Introduction
Radical cystectomy (RC) is the gold standard surgical treatment for patients with localized muscle-invasive bladder cancer, and also patients with Bacillus Calmette-Guerin (BCG) unresponsive non-muscle invasive bladder cancer disease and those with non-muscle invasive bladder cancer in very high-risk group for progression according to current European Association of Urology (EAU) Guidelines.
1 However, despite RC, the high rates of morbidity and mortality highlight the importance of identifying factors that can guide the selection of appropriate candidates for this procedure.
2 In addition to the existing clinical staging systems, the introduction of new parameters may assist clinicians in optimizing patient selection and treatment planning.
3–5Sarcopenia has recently received significant attention as an objective preoperative prognostic factor. It is strongly associated with poorer survival outcomes across various cancer types.
4,5 Based on recent studies, sarcopenia may be an important factor to consider when assessing the survival rate of BC patients.
6The diagnostic criteria for sarcopenia are widely established by the European Working Group on Sarcopenia in Older People (EWGSOP).
7,8 Most studies prefer to diagnose sarcopenia using radiological imaging methods, as these provide higher levels of standardization and objectivity.
There is no comprehensive study evaluating multiple gender-specific radiologic markers of sarcopenia that may be associated with overall survival in patients undergoing radical cystectomy, comparing these markers, and also providing relevant cut-off values for each marker in the Turkish population for this surgery. Therefore, in a retrospective study reflecting the experience of tertiary center, we aimed to contribute to the literature by evaluating the impact of radiologically defined sarcopenia parameters on overall survival in patients undergoing radical cystectomy for bladder cancer and by determining cut-off values specific to the our study. It is hypothesized that these parameters are independent prognostic factors for overall survival.
Materials and methods
Patient selection
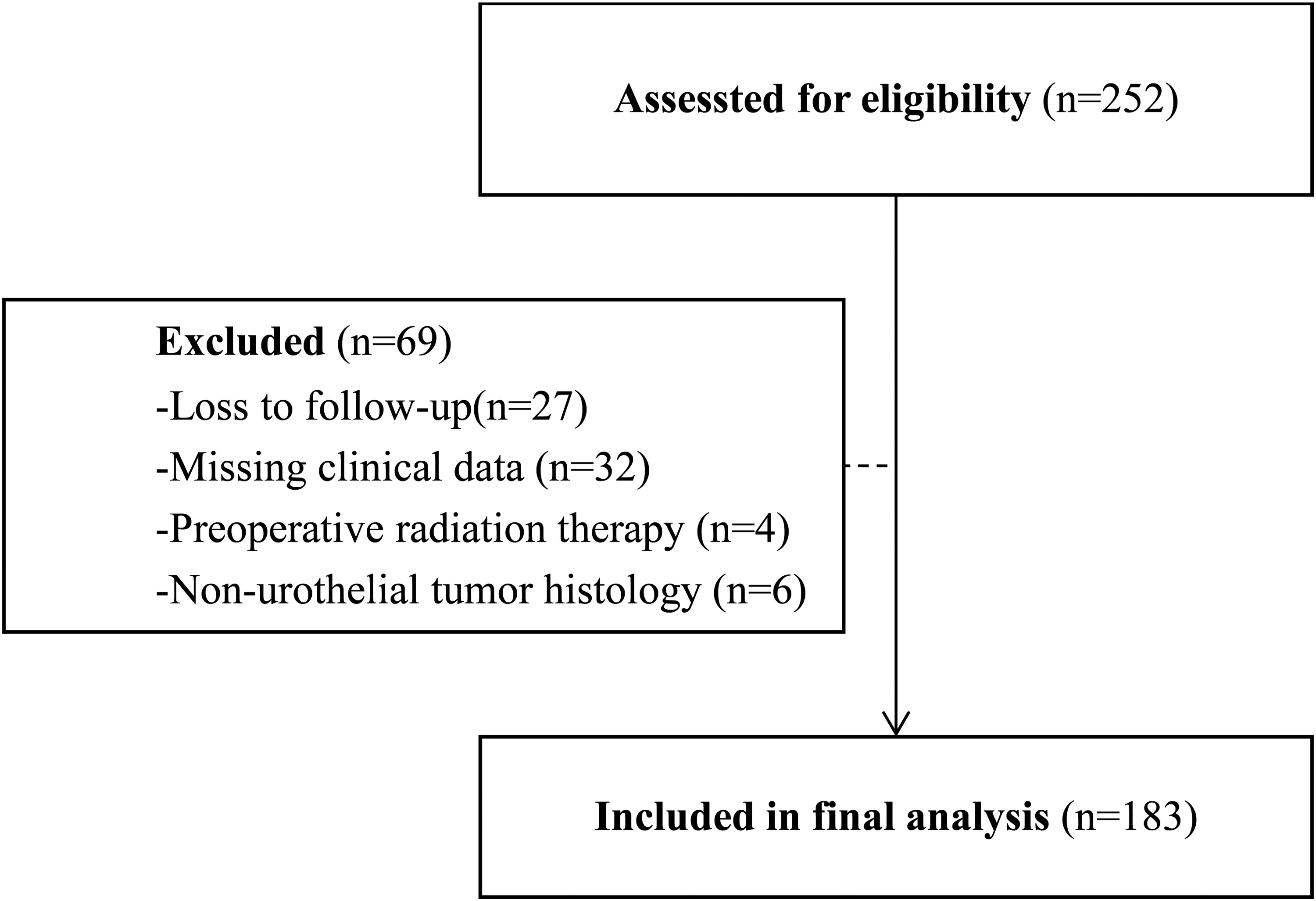
A retrospective review of data from a consecutive 252 patients who underwent radical cystectomy with bilateral pelvic lymphadenectomy for bladder cancer between January 2009 and January 2022 was conducted. A total of 69 patients were excluded from the study due to loss to follow-up, non-urothelial tumor biology in cystectomy specimen, preoperative radiation therapy history or missing clinical data (
