Introduction
The most prevalent protein with fibrous structure in all organisms, collagen (C), serves as the primary functional component of extracellular structures in tendons and ligaments. Glycine is found in every third location of the proteins considerably formed themselves triple-helix construction, which is arranged helicoidally. The substances from which collagen molecules are extracted and the post-processing conditions can change the crystalline structure and molecular features, particularly their ability to interact with the derived components. Recently, collagen fibers are being widely explored due to the growing interest in novel biomaterials enhanced with molecules of collagen for a variety of biological purposes.
1 Among the many biological sources of collagen used for health advantages, bovine skin, tendons, and intestines are potential candidates for widespread use as biomedicine. Collagen-based polymers have been employed in both pharmaceuticals and healthcare. Nearly every physical form, including liquids, gels, powders, fibers, membranes, sponges, tubes, etc., contains collagen.
Fracture healing is a complex biological phenomenon that involves the restoration of structural integrity through bone regrowth. Losing a tooth can be distressing on the mind. Replacement attempts for missing teeth were performed even in primitive times. Implant dentistry unique is in its ability to achieve this goal despite stomatognathic system atrophy, disease, or damage. To achieve the optimum objectives of implant dentistry, the hard and soft tissues must have appropriate volumes and attributes. If there is not enough bone, a variety of surgical methods can be used to reconstruct the weak ridge for implant implantation.
2 Bone tissue engineering technologies have been explored for a variety of dental applications such as salivary gland regeneration, tooth development, and pulp dentin complex regeneration, with promising results.
3 Javed et al.
4 report the bone growth factors have a significant impact on protein synthesis, chemotaxis, cellular proliferation, and differentiation. Non-collagenous proteins found in demineralized dentin and bone matrices include dentin matrix protein-1, dentin sialophosphoprotein (DSPP), osteonectin, osteopontin, and bone sialoprotein. Bone morphogenetic proteins (BMPs) and fibroblast growth factors are also found in demineralized matrices.
5Bone manipulation methods can affect the density of a person’s bones to improve their tensile strength and durability. These methods mobilize necessary bone through the plastic bending, shaping, or condensing of tissue into a bone flap or bone-periosteal flap. These change the size or shape of the bone while keeping it healthy and whole. The goal is to modify the residual bone to create an intra-body chamber that heals similarly to an extraction site and allows mesenchyme stem cell accessibility in addition to normal wound healing mechanisms. Choosing methods for ridge manipulation and the form of the bone defect could be taken into account. The main drawback of synthetic implants is their substantial modulus of elasticity, which results in a stress-shielding impact that increases bone resorption since cortical bone’s elastic modulus is incongruent with titanium’s elastic modulus.
6 Fractures and faults may already exist, or they may develop as a result of the implant. Blood transfusions are performed in the human body more frequently than bone transplants. Therefore, it is essential to carefully consider orthopedic device design to protect patients and efficiently treat bones and joint disorders.
7The parameters of design and choice of material for bone implants are determined by functional and biomechanical requirements. To achieve biocompatibility, a variety of biomaterials are used, including metals, ceramics, polymers, and composites. Biomedical implants can take the role of worn-out organs, joints, and tissues. For them to achieve integrity, according to International Standards Organization (ISO) 20160, a homogeneous and stable microstructure is required. An ideal implant should be able to work properly without requiring any more revision surgery. The majority of orthopedic biomedical implants are used for hip, knee, spinal, and maxillofacial replacements.
8Dental implant (DI) technology has progressed in the past few years, providing patients with unparalleled levels of affordability, convenience, and effectiveness. This is one of the main reasons why so many dentists believe that DIs are the best approach to replace missing teeth. Natural-based DIs can provide numerous advantages.
9 Previously, the researchers considered a natural-based implant in tissue engineering and dental surgery while focusing on cost-effective techniques. The main aim of this research work focused on the preparation of natural material-based DIs and evaluated using the mechanical, physicochemical, and biological properties.
