Introduction
The emergence of precision or personalized medicine, which seeks to improve diagnosis, prediction, prevention, and treatment by focusing on the individual rather than on groups of people, has instigated a need for patient-specific multidimensional data to evaluate an individual’s health, well-being, and environment. Diabetes mellitus is one example of a disease for which precision medicine approaches may be beneficial due to the recognized heterogeneity in its etiology, clinical presentation, and pathogenesis.
1 The resulting need for patient-specific multidimensional data collection (precision monitoring) has been highlighted in the latest American Diabetes Association and European Association for the Study of Diabetes consensus, along with other publications, that advocated for precision monitoring methods as the next crucial step toward precision medicine for diabetes.
1,2 Precision medicine in the field of diabetes research focuses on elucidating pathophysiological processes (molecular and environmental), and consequently the heterogeneity in glycemic dynamics, that underlie the disease. Although the primary cause of dysregulated glycemic dynamics concerns defects in at least one physiological pathway, multiple coherent factors including self-regulated behavior (e.g., dietary intake and physical activity) and health (e.g., medication and psychological status) also influence glycemic dynamics. Furthermore, lifestyle, defined by the long-term combination of these variables, impacts the risk for future complications.
3–7To capture glycemic variations, gain insight into the underlying glycemic effects of lifestyle, and ultimately monitor disease progression, all at an individual level, the availability of data is imperative. Although data measured in clinical and research settings are well controlled, detailed data collection over weeks or months of time is unfeasible from these settings and difficult to scale to large cohorts. Consequently, a transition must be made toward data measured in the comfort of people’s homes and throughout their daily lives. Furthermore, data from free-living conditions are representative of self-regulated behavior and habits, thereby offering the opportunity to capture the day-to-day glycemic variability typical for diabetes. The shift from clinical settings toward the real-world is enabled by the advent and enhancement of wearable devices. The continuous glucose monitor (CGM) has already provided tremendous insight into glycemic control for clinicians and people with diabetes.
8 Furthermore, smartphones and smartwatches have been shown to be promising for the monitoring of lifestyle variables.
9,10The use of wearable devices in diabetic populations for data collection in free-living conditions has, except for a few studies, been mostly limited to the effects on blood glucose levels of physical activity, diet, or psychological stress alone. Of the few studies that simultaneously collected data on a broader range of variables, nearly all of these studies have been conducted on individuals with type 1 diabetes. Sample sizes in these studies ranged from 9 to 497 participants, with study periods of up to 8 weeks, and measurements on blood glucose levels, exercise, insulin dosing, dietary intake, heart rate, psychological status, electrocardiograms, breathing, and accelerometers.
11–14 Similar studies in people with type 2 diabetes are even rarer, with two studies (94 and 14 subjects with non-insulin-treated type 2 diabetes) collecting data on blood glucose levels, dietary intake, heart rate, and accelerometer-derived step count.
15,16Data collection in free-living conditions can be burdensome for the participants and researchers, especially when using analog data collection methods (i.e., paper logbooks and questionnaires). The data collection process often requires data digitization, structuring, and manual preprocessing. Some studies also relied on participants or experts to acquire information on dietary macronutrient composition.
12,13 By integrating multiple wearable devices and applications, some of the burdens of data collection in the real world can be alleviated. The use of wearable devices negates the need for data inference and digitization. Moreover, the use of specifically consumer-grade wearable devices, rather than research-dedicated devices, offers a cost-effective, scalable, and readily available method of collecting data due to the widespread ownership of smartphones and the increasing popularity of smartwatches and fitness trackers. The consumer-grade wearable devices, used in this study, included a CGM for continuous blood glucose measurements, a smartphone application to collect patient-reported dietary intake and physical activity, and a smartwatch with in-house application to measure heart rate, acceleration, and step counts and collect participant-reported mood and insulin dosing.
This article describes the use of commercially available, consumer-grade wearable devices and applications in the DiaGame Study to collect data under real-world conditions in people with diabetes, predominantly type 2 diabetes. We provide an overview of the types, quantity, and quality of data collected. We subsequently investigate whether dietary intake, insulin dosing, physical activity, and mood (from here onwards referred to as lifestyle factors) are associated with glycemic excursions (defined by peaks in the CGM signal) in people with type 1 diabetes, insulin-naïve type 2 diabetes, and insulin-dependent type 2 diabetes.