Figure 1). Patients who underwent salvage cystectomy were excluded from the study because they were associated with a high risk of perioperative major complications and therefore early mortality. Patients who received NAC were also included in the study, especially since NAC is recommended before cystectomy by current guidelines. The study included 183 patients who had undergone preoperative abdominal computed tomography imaging within three months before surgery, had been diagnosed with urothelial carcinoma. The demographic, clinical, pathological, oncological, and radiological data of the patients included in the study were accessed. The demographic and clinical data included age, gender, height, weight, body mass index (BMI), smoking status, Age-adjusted Charlson Comorbidity Index (ACCI), time between transurethral resection and radical cystectomy, neoadjuvant chemotherapy history. Pathological data consisted of pathological T (pT) stage, pathological N (pN) stage, presence of variant histology of urothelial carcinoma, and number of lymph nodes removed, adjuvant chemotherapy history, recurrence of disease or death any cause in follow-up was for oncological data. Disease recurrence was defined as any recurrence in the loco-regional recurrence (i.e., the cystectomy site or pelvic lymph nodes), any metachronous recurrence (i.e., distant lymph nodes, bones, or visceral organs), and any urothelial recurrence. Radiological data included multiple numeric measurements based on skeletal muscle, psoas muscle, lomber third vertebrae, as comprehensively described by formulations below.
The oncological follow-up was conducted in accordance with institutional protocols and current EAU guidelines.
1Computed tomography image analysis based calculation of radiological sarcopenia parameters
A 64-detector computed tomography (CT) scanner (Toshiba Medical Systems, Otawara, Japan) was used for imaging. The imaging data were accessed through the Picture Archiving and Communication System (PACS). Images obtained with a 1.25 mm slice interval in non-contrast series were analyzed. Muscle analysis from the CT images was performed using the web-based software package CoreSlicer (version 1.0.0; Montreal, Quebec, Canada).