Discussion
Patients who have DIs could be able to talk and eat more effortlessly because their dentures are not at risk of dislodging. Dentures must be replaced when the gum tissue shrinks and the fit changes; implants are not affected by this issue. DIs require less upkeep and hygiene than dentures. Another factor driving the continuous expansion of the global market for DIs is the fact that DIs are an effective treatment for edentulism, as well as the increased desire for cosmetic dentistry across all age groups worldwide.
12Choosing the best bone implantation material from the many choices available is difficult. Although the level of failures and surgical factors must be addressed when selecting a graft material, the collagen-based DI may be offered an alternative when tooth extraction is essential due to its autogenous origin and favorable biocompatibility outcomes. In this study, DI was prepared using natural resources and it was evaluated using bone tissue engineering. The observed quality of DI prepared from MCF and Si-NPs were higher mechanical properties when compared to quality from other implants like Osuchukwu et al.
13Collagen has been found in living organisms as a part of bone, cartilage, and fibrous tissue. More than 90% of the protein in the bone matrix is collagen. The majority of the collagen in bone is Type I collagen, which is produced by osteoblasts. Cross-linking connections are generated between the molecules of collagen in collagen fibrils, which produce interconnected collagen filaments, which develop collagen molecules self-associate into aggregated to form collagen fibrils.
14 During the growth, remodeling, and regeneration of bone, osteoblasts produce collagen fibers that are then coated with calcium phosphate, completing the process of building a robust bone matrix.
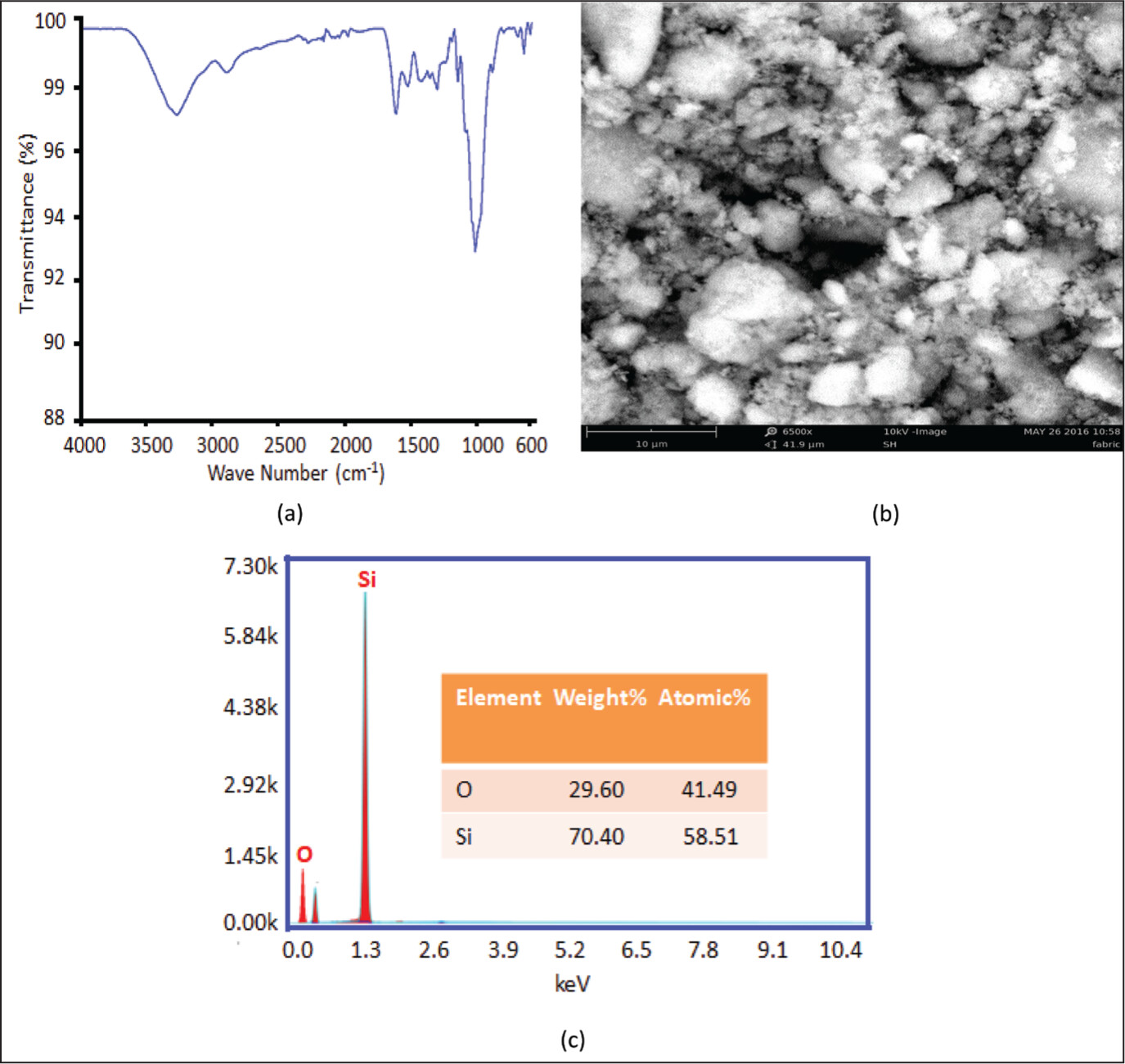
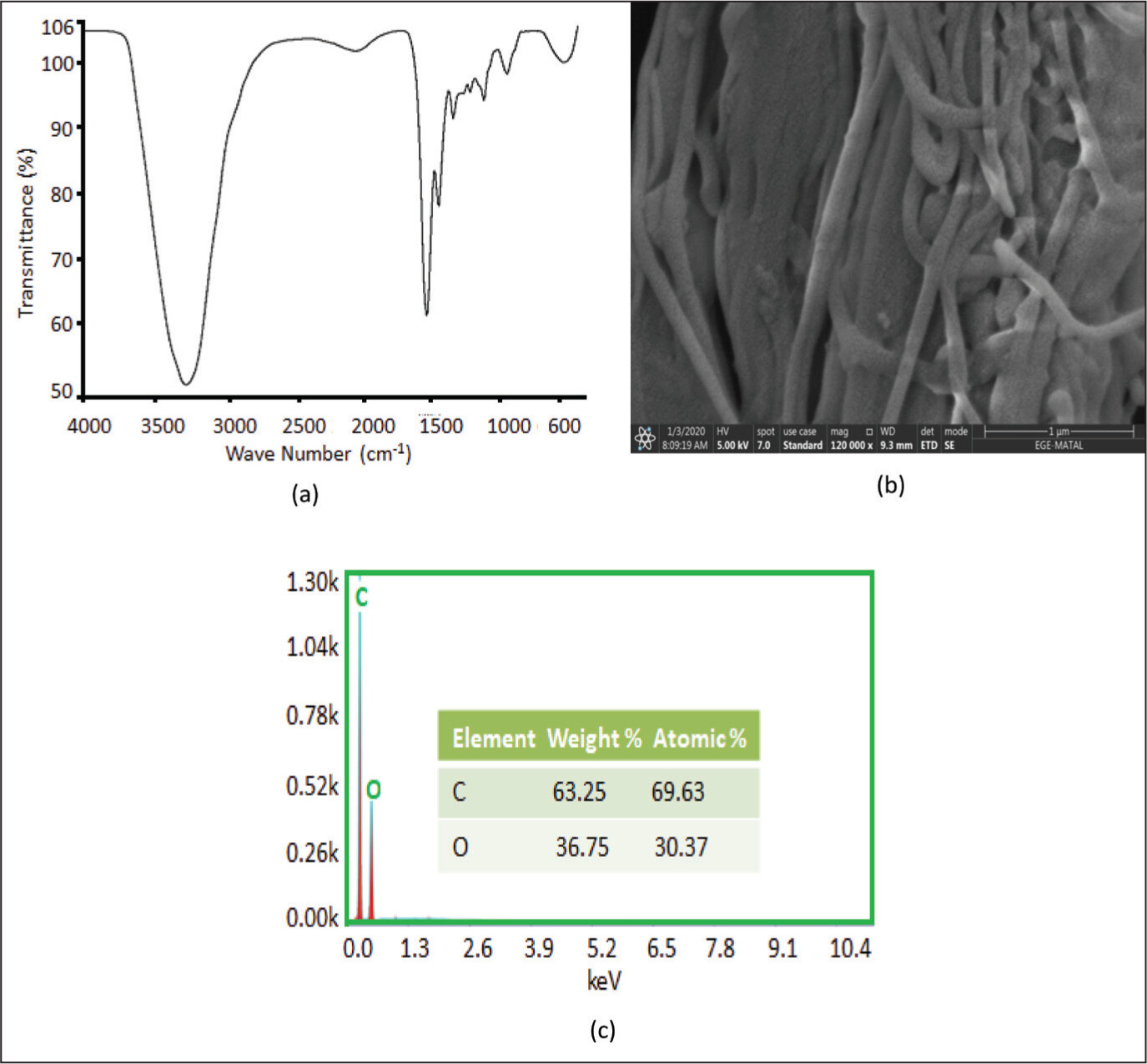
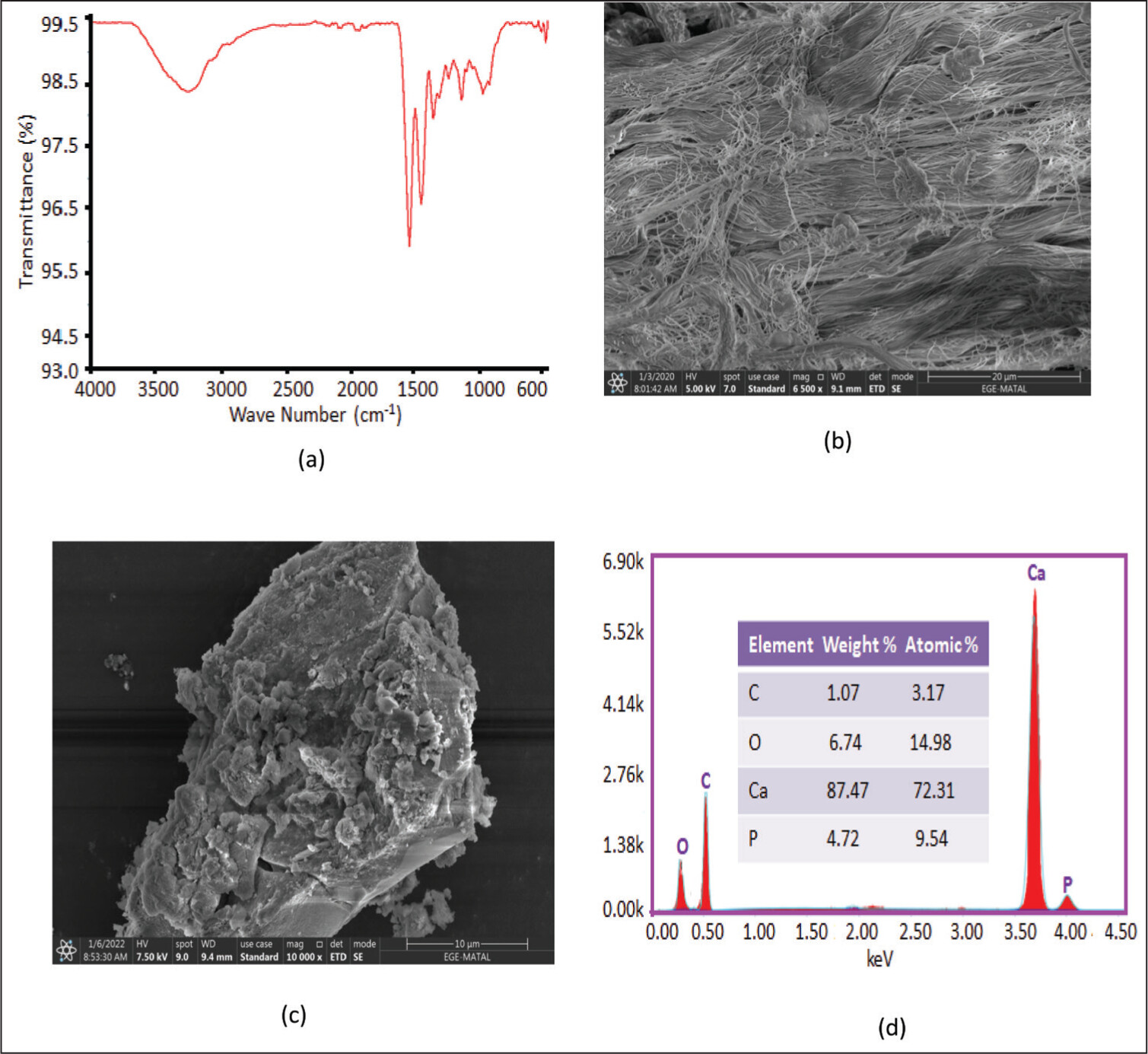
15 Silica nanoparticles (Si-NPs) are the ideal nanomaterials for bone tissue engineering because of their distinctive qualities, including high specific surface area, pore size, multi-functional ability, variable shape, and good biocompatibility.