Methods
Study design and participants
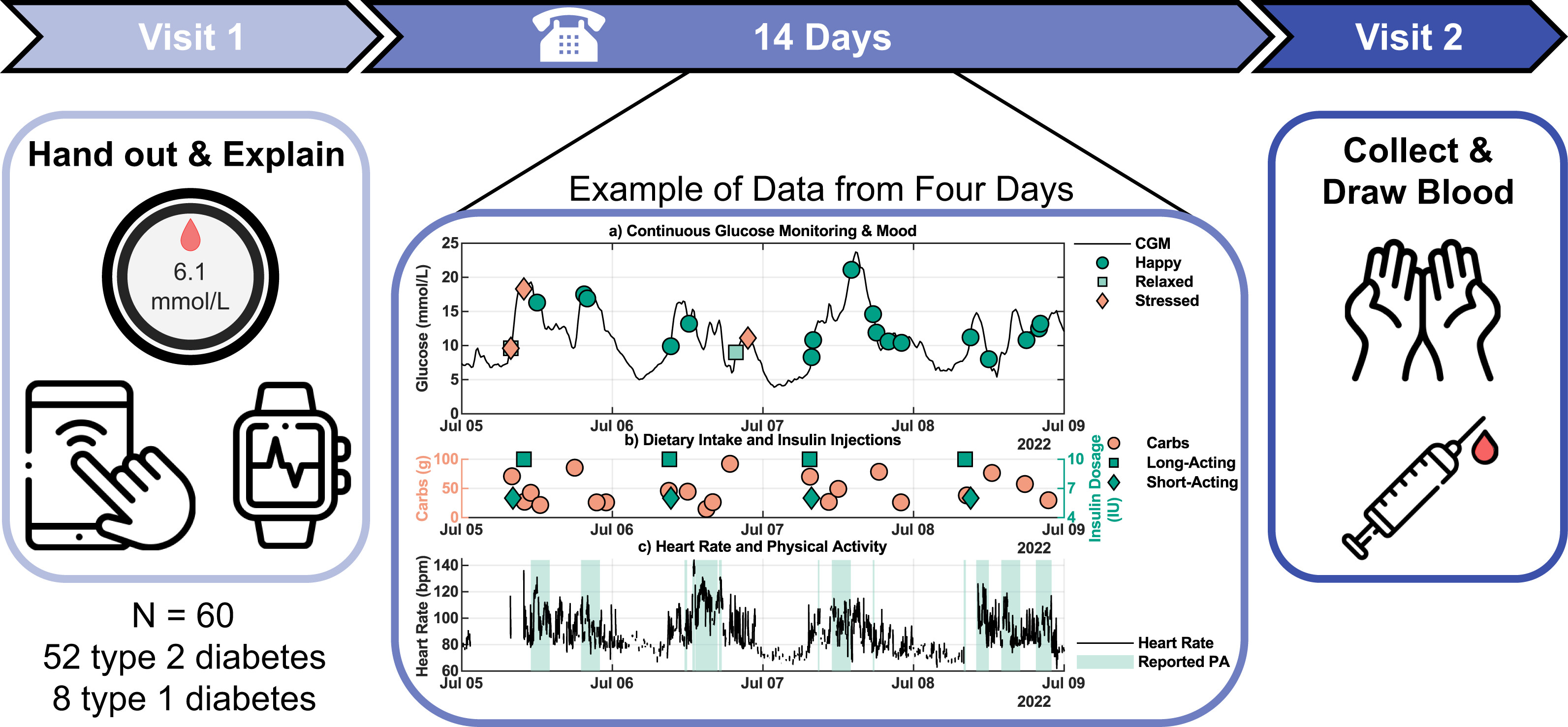
Between June 2022 and June 2023, 60 participants (type 1 and type 2 diabetes) were included, and data were collected over a consecutive 14-day period. All participants were recruited from the outpatient clinic of the Máxima Medical Center (The Netherlands). Participants were eligible for inclusion when they met the following criteria: (I) diagnosis of type 2 diabetes or type 1 diabetes (body mass index [BMI] <30 kg/m2 for type 1 diabetes), (II) age >18 years, and (III) possession of a smartphone that runs the required application. Exclusion criteria comprised (I) pregnancy or breastfeeding, (II) ongoing treatment for malignancy, (III) scheduled Magnetic Resonance Imaging (MRI) scan during the study, and (IV) non-Dutch speaking. All individuals provided written informed consent. The study was registered in the Dutch Trial Registry (NL9290), approved by the Medical Research Ethics Committee Máxima MC (nr. L20.102), and conducted according to the principles of the Declaration of Helsinki.