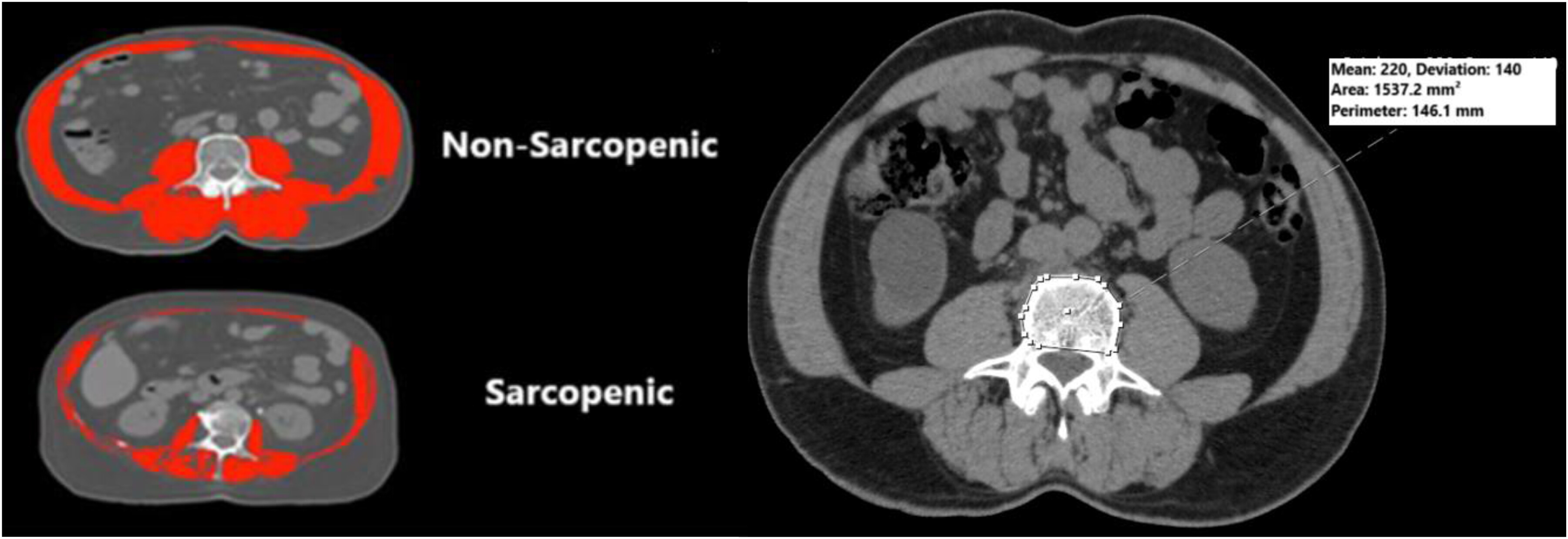
The CT image slices were retrieved from PACS and imported into CoreSlicer for analysis. The skeletal muscles at the level of the L3 vertebrae, including the rectus abdominis, lateral and oblique abdominal muscles, psoas major, quadratus lumborum, and erector spinae muscles, were assessed using a single cross-sectional slice.
9 The total lumbar skeletal muscle area (SMA) at the L3 level was used because it correlates strongly with whole-body muscle volume.
7To identify muscle structures, areas within the (−29) to (150) Hounsfield Unit (HU) range were automatically calculated by the software and manually adjusted when necessary. Through analysis on PACS and CoreSlicer, the SMA, psoas muscle area (PMA), and skeletal muscle density (SMD) values of the patients were calculated as follows:
The areas of muscle structures identified at the midaxial level of the L3 vertebra were measured in square centimeters (cm
2). To normalize the SMA and the (PMA, they were divided by the square of the patient's height (cm
2/m
2). This normalization yielded the skeletal muscle index (SMI) (abbreviated in this study as SMI type 1) and psoas muscle index(PMI) (abbreviated in this study as PMI type 1) as defined in the literature.
4,5,10 For this study, the vertebral cross-sectional area (VCA) at the L3 level was also used for normalization. SMA and PMA were normalized by VCA, resulting in SMI type 2 and PMI type 2. The average HU value of the skeletal muscles was calculated to obtain the SMD.
SMI type 1: Total muscle area at the L3 vertebra level (SMA) / height (cm2/m2).
SMI type 2: Total muscle area at the L3 vertebra level (SMA) / L3 VCA.
PMI type 1: Sum of the areas of the right and left psoas muscles at the L3 vertebra level (PMA) / height (cm2/m2).
PMI type 2: Sum of the areas of the right and left psoas muscles at the L3 vertebra level (PMA) / L3 VCA.
SMD: Average HU value of all muscles at the L3 vertebra level (Figure 2). Optimal threshold determination of radiological sarcopenia parameters
Since muscle mass varies between populations due to differences in ethnicity, geography, and dietary habits, which explains the varying threshold values for sarcopenia found in different studies regarding cancer survival.
