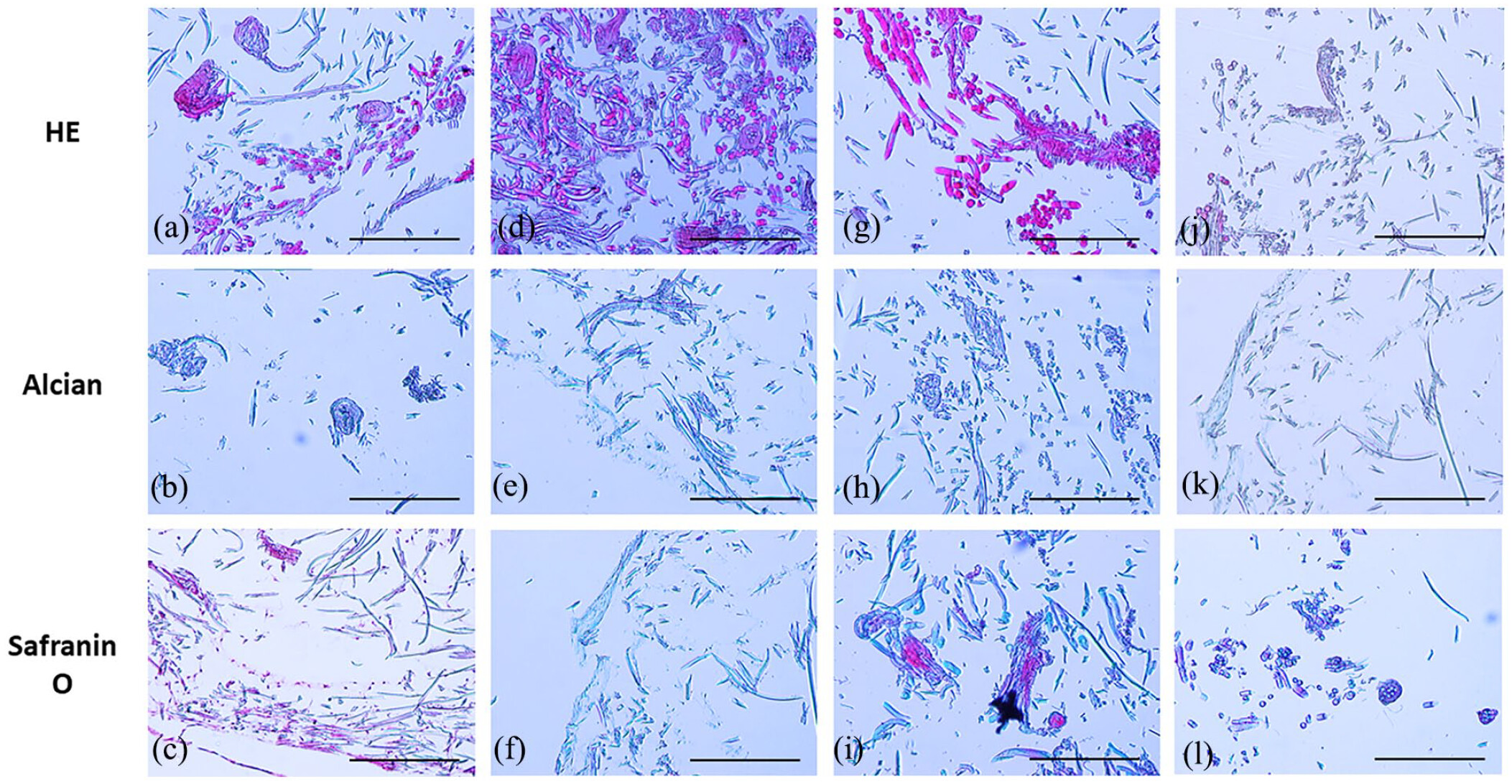
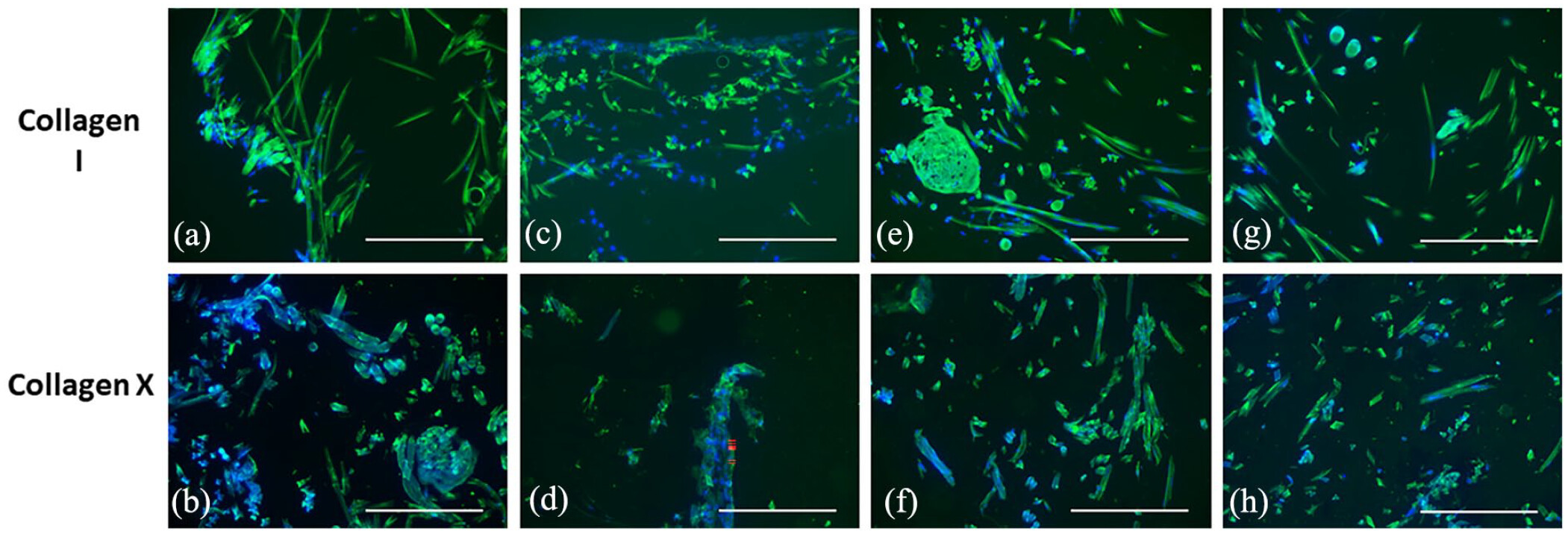
Cartilage tissue consists of chondrocytes and the extracellular matrix (ECM) that mainly composes of proteoglycans and collagens.
1 Chondrocytes produce and maintain the ECM and are essential for the cartilage integrity and function.
2 Due to the avascularity of cartilage tissue, there is almost no regenerative capacity.
3 In research, numerous efforts have been made to develop auspicious approaches for cartilage repair.
4 The approaches are diverse being based on tissue engineering principals, novel biomaterials, various cell types, growth factors and gene therapeutic options.
4 Chondroprogenitor cells, deriving from periosteum,
5 perichondrium,
6 and articular cartilage
7 as well as adipose or bone marrow derived stem cells
8 or allogeneic chondroblasts,
9 in combination with various scaffold materials have been investigated. The multi-lineage capacity of adipose-derived stromal cells (ASC) was discovered in 2001, and soon after they went into the focus of tissue engineering scientists.
10 ASC can differentiate to form bone, cartilage, muscle or fat tissue as well as various other cell types, representing an auspicious perspective for tissue engineering applications.
11–13 Due to their favorable secretion profile of growth factors and cytokines, ASC showed a highly regenerative potential in pre-clinical and clinical settings.
14,15 It was described earlier that ASC also enhance vascularization in de-novo tissue formation.
16 To improve chondrogenic differentiation, most cartilage tissue engineering strategies are combined with free or encapsulated growth factors.
