |
| Bisoprolol | 65 (1.71%) | Abatacept | 87 (3.50%) |
| Esomeprazole | 64 (1.68%) | Etanercept | 73 (2.94%) |
| Gemcitabine | 64 (1.68%) | Adalimumab | 66 (2.66%) |
| Oxaliplatin | 63 (1.66%) | Vincristine | 66 (2.66%) |
| Doxorubicin | 60 (1.58%) | Hydroxychloroquine | 47 (1.89%) |
Discussion
Limitations
Conclusion
Acknowledgments
ORCID iDs
Footnotes
References
Cite
Cite
Cite
Download to reference manager
If you have citation software installed, you can download citation data to the citation manager of your choice
Information, rights and permissions
Information
Published In
Keywords
Authors
Metrics and citations
Metrics
Journals metrics
This article was published in Therapeutic Advances in Drug Safety.
View All Journal MetricsPublication usage*
Total views and downloads: 3361
*Publication usage tracking started in December 2016
Altmetric
See the impact this article is making through the number of times it’s been read, and the Altmetric Score.
Learn more about the Altmetric Scores
Publications citing this one
Receive email alerts when this publication is cited
Web of Science: 21 view articles Opens in new tab
Crossref: 21
- Analysis of interstitial lung disease in pharmacovigilance databases: Coding challenges and interpretation biases—An update
- Preservative-free latanoprost induces meibomian gland dysfunction through inflammatory and oxidative stress pathways
- Safety of immune checkpoint inhibitors: A systematic review of disproportionality analysis studies
- 0.005% Preservative-Free Latanoprost Triggers Meibomian Gland Dysfunction in Mice via Inflammation and Oxidative Stress Modulation
- Pooled analysis of trastuzumab deruxtecan retreatment after recovery from grade 1 interstitial lung disease/pneumonitis
- Drug-Induced Interstitial Lung Disease: A Real-World Pharmacovigilance Study Based on an Adverse Event Reporting System
- Infliximab-induced pulmonary interstitial disease in a male patient with ulcerative colitis: a case report and review of literature
- Tyrosine Kinase Inhibitors and Interstitial Lung Disease: A Disproportionality Analysis Using the European Post-marketing EudraVigilance Database
- Identifying signals of disproportionate reporting for calcitonin gene-related peptide inhibitors: real-world evidence from the FDA adverse event reporting system
- Disproportionality analysis of interstitial lung disease associated with novel antineoplastic agents during breast cancer treatment: a pharmacovigilance study
- View More
Figures and tables
Figures & Media
Tables
View Options
View options
PDF/EPUB
View PDF/EPUBAccess options
If you have access to journal content via a personal subscription, university, library, employer or society, select from the options below:
loading institutional access options
Alternatively, view purchase options below:
Purchase 24 hour online access to view and download content.
Access journal content via a DeepDyve subscription or find out more about this option.