Participation consisted of two visits to the clinic and a telephone call. During the first visit, a brief overview of the participant’s medical history was registered, along with medication use. Subsequently, blood pressure, resting heart rate, weight, and length were measured, and a blinded CGM was applied. Finally, participants received a smartwatch with preinstalled study application and were instructed on how to download the smartphone application and use the devices.
Four to six days after the first visit, participants were contacted by telephone to address specific issues identified through a remote assessment of data reported on dietary intake and physical activity. Instructions were reiterated if necessary. Subsequently, after 14 days, participants returned for the second visit in a fasted state to draw blood and return all devices. The data on these devices were subsequently pseudonymized and exported to a secure database.
Data collection
Glucose concentrations were measured using a FreeStyle Libre Pro iQ sensor (Abbott Diabetes Care, Illinois, USA), a blinded CGM that measured interstitial glucose levels every 15 min, 24 hours per day, for 14 consecutive days. Glycated hemoglobin (HbA1c), fasting glucose, and C-peptide concentrations were measured using the standard protocol of the hospital’s clinical chemical laboratory.
Participants received a
Samsung Galaxy Watch 2 Active 44 mm smartwatch during visit 1, preinstalled with the developed application (built for the Tizen mobile operating system). The application allowed participants to report mood and insulin dosing and automatically recorded measurements from the internal sensors (heart rate monitor, pedometer, and triaxial accelerometer). First, (insulin-dependent) participants were instructed to self-report the type of insulin injected (short-acting, long-acting, or mixed insulin), dose, and time of injection through the dedicated screen. Second, participants were nudged four times a day at fixed times (8 AM, 12 PM, 6 PM, 8 PM) through short vibrations and activation of the smartwatch showcasing the dedicated mood screen. The screen provided the option of five moods with matching emojis: happy, relaxed, stressed, angry, and sad. Participants were instructed to respond to these nudges and to report their mood when they perceived a change in their mood. Nonetheless, participants retained autonomy to report mood at any time via the application. An overview of the smartwatch application can be seen in
Supplementary Fig. S1.
Dietary intake, both food and beverage consumption (excluding water, tea, and coffee without any additives), and physical activity were self-reported every day through the
Mijn Eetmeter smartphone application (translates to “
My Eating Meter”) on the participant’s smartphone (Voedingscentrum, The Hague, The Netherlands).
17 Reporting dietary intake required participants to enter the meal moment (breakfast, lunch, dinner, and snacks in between these meal moments), meal contents, and amounts of these meal contents. Options for meal contents are synchronized with the Dutch Food Composition Information Portal (NVIP) from the Dutch National Institute for Public Health and the Environment.
18 The application often provides predefined portion sizes per product (e.g., one standard serving of a product or the distinction between a small, medium, or large portion with an indicated weight) next to the option to report the weight of the product. Instructions were given to report mealtimes in an adjacent text field that listed the different meal moments. Furthermore, participants were instructed to report physical activity in the application. This action consisted of selecting the type of activity from a wide range of possibilities (from walking to cleaning the house), the duration of the activity, and noting the starting time in a text field. For an impression of the application, see
Supplementary Fig. S2.
Data security
Participants were provided with preset study Mijn Eetmeter accounts to promote data security and privacy, aligning with the General Data Protection Regulation. In addition, the smartwatch and CGM operated offline during the study period.