16 Bioactive bone fillers composed of collagen and calcium phosphate have been used in bone repair treatment for many years. They are renowned for their excellent biocompatibility, osteoconductivity, and surgical handling efficiency. However, because of their poor mechanical qualities, such as their extreme brittleness, these bone replacements are frequently not suitable for weight-bearing purposes.
17The presence of these collagen triple helix absorption peaks suggests that the triple helix of structural collagen was mostly retained after mineralization. The P and Ca elements throughout the mineralization process changed the calcium-deficient HA and apatite crystals into amorphous calcium phosphate (ACPs).
18 The scissor-like bend vibration of water molecule H
2O causes the band at 1637 cm
–1. Similar to this, the Si–O and Si–O–Si linkage vibrations’ asymmetric stretching can be seen in the band at 1097 and 802 cm
–1, respectively. Additionally, the peak at 463 cm
–1 was related to the O–Si–O modes’ bending vibration.
19 The Si-NPs result is explained by the greater surface area and persistent affinity of nanoparticles throughout the dehydration process, which results in the agglomeration of the nanoparticles.
20 The phytochemicals present in plants and the extraction technique has a major impact on the longevity of NPs and their aggregation into aggregates. The Si-NPs cling together as a result, and they independently produce asymmetrical clusters.
21 The measurement efficiency depends on Si-NPs compounds with a low standard number of atoms, like polymers and oxides, making it difficult to identify Si-NPs smaller than 20 nm with modern instruments.
22Hydrogen bonding between collagen, calcium carbonate, and Si-NPs causes the implant to widen and OH vibration of stretching to a lower wave number. It is stated that electrostatic and hydrogen bonding interactions between collagen and the Si-NPs are expected non-covalently.
23 The results of the research support the earlier publication about collagen-based bone implants. The collagen-based implant materials need to have enough mechanical strength to handle them and be used in the intended medical applications.
24 The expected characteristics from the implants are near to the features of the desired bone in terms of sufficient mechanical strength, modulus of elasticity, corrosion behavior, as well as biocompatibility to prevent adverse effects on humans.
25 Design parameters and material selection for bone implants are determined by functional and biomechanical requirements. To achieve biocompatibility, a variety of biomaterials are used, including composites, polymers, ceramics, and metals.
26 According to Kokubo and Takadama,
27 the development of a phosphate-containing calcium layer on an artificial substance is a crucial step for in vivo bone growth, commonly referred to as bone-like apatite, on the surface of the material. The bone-like apatite seems to trigger signaling proteins and tissues to start the chain of reactions that results in bone formation. DIs’ porous design not only aided in cell migration but also allowed for the transfer of oxygen and nutrients with cells, which is crucial for the formation of the Matrix and vascularization during bone tissue regeneration.
