Submission guidelines

Instructions for Authors
- Scope and editorial policy................................................................................. 3
- Copyright considerations........................................................................... 3
- Ethical considerations................................................................................ 4
- Authorship and artificial intelligence.......................................................... 4
- Preprints.................................................................................................. 6
- Fees and open-access policy.............................................................................. 6
- Review and acceptance criteria.......................................................................... 7
- Novelty of contents and impact/usefulness to veterinary laboratory diagnosticians........................................................................................... 7
- Adequacy of experimental design, test validation, and statistical analysis....... 8
- Adequacy of title, references, figures, and tables........................................ 10
- Manuscript preparation.................................................................................... 10
- General format and style.......................................................................... 10
- Layout and media............................................................................ 10
- Language and style.......................................................................... 11
- Units of measurement...................................................................... 12
- Tumor pathology............................................................................. 12
- Immunohistochemistry..................................................................... 12
- Sources and manufacturers............................................................... 13
- Detailed manuscript layout....................................................................... 14
- Title page........................................................................................ 14
- Abstract.......................................................................................... 14
- Keywords....................................................................................... 14
- Manuscript sections......................................................................... 15
- Figures.......................................................................................................... 17
- Supplemental material..................................................................................... 24
- New manuscript submission............................................................................ 25
- Revised manuscript submission........................................................................ 26
- Page proofs.................................................................................................... 26
- Help............................................................................................................ 26
- General format and style.......................................................................... 10
APPENDIX A: PCR guidelines—suggested dossier or publication information.......... 27
APPENDIX C: Abbreviations that may be used without expansion........................... 31
Manuscript submission checklist
- Please note the page charge of $75 for each printed page published in
- Please take the time to read our Best Practices for submitting, reviewing, and publishing in JVDI. All manuscripts should adhere to Sage’s accessibility guidelines.
- Microsoft Word file, double-spaced, 12-point Times New Roman font, left-aligned (not justified), 25 mm (1 in.) margin on all sides, pages numbered at the bottom center (i.e., Page X of Y).
- For styles, do not use Heading 1, Use the Normal style setting.
- Number text lines consecutively throughout the manuscript; begin page 1 with line 1; do not restart numbering on each subsequent page.
- Indent paragraphs; do not include spaces between
- Allow 1 space (not 2) after a word or
- JVDI number style is one, 2, 3, 4… within the text, but 1, 2, 3, 4… when in a series in the same sentence.
- Include tables in the main document, but do not embed
- SI units of measurement (International System of Units) must be used (may include conventional units in brackets; see section 4.1.3).
- For anatomic terms, use the English equivalents of terms in Nomina Anatomica Veterinaria. Names of infectious agents should follow the current published standards for viruses (ICTV, International Committee on Taxonomy of Viruses), bacteria (NCBI), and fungi (NCBI).
- Cite the brand and manufacturer in parentheses at the appropriate location within the Do not include address.
- Reference citations in text are listed as superscripts after the punctuation, as 1,2-4,8
- Arrange references alphabetically, numbered See section 4.2.4, References.
- Submit figures in .tiff or .jpg formats only, preferably in color, if applicable (see section 5). Do not exceed the maximum file size of 5 MB per figure.
- Submit supplemental tables as Microsoft Word files (see section 6). Submit supplemental figures following the figure guidelines (see section 5).
1 Scope and editorial policy
The Journal of Veterinary Diagnostic Investigation is the official journal of the American Association of Veterinary Laboratory Diagnosticians. The mission of the Journal is to educate by informing readers of progress in veterinary laboratory medicine and related fields of endeavor. The key objectives of the JVDI are to promote the science of veterinary laboratory medicine and the betterment of animal and public health. JVDI fully supports diversity, equity, and inclusion in our publishing activities.
JVDI is devoted to all aspects of veterinary laboratory medicine. The major disciplines are anatomic pathology, bacteriology/mycology, clinical pathology, epidemiology, immunology, laboratory information management, molecular biology, parasitology, public health, toxicology, and virology. For more information about JVDI, please visit /home/vdi
Three manuscript formats are accepted for review: Reviews, Full Scientific Reports, and Brief Reports (see section 4.2.4). Review articles are strongly encouraged provided they cover subjects of current and broad interest to veterinary laboratory diagnosticians. The suitability of Letters to the Editor, Book Reviews, and Commentaries is determined by the Editor-in-Chief, and a pre-submission inquiry to the editor is recommended.