17 As reported elsewhere, ASC differentiated into chondrocytes by applying transforming growth factor beta (TGF-β) as well as ascorbate and dexamethasone to a three-dimensional culture system.
10,18 TGF-β proteins induced chondrogenesis in embryonic
19 and adult stem cells,
20,21 increased ECM synthesis
22 and enhanced proliferation of chondrocytes.
23 TGF-β1 is known to influence cell-cell interactions between chondrogenic progenitor cells,
24 whereas TGF-β2 interferes hypertrophic differentiation, and TGF-β3 influences stem cell differentiation.
25 Other studies achieved chondrogenic differentiation of ASC by supplementing the culture medium with bone morphogenic proteins (BMP), as BMP is known to influence chondrogenesis and osteogenesis.
26 It was shown earlier that BMP-2 and BMP-7 enhance ECM production in chondrogenic progenitor cells and mature chondrocytes.
27 By adding BMP-2 to tissue engineering approaches, increased expression levels of sex-determining region Y box gene 9 (SOX-9),
28 collagen type II,
29 and aggrecan
30 could be observed. As reported elsewhere, BMP-7 increases the ECM production but at the same time reduces the infiltration of fibroblasts into the scaffold material.
31 By adding a combination of 5 ng/mL TGF-β2 and 100 ng/mL BMP-7, it was possible to grow cartilage tissue that was most similar to ordinary cartilage.
32 Besides chemical induction of differentiation, the application of cyclic pressure, which causes mechanotransduction via binding motif-dependent integrins and thus induction of chondrogenic differentiation has been investigated.
26,33 The application of pressure in dynamic cell cultures usually requires a bioreactor model. As published earlier, scaffolds grown in dynamic bioreactor perfusion models showed enhanced cell proliferation and biochemical secretion profiles compared to static cultures.
34 For long-term in vitro cultures in bioreactor systems, shear stress seemed to increase ECM synthesis resulting in greater tissue production.
35 A plethora of custom-made bioreactor systems has been investigated in cartilage tissue engineering including parallel-plate bioreactors,
36 rotating wall bioreactors,
37 wavy-wall bioreactors,
38 and concentric cylinder bioreactors.
39 Regarding successful cartilage tissue engineering purposes, chondrogenic differentiation of ASC could be achieved in a three-dimensional collagen scaffold with treatment of cyclic hydrostatic pressure in a bioreactor model.
26For cartilage tissue engineering, different biomaterials have been investigated such as alginate, agarose, cellulose, chitosan, chondroitin sulfate, collagen, fibrin, and gelatin.
35 As described elsewhere, a three-dimensional scaffold structure that enhances cartilage-like tissue production should be provided by the biomaterial.
35,40 High initial cell seeding density as well as cell-cell interactions showed increased ECM production and deposition.
41 Furthermore, the scaffold material should be highly biocompatible and biodegradable as well as sufficiently mechanically loadable.
42 The mechanical properties of the chosen scaffold material depend on mechanical strength, elasticity and biodegradation.
42 Due to its unrivaled biomechanical properties, spider silk of the species
Nephila edulis represents a highly promising scaffold material for tissue engineering. According to current knowledge, there are seven different types of spider silk, each with different biomechanical properties.
43 For example, silk from the tubuliform gland, used for the egg cocoons, provides a porous structure with high toughness whereas dragline silk from the major ampullate gland has a high tensile strength, reaching up to 4 × 10
9 N·m.
1,44 At the same time, dragline silk showed auspicious results regarding in vitro and in vivo biocompatibility.
45,46 Due to its three-dimensional porous structure, its compressive strength and the successful support of chondrocyte growth, cocoon silk seems promising for cartilage tissue engineering purposes.
47 The concept of the present proof-of-concept study was the investigation of spider silk cocoons as scaffold seeded with ASC in a bioreactor model to engineer cartilage-like tissue. For chemical induction of differentiation, BMP-7 and TGF-β2 were added to enhance chondrogenic differentiation.