Statistical analysis
The association between glycemic excursions and lifestyle variables was evaluated by determining whether peaks in the CGM signal were preceded, within a 2-hour window, by a logged meal, insulin dose, physical activity, or mood. CGM data were first smoothed using a third-order Savitzky-Golay filter (frame length of 7), and peaks were identified using MATLAB R2024a’s
findpeaks algorithm.
19 Days with reported caloric intake below 1000 kcal were excluded. To assess whether the observed associations were greater than expected by chance, a control window from 2.5 to 4.5 hours prior to each peak (nonoverlapping with any test window) was defined. For each lifestyle variable, the proportion of test versus control windows containing a respective log entry was compared using a nonparametric one-sided Wilcoxon rank-sum test. Bonferroni correction was applied to adjust for multiple comparisons (
α = 0.05). Furthermore, associations between lifestyle factors and CGM peaks were quantified using mixed-effects logistic regression models. Odds ratios (ORs) for each lifestyle factor were estimated by including all lifestyle variables simultaneously as fixed effects, while accounting for repeated measurements and time-invariant confounders within individuals via participant-level random intercepts. Both analyses were stratified by diabetes type and insulin therapy: type 1 diabetes, insulin-naïve type 2 diabetes, and insulin-dependent type 2 diabetes to account for differences in insulin therapy.
The wearing of the smartwatch was determined by the presence of a valid (positive value) reading of the heart rate sensor within 1 min of nudging. Response percentage was defined as the percentage of responses to a nudge within 5 min of the nudge, given the smartwatch was worn according to the previous heart rate criterion. 95% binomial proportion confidence intervals (CIs) were calculated using Clopper–Pearson “exact” CIs, unless stated otherwise.
Discussion
The DiaGame study extends prior work on glucose and lifestyle monitoring by demonstrating that consumer-grade wearable devices and applications, supported by a nudging mechanism, can be used to capture multimodal real-world data on glycemic dynamics alongside lifestyle factors. By integrating these diverse data streams, we illustrate how these everyday behaviors relate to spikes in blood glucose levels in people with type 1 diabetes, insulin-naïve type 2 diabetes, and insulin-dependent type 2 diabetes.
Our study showcased the opportunity to harness wearable devices as a feasible method of multimodal longitudinal data collection under real-world conditions in individuals. These devices required minimal calibration steps, could be operated by participants following a short instruction session, and no major problems were identified by the research team (except for two CGM sensor failures) or reported by participants. Moreover, the interaction of the participants with the applications was effective, reflected by the high percentage of participants providing self-reported information. In addition, the use of a blinded CGM negated the need to scan every 8 hours, potentially leading to loss of data and avoiding influencing the participant’s decision-making. The commercial availability of these consumer-grade devices and public access to the applications allow for time-effectiveness and easy scalability to large cohorts. Thereby, providing the means to obtain continuous insight into lifestyle and personalized glycemic dynamics to the public and, optionally, their health care providers.
The combination of a CGM, a smartwatch, and a smartphone application provided insight into the daily life of an individual, from the comfort of their home, of which limited information was otherwise available. However, data collected under free-living conditions are usually not a comprehensive description of all lifestyle events. Evaluation of data quality indeed indicated that participant-reported data did not always compare with expectations, with heterogeneity in the magnitude of these differences between participants. First, the energy intake reported through the smartphone application was, on average, approximately 450 kcal/day lower than expected when compared with the European reference values for moderately active women and men aged 60–69 years.
20 This discrepancy may reflect known challenges in dietary self-reporting, including conscious or unconscious underreporting, particularly in studies focusing on lifestyle behavior. Importantly, however, the mean reported energy intake in the present cohort was comparable with that reported in a younger population without diabetes using the same smartphone application (1830 kcal/day, SD 485 kcal/day,
n = 100), in which the application was shown to provide estimates comparable with 24-hour dietary recalls. Another study that relied on picture-based food annotations by a dietitian reported comparable daily energy intakes (1672 kcal/day, SD 1038 kcal/day,
n = 8).
13,21 In addition, mean daily carbohydrate intake was consistent with participant-reported values in people with type 1 diabetes.