28Conclusion
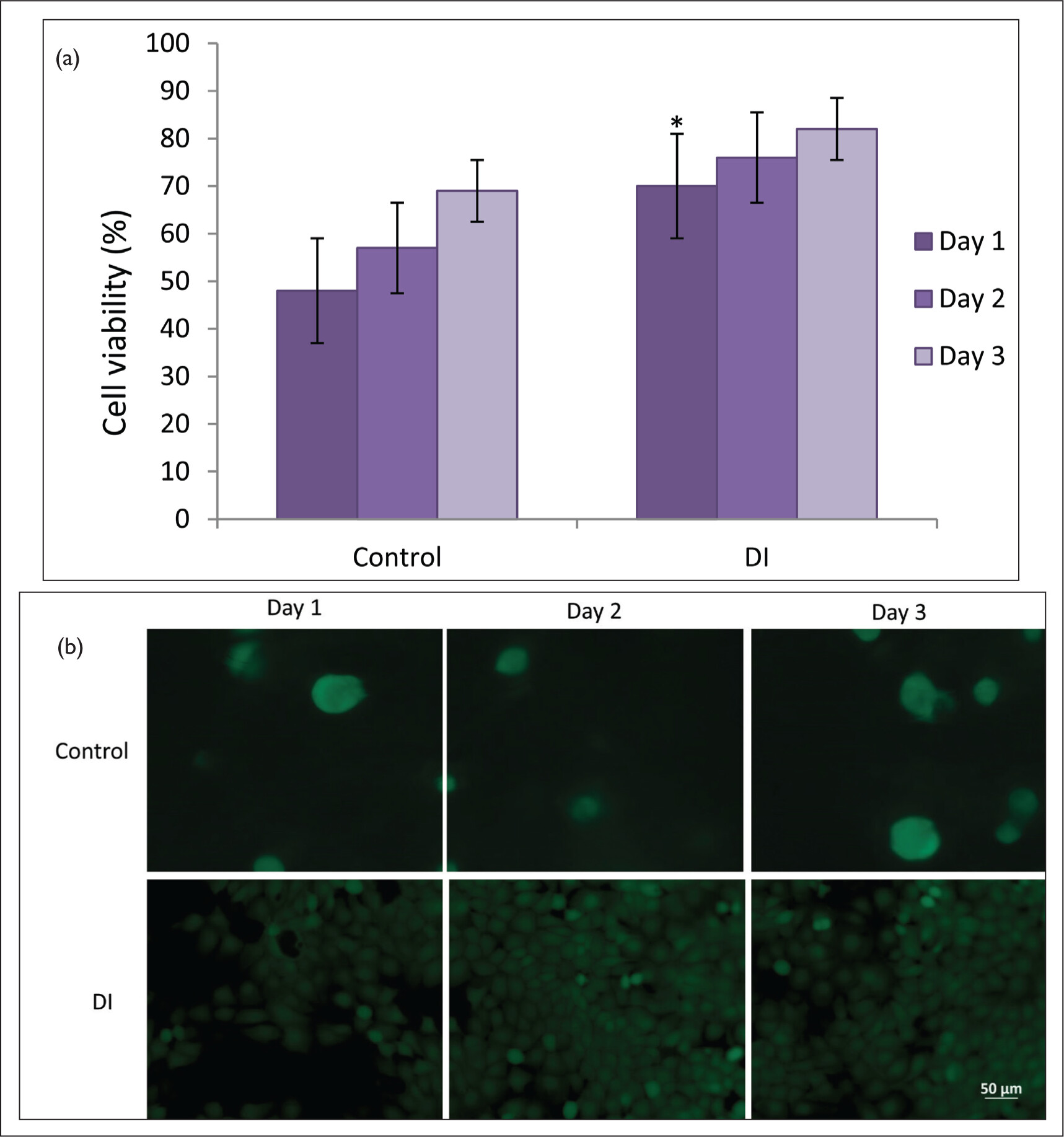
In this research, we introduce a DI that contains Si-NPs with the potential for bone healing. The prepared DI has shown outstanding mechanical properties, porous structure, bioactivity test, and biocompatibility study. Studies done in vitro showed that the implant increased cell viability, making it the best substrate for bone tissue engineering. Overall, the DI offers potential uses in bone tissue engineering, including the promotion of bioactive materials, dental soft and hard tissue healing, and new bone tissue formation. The present difficulty is scaling up the manufacturing of collagen and making it inexpensive for the general population while effectively incorporating MCF and Si-NPs into the development of nano-based biomaterials.