4,5,10,11 In this study, the threshold values (separately for males and females) and the area under the Receiver Operating Characteristics (ROC) curves for radiological parameters used to assess the relationship between these parameters and overall survival were calculated using ROC analysis. Based on the gender-specific thresholds identified for each radiological parameter, patients were classified into two groups: sarcopenic and non-sarcopenic, and overall survival analysis was performed.
Statistical analysis
Statistical analysis was performed using IBM SPSS version 20.0 (IBM Corp., Armonk, NY, USA). The normality of the data distribution was assessed using the Shapiro-Wilk and Kolmogorov-Smirnov tests. Variables with a normal distribution were presented as mean ± standard deviation, while non-normally distributed variables were expressed as median (25th-75th percentile).
Cut-off values, area under the curve (AUC), sensitivity, and specificity were calculated using ROC analysis in the MedCalc® version 12.5.0.0(MedCalc® Software, Mariakerke, Belgium). Categorical variables were presented as frequencies (percentages). Differences between groups were determined using the independent samples t-test for normally distributed data or the Mann-Whitney U test for non-normally distributed data. The Chi-square test was used for the statistical evaluation of categorical groups.
Univariate and multivariate regression analyses were performed using Cox regression analysis. Overall survival between groups was estimated using the Kaplan-Meier method, and statistical differences between these curves were assessed using the Log-Rank test.
A p-value of <0.05 was considered statistically significant for all hypothesis tests.
Ethical approval
This study was conducted in accordance with the project approval numbered 2022/281 obtained at the Kocaeli University Non-Interventional Clinical Research Ethics Committee meeting dated 24.11.2022.
Results
Of the 183 patients included in the study, 156 (85.2%) were male. The median age was 65 (60–71,IQR) years. Preoperatively, the mean height was 168.9 (±7.26) cm, the mean weight was 73.9 (±10.63) kg, and the median BMI was 25.4 (23.6–27.6) kg/m2.
A history of smoking was present in 146 patients (79.8%). The median age-adjusted Charlson Comorbidity Index (CCI) was calculated as 3 (2–4). Variant histology was observed in 27.3% of patients, with squamous variant being the most common (78%).
According to final pathological specimen analysis, 65 patients (35.5%) had localized disease (pT0-2N0), while 118 patients (64.5%) had locally advanced disease (pT3-4N0 or pT0-4N+). Lymph node involvement was observed in 38.3% of patients. Positive surgical margins were found in 7.7% of patients, and carcinoma in situ (CIS) was identified in 24.3%. Median number of lymph nodes removed were 18 (13–27).
A total of 38 patients (20.8%) received neoadjuvant chemotherapy, while 82 (44.8%) underwent adjuvant chemotherapy. Recurrence was observed in 44.8% of patients, with 21% of these being loco-regional, 51.8% distant metastases, 6.2% metachronous urinary tract recurrence, and 21% multiple site recurrences (both loco-regional and distant). The mean hospital stay was 13 days (±5), and the median follow-up period was 22 months (10–50). During the follow-up period, 120 (65.6%) patients died (
Table 1).
As shown in
Table 1, significant differences were found between genders in terms of height, weight, smoking history, and radiological parameters related to sarcopenia (p < 0.05). Radiological parameters related to sarcopenia did not significantly different in patients treated neoadjuvant chemotherapy and those not treated with neoadjuvant chemotherapy (p > 0.05).