JVDI content is open-access after a 12-month embargo for non-subscribers and non-AAVLD members. NIH- and government-funded research is not included in the embargo. Our time-to-first decision (reject, revise, accept) averages 20 days.
Accessibility guidelines. Making your article accessible helps more people benefit from your work. It’s also increasingly a legal requirement for content to meet recognized accessibility standards. For more information, see Sage’s accessibility page.
1.1 Copyright considerations
JVDI accepts original manuscripts for consideration with the understanding that the same material or a substantial part thereof is not presently being considered for publication or has not been published elsewhere. The Corresponding Author must secure the approval of all authors and institution(s) where the work was carried out. A statement to the Editor confirming that such approval has been received must be included in the submission cover letter.
Please note that you must sign a license for us to publish your article. Upon acceptance, authors will receive an email from Sage Journals: Licensing & Payment ([email protected]) with a link to sign an appropriate license.
- If you require Open Access, you can select this as an option before you sign the Contributor
- If you are eligible for Open Access via an agreement that Sage has with your institution, Sage will identify you automatically, and you will be asked to sign an Open Access Creative Commons license. The email will come from [email protected].
If your manuscript incorporates any previously copyrighted material that is not in the public domain, you must obtain written reprint permission from the copyright owner and submit a scanned PDF of the permission (as a Supplemental File) along with your manuscript files via our online manuscript submission portal (see section 8). No manuscript containing previously copyrighted material will be accepted for review in JVDI without submission of satisfactory proof that copyright permission has been obtained.
1.2 Ethical considerations
1.2.1 Animal welfare
JVDI requires that authors obtain the relevant national/state/institutional approval prior to animal experimentation. In the United States, this means the Institutional Animal Care and Use Committee for approval for any animal experiment.
Authors are encouraged to register their clinical trials at https://www.clinicaltrials.gov/ or other suitable databases identified by the International Committee of Medical Journal Editors.
1.2.2 Plagiarism
JVDI employs the software program iThenticate to detect plagiarism. From the U.S. Office of Research Integrity, plagiarism of text is “copying a portion of text from another source without giving credit to its author and without enclosing the borrowed text in quotation marks” and plagiarism of ideas is “appropriating someone else’s idea (e.g., an explanation, a theory, a conclusion, a hypothesis, a metaphor) in whole or in part, or with superficial modifications without giving credit to its originator.” Detection of plagiarized material in a manuscript will result in its immediate rejection, regardless of its scientific merit. The author’s institution may be notified.
1.3 Authorship
All authors must be entered into SageTrack during the submission process so that they receive all correspondence during the peer-review process. In addition, the JVDI Authorship Form, which is available as a downloadable PDF, must be completed and submitted during the submission process. The form is located in step 5 of the online submission process. We also encourage all authors to have an ORCID iD (see section 7).
Manuscripts should only be submitted for consideration once consent is given by all contributing authors. Submitting authors should carefully check that all those whose work contributed to the paper are acknowledged as contributing authors. The list of authors should include all those who can legitimately claim authorship. These are all those who:
- Made a substantial contribution to the concept or design of the work; or acquisition, analysis, or interpretation of data.
- Drafted the article or revised it critically for important intellectual
- Approved the version to be
- Agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Authors should meet the conditions of all of the points above. Each author should have participated sufficiently in the work to take public responsibility for appropriate portions of the content.
When a large, multicenter group has conducted the work, the group should identify the individuals who accept direct responsibility for the manuscript. These individuals should fully meet the criteria for authorship.
Acquisition of funding, collection of data, or general supervision of the research group alone does not constitute authorship, although all contributors who do not meet the criteria for authorship should be listed in the Acknowledgments section.
For more information, see the International Committee of Medical Journal Editors on the roles and responsibilities of authors and contributors.
Artificial intelligence
“The use of artificial intelligence (AI) tools such as ChatGPT or Large Language Models in research publications is expanding rapidly. COPE joins organisations, such as WAME and the JAMA Network among others, to state that AI tools cannot be listed as an author of a paper. AI tools cannot meet the requirements for authorship as they cannot take responsibility for the submitted work. As non-legal entities, they cannot assert the presence or absence of conflicts of interest nor manage copyright and license agreements. Authors who use AI tools in the writing of a manuscript, production of images or graphical elements of the paper, or in the collection and analysis of data, must be transparent in disclosing in the Materials and Methods (or similar section) of the paper how the AI tool was used and which tool was used. Authors are fully responsible for the content of their manuscript, even those parts produced by an AI tool, and are thus liable for any breach of publication ethics.” (COPE, 2023 Feb 13)
JVDI has adopted recommendations from WAME (Chatbots, ChatGPT, and Scholarly Manuscripts—WAME Recommendations on ChatGPT and Chatbots in Relation to Scholarly Publications), namely:
- Chatbots cannot be
- Authors should be transparent when chatbots are used and provide information about how they were used.