12 Collectively, these comparisons suggest that, despite lower-than-expected absolute energy intake relative to dietary guidelines, the reported intake is in line with values obtained using other approaches in free-living settings. Furthermore, all participants reported dietary intake for at least 11 days, and participant age did not appear to be a limiting factor in reporting dietary intake using a smartphone application, even in this generally older cohort, typical of type 2 diabetes. Thus, even though energy intake may deliberately be reduced or underreported, partially accounting for the discrepancy, accurate dietary intake assessment in free-living conditions remains challenging.
Second, the smartwatch offered an easily accessible means for insulin usage registration, which needed no connection to the internet or a smartphone and was always at hand for participants. These reported insulin doses were accurate on most of the study days, when they could be compared with prescribed insulin treatments. These comparisons demonstrated that only 5 out of 36 long-acting insulin users, and no short-acting insulin users, failed to report at least 1 day for which the accumulated daily insulin dosages were comparable with their health records. However, the accumulated daily insulin dosages derived from insulin reports were frequently lower than prescribed (especially evident when one dose a day was prescribed, and none were reported). It is unclear why insulin administration was underreported. While daily dose adjustments are inherently part of the short-acting insulin treatment, the quality of reported data on insulin use may be affected by the otherwise repeated nature of daily insulin dosing, leading to a lack of urgency to consistently report these events. It has already been shown that data on insulin dosing can be effectively collected from insulin pumps,
22 which are predominantly used by people with type 1 diabetes, whereas the recent development of smart insulin pens could potentially alleviate the burden of insulin dose registration altogether.
Third, ecological momentary assessment of mood using the smartwatch, supplemented with a nudging mechanism four times a day, was an effective method to collect information on the perceived mood of participants. Participant involvement was consistent throughout the study period with high wearing percentages (except in the early morning when several participants may not yet have woken up) and overall steady response percentages. Participants for whom the nudging mechanism was less effective still provided ample mood reports without this nudging encouragement. Nonetheless, participants predominantly reported non-negative (happy or relaxed) moods, which could be valid but could also indicate participants were biased to report socially desirable behavior by using the nudging system. In addition to providing a means to report mood and insulin doses, the smartwatch enabled the measurement of heart rate, acceleration, and step count to independently inform on physical activity (next to participant-reported physical activity, during which heart rate was shown to be elevated). These measurements may supplement registration of physical activity, since the number of days with self-reported physical activity was inconsistent across individuals. Lastly, the mean daily value of none of the variables reported by the participants decreased considerably throughout the study period, which, if this had happened, could have suggested that the data collection protocol may have placed too much of a burden on the participants. Thus, while the collected data may not be a complete description of all daily events, these wearable devices can effectively be used to acquire multimodal real-world data. Hereby answering the call for precision monitoring methods as the next step toward precision prevention, diagnostics, and prognostics.
1,2The acquired data can subsequently be harnessed to assess the association of lifestyle variables with glycemic dynamics, such as peaks in the CGM signal, at an individual level. We have been able to show that peaks in the CGM signal are independently associated with reported meal intake and insulin dosing, demonstrated by higher within-individual proportions of these lifestyle events prior to peaks and by higher ORs for glucose peaks at a subgroup-level, accounting for repeated measurements and time-invariant participant characteristics. The observed association between glycemic excursions and dietary intake is in line with previous research reporting a similar association between dietary intake and CGM peaks in adults without diabetes.
23 Negative mood and physical activity, which have not previously been reported to result in acute glycemic excursions (except for high-intensity exercise), were not associated with peaks in the CGM signal at an individual or subgroup level. Importantly, we cannot exclude that the demonstrated underreporting of data could have affected the estimated associations. However, unless underreporting systematically differs between peak and control windows, this misclassification is expected to bias effect estimates toward the null hypothesis rather than generate spurious associations. Therefore, the reported associations are likely conservative estimates of the true underlying relationships. Furthermore, the within-individual study design mitigates between-person reporting differences, as each participant serves as their own control. Future studies incorporating more structured and comprehensive assessments of lifestyle behaviors may further clarify how these factors shape glycemic dynamics at the individual level.