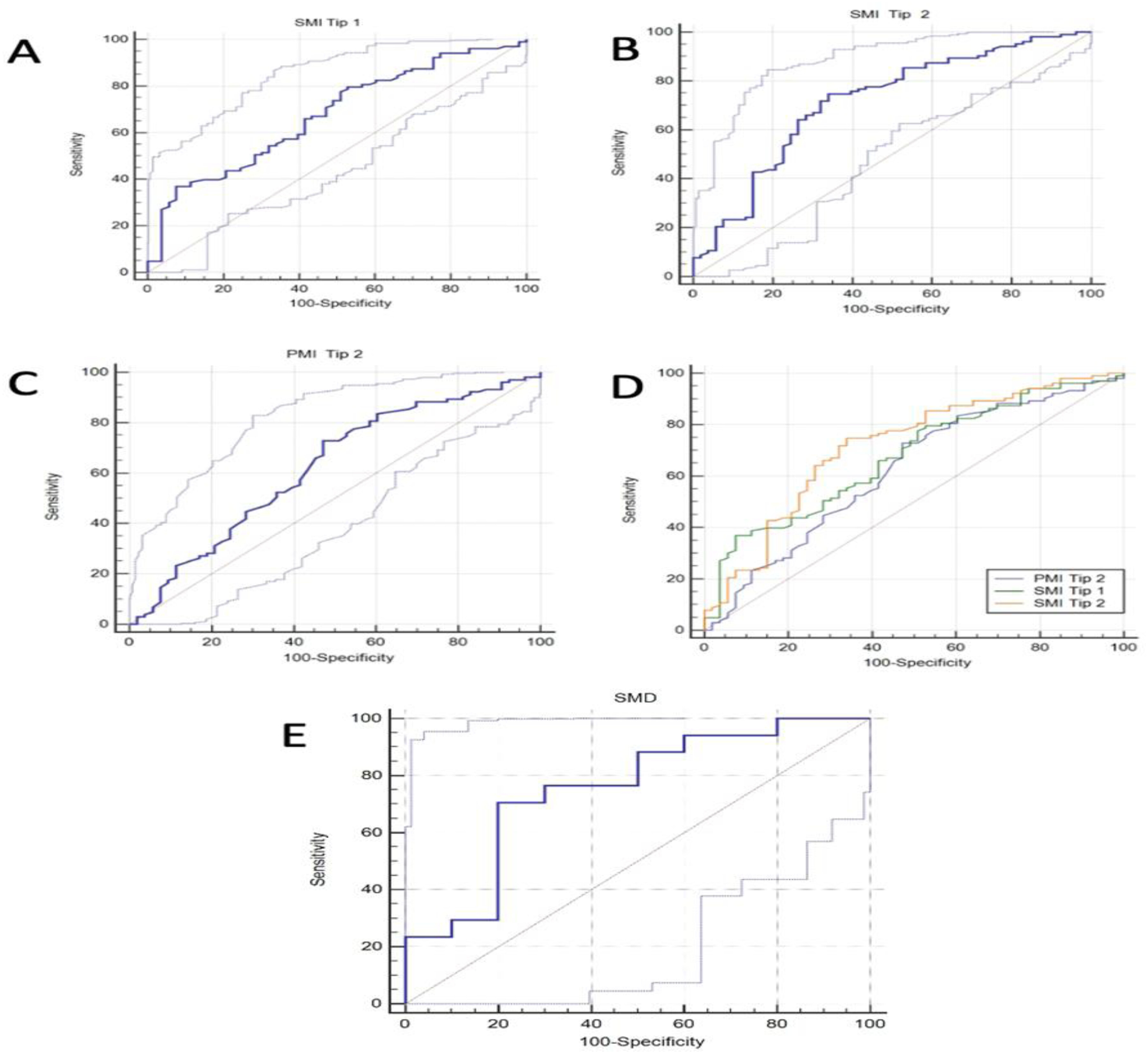
Figure 3 shows ROC analysis for sarcopenic parameters for overall survival. For male patients, the threshold values were calculated as follows: 45.3 cm
2/m
2 for SMI type 1, with an AUC value of 0.67; 9.53 for SMI type 2, with an AUC value of 0.71; and 1.47 for PMI type 2, with an AUC value of 0.62. In female patients, only the SMD threshold value was determined to be 39 HU, with an AUC value of 0.75.
Among male patients, there was a statistically significant difference in age and BMI between sarcopenic and non-sarcopenic patients based on radiological parameters (p < 0.05). In female patients, when comparing sarcopenic and non-sarcopenic groups based on radiological parameters, only age showed a significant difference (p < 0.05, Supplement Table 1).
Univariate and multivariate analyses were conducted to identify factors affecting overall survival. In male patients, univariate Cox regression analysis showed that age (HR: 1.56 95% CI:1.04–2.34), pT stage (HR: 2.22 95% CI:1.45–3.39), pN stage (HR: 1.89 95% CI:1.26–2.86.), SMI type 1 (HR: 2.48 95% CI:1.64–3.73), SMI type 2 (HR: 2.75 95% CI:1.75–4.32), and PMI type 2 (HR: 2.07 95% CI:1.33–3.2) were significantly associated with overall survival (p < 0.05). In female patients, univariate Cox regression analysis revealed that pT stage (HR: 3.64 95% CI:1.26–10.52), pN stage (HR: 3.21 95% CI:1.15–8.92), and only SMD (HR: 3.36 95% CI:1.17–9.64) were significant predictors of overall survival (p < 0.05) (
Table 2).