- Authors are responsible for the work performed by a chatbot in their manuscript (including the accuracy of what is presented, and the absence of plagiarism) and for appropriate attribution of all sources (including for material produced by the chatbot).
Corresponding author
The one individual who takes primary responsibility for communication with the journal during the manuscript submission, peer review, and publication process, and typically ensures that all the journal’s administrative requirements (such as providing details of authorship, ethics committee approval, clinical trial registration documentation, and gathering conflict of interest forms and statements) are properly completed, although these duties may be delegated to one or more co-authors.
The corresponding author is the person who signs the Contributor Form on behalf of all of the authors and whose contact details are included in the article. The corresponding author should be available after publication to respond to critiques of the work and cooperate with any requests from the journal for data or additional information should questions about the paper arise after publication.
Equal credit for authorship.
On the title page, use an asterisk (*) after author names to indicate if authors should receive equal credit—no more than 2 authors per manuscript. Below the corresponding author information, add the following text: *These authors contributed equally to this work.
Changes in authorship.
“Any requests for changes in authorship after initial manuscript submission and before publication should be explained in writing to the editor in a letter signed by all authors, or if sent by email, all authors should be copied (i.e., included as recipients of the email).” (Christiansen S, Flanagin A. What’s new in AMA style? Am Med Writers Assoc Ann Meet; Nov 2019; hereafter AMA Manual of Style, 11th ed.)
Third-party submissions
Where an individual who is not listed as an author submits a manuscript on behalf of the author(s), a statement must be included in the Acknowledgments section of the manuscript and in the accompanying cover letter. The statements must:
- Disclose this type of editorial assistance—including the individual’s name, company, and level of
- Identify any entities that paid for this
- Confirm that the listed authors have authorized the submission of their manuscript via third party and approved any statements or declarations (e.g., conflicting interests, funding, etc.).
Where appropriate, SAGE reserves the right to deny consideration to manuscripts submitted by a third party rather than by the authors themselves.
1.4 Preprints
As part of the submission process, you will be required to warrant that you are submitting your original work, you hold the rights to the work, that you have obtained and can supply all necessary permissions for the reproduction of any copyright works not owned by you, you are submitting the work for first publication in the Journal, and the work is not being considered for publication elsewhere and has not already been published elsewhere. Note that JVDI may accept submissions of papers that have been posted on preprint servers; include the DOI for the preprint in the designated field during the submission process. Authors should not post an updated version of their paper on the preprint server while it is being peer-reviewed. If the article is accepted for publication, the author may re-use their work according to SAGE’s author archiving policy. If your paper is accepted, you must include a link on your preprint to the final version of your paper.
2 Fees and open-access policy
Authors will not be asked for any payment related to their submitted articles until the article is accepted and in production. Please report any suspicious communications you receive to the Managing Editor at [email protected]
Once an article has been accepted and files received into SAGE’s SMART program, authors will receive an email link to the SAGE payment portal within 6 wk.
JVDI page charges. There is a page charge of $75 for each typeset (digital) page published in JVDI. For color figures, authors are given the choice of online-only (free) or print & online ($800 for the first page of color figures, then $200 for each additional page). Black & white images do not incur an additional cost. Payment is expected within 30 days.
Open-access fee. Open-access publication is an additional publication fee to the JVDI page charges. Open-access publishing is offered via the SAGE Choice program. The open-access fee is $3,750.
Payment is expected within 30 days. When publishing your work open access, your institution may be able to help cover the open-access article processing charge (APC) through an Open-Access agreement it has with Sage. See Sage’s full list of agreements here.