We acknowledge that the study cohort consisting of known patients does not provide a complete cross-sectional overview of the diabetic population in the Netherlands due to selection from the region and restriction to Dutch-speaking individuals. Nonetheless, the study cohort is typical for a type 1 and 2 diabetes cohort when compared with other studies in terms of patient characteristics. Inclusion may be biased for participants with at least some affinity for digital devices, as those unfamiliar or hesitant to use wearable devices may be more likely to refuse participation. These latter people may need additional support to promote adequate use of these wearable devices.
Multiple enhancements could further enrich the collected data through the inclusion of standardized at-home meals and meal tolerance tests,
23 while automated data capturing systems (e.g., insulin pumps and smart insulin pens to register insulin dosing) could potentially improve the quality of the data and reduce the burden on participants. Furthermore, the registration of medication usage can be extended from the use of solely exogenous insulin to oral medications (e.g., metformin and GLP-1 agonists). Adherence to these medications was currently assumed to be consistent throughout the study period, even though in practice adherence has been shown to be inconsistent.
24 In addition, real-time communication between devices can enable personalized nudging mechanisms, by incorporating information from all devices, for Just-In-Time Adaptive Interventions.
25 Tailoring the timing, frequency, and format of the nudging to the individual may improve effectiveness, while just-in-time nudging could be used to promote reports on dietary intake or insulin injections when blood glucose excursions are observed (currently the nudging mechanism was limited to data collection on mood due to the inherent continuity of human mood, which is lacking in all other variables). Overall, the study protocol, with or without these enhancements, can be used in future studies with prolonged study periods to elucidate long-term behavioral patterns and seasonal variability.
The data collected in real-world conditions hold the potential for various clinical purposes, as indicated by the growing interest and recent increase in research within this field. First, it offers a deeper understanding of intra-day and inter-day glycemic variability at an individual level. This understanding enables health care providers to deliver targeted and personalized care (precision medicine), which may contribute to improved health outcomes (e.g., to deliver app-based personalized lifestyle recommendations
15). Second and more importantly, the collected data offer valuable insights when analyzed across broader groups. It can reveal interindividual variations, facilitating the identification of distinct subgroups within the diabetes population and unveiling their distinct characteristics and needs (e.g., to stratify meal events for digital phenotyping
16). Lastly, these data can support the development of next-generation simulators and digital twins, personalized dynamic metabolic models capable of predicting glucose levels and their modulation due to daily activities.
26 Such models have the potential to serve multiple purposes, from enhancing broader group-level education by showcasing the response of blood glucose levels to lifestyle decisions to in silico experiments with virtual patient cohorts. A deeper understanding of a disease like diabetes and the various factors influencing blood glucose levels can foster increased self-confidence and effective self-management in people with diabetes. Given that improved self-management in diabetes correlates with better health and reduced short- and long-term complications, the significance of this understanding is clear.
27 Furthermore, diabetes self-management education and support currently occur predominantly through one-on-one consultations with health care providers. However, with the escalating numbers of patients with diabetes globally, there is a growing need for more scalable, sustainable, and affordable support. Electronic resources and digital interventions, such as advanced simulators predicting glucose levels at a personal level, could offer a crucial addition to future health care. They hold the potential to make health care more efficient and accessible while simultaneously improving its quality.
Authors’ Contributions
R.d.V. was responsible for conceptualization, methodology, software, validation, formal analysis, investigation, resources, data curation, visualization, and writing the first draft of the article. E.F.H.R. was responsible for conceptualization, methodology, validation, investigation, resources, and writing the first draft of the article. D.F.D.C. worked on conceptualization, methodology, software, validation, investigation, resources, and data curation. E.M.W.L.C.-V.d.M. was responsible for conceptualization, methodology, validation, investigation, and resources. P.M.E.v.G., P.C.M.W.-v.P., U.K., P.A.J.H., N.A.W.v.R., and H.R.H. contributed to conceptualization, methodology, and reviewing of the article. In addition, N.A.W.v.R. and H.R.H. provided supervision and acquired funding, while N.A.W.v.R. was also responsible for project administration. All authors approved the definitive version of the article.