Multivariate Cox regression analysis in male patients involved three separate models adjusting age, pT, pN and each radiological sarcopenia parameter (SMI type 1, SMI type 2, and PMI type 2), which had been found as significant predictor in univariate analysis. In all multivariable models, the SMI type 1, SMI type 2, and PMI type 2 remained significant for overall survival (p < 0.05). On the other hand, in female patients, when modeling based on SMD, the significance of pT and N stages was disappeared, while SMD remained a significant independent prognostic factor for overall survival (p = 0.018)
(Table 3).
There was no significant difference in overall survival rates between male and female patients. The 24-month cumulative overall survival rate was 53.4% for men and 38.9% for women (Log-rank p = 0.704, Supplement Figure 1).
Among 156 male patients, there was a significant difference in overall survival between sarcopenic and non-sarcopenic groups categorized according to radiological sarcopenic markers including SMI type I, SMI type 2, and PMI type 2. In the SMI type 1 classification, the 24-month cumulative overall survival rate was 33.2% for sarcopenic patients (n = 42) and 61.1% for non-sarcopenics (n = 114) (Log-rank p < 0.001). In the SMI type 2 classification, the 24-month cumulative overall survival rate was 42% for sarcopenic patients (n = 95) and 71.7% for non-sarcopenics (n = 61) (Log-rank p < 0.001). When classified by PMI type 2, there was also a significant difference between sarcopenic (n = 100) and non-sarcopenic (n = 56) male patients, with 24-month cumulative overall survival rates of 43.9% and 70.8%, respectively (Log-rank p = 0.001) (Supplement Figure 2).
In female patients classified by SMD, the 24-month cumulative overall survival rate was 21.4% for sarcopenic patients (n = 14) and 58.7% for non-sarcopenics (n = 13), with a statistically significant difference (Log-rank p = 0.014).
Subgroup analyses performed for localized (pT0-2N0) and locally advanced disease (>pT2 or pN+) by Kaplan-Meier survival curves indicated that 101 male patients with locally advanced bladder cancer when classified by SMI type 1, the 24-month cumulative overall survival rate was 27.6% for sarcopenic patients (n = 29) and 52.1% for non-sarcopenics (n = 72) to be statistically different (Log-rank p = 0.001). In the SMI type 2 classification for this group, the 24-month cumulative overall survival rate was 34.4% for sarcopenic patients (n = 64) and 63.8% for non-sarcopenics (n = 37) to be statistically different (Log-rank p < 0.001). Similarly, in the PMI type 2 classification, the 24-month cumulative overall survival rates for sarcopenic (n = 68) and non-sarcopenic (n = 33) male patients with locally advanced bladder cancer were 36.8% and 62.5%, respectively, as statistically different (Log-rank p = 0.005 (Supplement Figure 3).
Among 55 male patients with localized bladder cancer (pT0-2 and pN-), the 24-month cumulative overall survival rates were 44.9% for sarcopenic patients (n = 13) and 76.2% for non-sarcopenics (n = 42) to be statistically different when classified by SMI type 1 (Log-rank p = 0.002). When classified by SMI type 2, the 24-month cumulative overall survival rates were 57.5% (sarcopenic, n = 31) and 83.3% (non-sarcopenic, n = 24), respectively, as statistically different (Log-rank p = 0.021)(Supplement Figure 4). However, there was no statistically significant difference in 24-month cumulative overall survival between male patients classified as sarcopenic or nonsarcopenic according to PMI Type 2 with localized bladder cancer (Log-rank p = 0.118, Supplement Figure 5).
In 17 female patients with locally advanced bladder cancer (> pT2 or pN+), all sarcopenic (n = 10) patients classified by SMD died within 24 months, while the 24-month overall survival rate in the non-sarcopenic group (n = 7) was 34.3% as statistically different (Log-rank p = 0.031). There was no statistically significant difference in 24-month cumulative overall survival between female patients classified as sarcopenic or nonsarcopenic according to SMD with localized bladder cancer (Log-rank p = 0.295) (Supplement Figure 6).