Waiver of page charges. JVDI has a limited fund available from the AAVLD Foundation to help researchers from resource-limited countries overcome financial barriers to publication. Authors may apply for a waiver of page charges at the time of submission. Waivers depend on the location of the authors’ institution. First authors with primary affiliations based in countries defined by the World Bank as “Low-Income Economies” (group A) can apply for a 100% page-charge waiver. First authors with primary affiliations based in countries defined by the World Bank as “Lower-Middle-Income Economies” (group B) can apply for a 50% discount on the usual page charge. If accepted for waiver status, the proffered manuscript will proceed through the usual peer-review process. NOTE: At submission, if you are applying for a waiver, please explain in your cover letter how you qualify.
For information on funding body compliance and depositing your article in repositories, please visit SAGE Publishing Policies on the SAGE Journal Author Gateway.
3 Review and acceptance criteria
Based on an assessment by the JVDI editorial office of compliance with JVDI acceptability criteria, manuscripts will be reviewed by 2 or more people selected by the Editors based on their expert knowledge and/or experience in the subject matter. Authors may provide the editorial staff with a list of suggested reviewers for their work and may also request that specific individuals be excluded as reviewers because of potential conflicts of interest. The peer-review process is single-anonymized, that is, editors and reviewers are aware of the authors’ identities, but authors are not informed of the reviewers’ identities. Acceptance of a manuscript for publication is determined by the Editors based on peer review, scientific merit, and value to JVDI readers.
Prior publication of an abstract or poster presented at a conference will generally not impact the manuscript’s eligibility for publication (conference title, date, and location should be added in the Acknowledgments section).
Potential reviewers of all manuscripts submitted to JVDI are asked to consider any potential conflicts of interest they may have before agreeing to review a manuscript. We expect that reviewers with a substantial conflict of interest will disqualify themselves from reviewing a manuscript. More information, as well as general Instructions to Reviewers, is available from SAGE Journal Reviewer Gateway.
Review and acceptability criteria include, but are not limited to, the following: novelty/usefulness and adequacy of experimental design, tables, and figures.
3.1 Novelty of contents and impact/usefulness to veterinary laboratory diagnosticians
3.1.1 Novelty
JVDI is devoted to the publication of original work. Before submitting a manuscript for review, it is the responsibility of all authors to review the literature to ensure that work similar to their own has not been published previously. Authors, please take note of the following examples:
- The development of detection assays (notably PCR procedures) for pathogens that have existing assays already published in the literature: JVDI will consider these papers if it can be demonstrated that the submitted manuscript constitutes a significant improvement over published methodology. Please note that if one or more previously described PCR procedures for any given pathogen exist, subsequent submissions will be acceptable for publication only if the authors demonstrate equivalency (or preferably superiority) via a side-by-side comparison between the existing assay and the new assay. The manuscript must also assess important test-related criteria such as sensitivity, specificity, accuracy, robustness, rapidity, throughput, and cost (see Appendix A).
- The isolation and/or identification of infectious agents from host species that have previously been described in the literature will be considered if the submitted manuscript adds impactful new information, such as a new detection method, novel virulence or pathogenicity data, or unique antimicrobial susceptibility information. In contrast, the detection of a well-known pathogen in a new animal species is not considered sufficiently novel information to warrant publication.
- Single case reports will be considered for publication only if they demonstrate excellence in the diagnostic investigation, including a detailed discussion of the differential diagnosis. Preferred submissions would be novel, emerging, or unique case reports; case series summaries; classic diseases that have significantly evolved or changed in some fashion; or demonstration of the usefulness of new technologies to the diagnostic Case reports combined with a review of the literature on the topic of the case report may increase the chances of having a manuscript accepted for publication.
Although authors may believe that theirs is the first report of a finding, it may have been reported elsewhere in an obscure site, and we prefer to let history decide on primacy. In the Abstract, you might state, for example: ”Disease X has not been reported previously in [species], to our knowledge.” To our knowledge” requires substantiation. Please expand further in the text.
Example.
Information about congenital intestinal anomalies in goats is scant. We retrieved no cases of intestinal vascular malformations in goats after searching Google, PubMed, CAB Direct, Web of Science, and Scopus using the search terms “hemangioma intestine goat,” “hemangiosarcoma intestine goat,” “vascular hamartoma intestine goat,” and “vascular malformation intestine goat.”
3.1.2 Usefulness and impact
The target readership of JVDI is veterinary laboratory diagnosticians. The contents of manuscripts published in JVDI must be applied science and relevant to the professional activities of our core group. Examples of manuscripts that do not fit within the scope of JVDI include the following:
- Clinically oriented manuscripts regarding therapy and clinical diagnostic techniques (e.g., ultrasonography, radiology).