Discussion
Radical cystectomy is a challenging surgical procedure, often performed in older adults, with high rates of morbidity and mortality. A decrease in muscle density in the body may be an indicator of a catabolic process such as muscle breakdown. In this context, recent studies have demonstrated that muscle mass can predict complication rates, perioperative mortality, and long-term mortality in patients with bladder cancer treated with radical cystectomy.
12–14 Especially in the era of developing radiologic-based artificial intelligence, radiologic sarcopenia assessment of patients scheduled for radical cystectomy may provide us with a non-invasive prediction of how the survival of patients will progress. Therefore, determining whether patients undergoing radical cystectomy are sarcopenic may help in patient selection and prognosis prediction.In this study, sarcopenia-associated markers were identified by analyzing computed tomography images and calculating the cross-sectional areas of skeletal muscles at the L3 vertebral level. Based on these measurements, gender-specific optimal cutoff values were established, and sarcopenic patients were categorized according to the specific markers. Radiologically based sarcopenic assessments were used to predict overall survival, with each marker being evaluated individually and comparatively. Our study has shown that there are clear differences between male and female patients in terms of muscle mass or sarcopenia markers. It was necessary to examine the markers associated with sarcopenia separately in these two genders with different muscle masses, and we emphasized this point.
The optimal cutoff value for SMD was determined as 39 HU in females, while in males, the optimal cutoff values for SMI Type 1, SMI Type 2, and PMI Type 2 were calculated as 45.3 cm2/m2, 9.53, and 1.47, respectively. Patients identified as sarcopenic according to these markers were found to have significantly poorer overall survival outcomes.
In the literature, the most commonly used threshold values for SMI Type 1 are those established by Martin et al.(For men: SMI <43 cm
2/m
2 for BMI <25 kg/m
2 and SMI <53 cm
2/m
2 for BMI ≥ 25 kg/m
2, women: SMI <41 cm
2/m
2), Prado et al. (For men: SMI ≤52.4 cm
2/m
2 and women SMI ≤38.5 cm
2/m
2), and the Lancet Oncology Consensus (For men: SMI ≤55 cm
2/m
2 and women SMI ≤39 cm
2/m
2). Martin et al. employed a formula incorporating BMI for male patients. More recent publications predominantly use thresholds independent of BMI, as defined by Prado et al. or the Lancet Oncology Consensus.
5,11 We believe the relatively lower threshold value for SMI Type 1 identified in our study may be attributable to factors such as ethnicity, dietary habits, and geographical variations.
The five-year overall survival rate for patients undergoing radical cystectomy due to muscle-invasive bladder cancer is approximately 50%.
15 A review of the literature reveals that more than half of the patients had tumors staged at pT2 or lower.
15,16 The VESPER randomized controlled trial, one of the more recent studies, reported a 5-year overall survival of approximately 60% in patients who received neoadjuvant chemotherapy before cystectomy.
17 In our study, 59% of patients had tumors staged at pT3 or higher, and the rate of adjuvant chemotherapy was lower than this proportion (44.8%). This likely explains, at least in part, the lower overall survival rates observed in our study compared to the literature.
Several studies have demonstrated the prognostic value of sarcopenia in cancer patients.
12,18,19 Consistent with the literature, the overall survival rates of patients classified as sarcopenic or non-sarcopenic based on the thresholds determined for SMI Type 1, PMI Type 2 in males, and SMD in females were significantly lower for the sarcopenic groups.
4,5,10,12,20The most commonly used threshold values for SMI Type 1 in the literature are those determined by Martin et al.
4. This study demonstrated that SMI Type 1 is an independent prognostic factor for overall survival in patients with gastrointestinal and lung cancers. Psutka et al. were among the first to show that sarcopenic patients, classified by SMI Type 1, undergoing radical cystectomy for bladder cancer had worse overall survival compared to non-sarcopenic patients (39% vs. 70%).
12 Similarly, a study by Mayr et al. demonstrated that sarcopenia defined by SMI Type 1 is an independent risk factor for overall survival in patients undergoing radical cystectomy for bladder cancer (HR 1.43; 95% CI: 1.09–1.87; p = 0.01).
21 In our study, sarcopenia classified as SMI Type 1 was demonstrated to be a strong independent predictor of overall survival in male patients. Sarcopenic patients exhibited an approximately 2.5-fold lower overall survival rate, with a 24-month cumulative overall survival rate of 33.2%.
Unlike prior studies, our study was the first to evaluate sarcopenia using SMI Type 2 (total muscle area at the L3 vertebra divided by L3 vertebral cross-sectional area). Male patients were classified as sarcopenic or non-sarcopenic based on SMI Type 2. Our analysis revealed that SMI Type 2 is a strong independent prognostic factor for overall survival in male patients. Sarcopenic patients demonstrated an approximately 3-fold lower overall survival rate, with a 24-month cumulative overall survival rate of 42%.
Another parameter for sarcopenia based on muscle area is PMI. Various methods and thresholds for PMI have been reported in the literature.
10,18,22–24 In our study, appropriate thresholds for PMI Type 1 could not be determined for either gender. However, using the novel PMI Type 2 defined in our study, we established a threshold for male patients and classified them as sarcopenic or non-sarcopenic. Both univariate and multivariate regression analyses demonstrated that PMI Type 2 is a strong independent prognostic factor for overall survival in male patients. Sarcopenic patients exhibited an approximately 1.75-fold lower overall survival rate, with a 24-month cumulative overall survival rate of 43.9%. In particular, some systematic reviews have shown that the L3 or L4 vertebrae are the sections that best reflect muscle density in studies on sarcopenia.
25,26 In our study, we emphasized the prognostic value of SMI 2 and PMI 2, obtained from the L3 VCA-based psoas muscle and general cross-sectional muscle assessments in patients undergoing radical cystectomy for bladder cancer.
Another prognostic factor in cancer patients is SMD, which reflects the fat infiltration within muscle tissue. Lower SMD values indicate greater fat content and reduced muscle quality. Sarcopenia defined by SMD is also referred to as myosteatosis in the literature. Few studies have evaluated myosteatosis as a prognostic factor in bladder cancer. Yamashita et al. assessed 123 patients undergoing radical cystectomy for bladder cancer and reported that myosteatosis was an independent prognostic factor for cancer-specific survival (HR 3.53; 95% CI: 1.30–12.50; p = 0.04) but not for overall survival (p = 0.10).
20 Engelmann et al., in a recent study involving 657 patients undergoing radical cystectomy, demonstrated that low SMD values were significantly associated with decreased overall survival.
14 Our study showed that female patients classified by SMD had lower overall survival in the sarcopenic group compared to the non-sarcopenic group. SMD was identified as a strong independent prognostic factor for overall survival in women in the multivariate Cox regression model (HR 3.68; 95% CI: 1.24–10.89) (
Table 3). The 24-month cumulative overall survival rates for sarcopenic and non-sarcopenic groups were 21.4% and 58.7%, respectively (p = 0.014). Evaluating the above studies emphasizing myosteatosis as an important prognostic factor for bladder cancer, it is clear that male and female patients were seperately analyzed in these papers. It should be noted that male and female have different muscle masses in both quantity and structure. In our study, we also demonstrated a significant difference in muscle mass between the sexes. We speculate that one underlying mechanism may be hormonal, and that estrogen may also be associated with myosteatosis. Therefore, unlike in men, we believe that a different sarcopenic prognostic marker (SMD) may have been identified in female cohort, due to the fat density in their musculature, regardless of the small number of patients.
An additional significant finding was the evaluation of sarcopenia parameters based on localized versus locally advanced disease. Fukushima et al. demonstrated that sarcopenia is an independent risk factor for shorter overall survival in patients with advanced urothelial carcinoma (11 vs. 31 months).
27 Kasahara et al. evaluated 27 patients with advanced bladder cancer treated with gemcitabine and nedaplatin, reporting mean overall survival of 223 days in the sarcopenic group compared to 561 days in the non-sarcopenic group.
22 In our study, SMI Type 1, SMI Type 2, and PMI Type 2 were significant predictors of overall survival in males with locally advanced disease, while SMD was significant in females. In localized disease, SMI Type 1 and SMI Type 2 remained as significant predictors in males, but PMI Type 2 and SMD were not in both male and female patients.