- Basic science manuscripts (e.g., mapping genes of infectious agents without a practical diagnostic application).
3.2 Adequacy of experimental design, test validation, and statistical analysis
The experimental design used must be appropriate and adequate. Similarly, the interpretations and conclusions must be valid and supported by appropriate statistical analysis.
- All tests used are expected to have been verified or validated in-house, as appropriate. Variants of standard tests may be referenced to a previous peer-reviewed report. No reference is needed for standard techniques, such as tissue sectioning and H&E If standard tests are modified, the authors must present comparability data or reference published data that show comparability.
- From the AAVLD Requirements for an Accredited Veterinary Medical Diagnostic Laboratory, v2021-01.
- Validation: The act of confirming, through objective evidence, that the requirements which define an intended use or application have been The process through which a test method is confirmed to be fit for the intended purpose.
- Verification: The process of determining accuracy using a reference standard; g., comparing the accuracy of a piece of equipment to a NIST-calibrated reference standard; the internal process to determine if a laboratory can perform a validated assay and obtain expected results.
- For further information, see: Arnold JE, et al. ASVCP Guidelines: Principles of Quality Assurance and Standards for Veterinary Clinical Pathology (version 0). Vet Clin Pathol 2019;48:542–618.
- If the subject of the article is the validation or modification of a PCR assay, then the following information should be described: PCR primer and probe sequences; analytical sensitivity and specificity performance characteristics; repeatability and reproducibility; and diagnostic sensitivity and specificity (Toohey-Kurth K, et al. Suggested guidelines for validation of real-time PCR assays in veterinary diagnostic laboratories. J Vet Diagn Invest 2020;32:802–814; see Appendix A). Alternatively, the author may follow the most recent revision of the OIE validation algorithm (Principles and methods of validation of diagnostic assays for infectious diseases. In: Manual of Diagnostic Tests and Vaccines for Terrestrial OIE, 2019). Novel molecular assays without a PCR step and non-molecular tests should follow the most recent revision of the OIE validation algorithm.
- For clinical pathology test guidelines, see current ASVCP Quality Assurance and Laboratory Standards Guidelines. Authors should state which guideline they are following.
- The usual level of significance accepted is p ≤ 05. “Fisher suggested a probability of one in twenty (0.05) as a convenient cutoff level to reject the null hypothesis.” “The null hypothesis is the default assumption that nothing happened or changed” (https://en.wikipedia.org/wiki/Statistical_significance). Note that “p values do not give the probability that a null hypothesis is false, they give the probability of obtaining data at least as extreme as those observed if the null hypothesis was true. It is by convention that smaller p values are interpreted as stronger evidence that the null hypothesis is false” (Woolston C. Psychology journal bans p values. Nature 2015;519:9).
- We can still apply tests and report p-values—it is a valid method, just don’t label results as “statistically significant,” but rather report how strong an effect is, and what this means or As summarized in the ASA statement on p-values: “Good statistical practice, as an essential component of good scientific practice, emphasizes principles of good study design and conduct, a variety of numerical and graphical summaries of data, understanding of the phenomenon under study, interpretation of results in context, complete reporting, and proper logical and quantitative understanding of what data summaries mean. No single index should substitute for scientific reasoning” (Wasserstein RL, Lazar NA. The ASA’s statement on p-values: context, process, and purpose. Am Stat 2016:70:129–133).
- “Questionable research practices (QRPs) in the statistical analysis of data and in the presentation of the results in research papers include HARKing, cherry-picking, p-hacking, fishing, and data dredging or HARKing (Hypothesizing After the Results are Known) is the presentation of a post hoc hypothesis as an a priori hypothesis. Cherry-picking is the presentation of favorable evidence with the concealment of unfavorable evidence. P-hacking is the relentless analysis of data with an intent to obtain a statistically significant result, usually to support the researcher’s hypothesis. A fishing expedition is the indiscriminate testing of associations between different combinations of variables not with specific hypotheses in mind but with the hope of finding something that is statistically significant in the data. Data dredging and data mining describe the extensive testing of relationships between a large number of variables for which data are available, usually in a database. This article explains what these QRPs are and why they are QRPs. This knowledge must become widespread so that researchers and readers understand what approaches to statistical analysis and reporting amount to scientific misconduct” (Andrade C. HARKing, cherry-picking, P-hacking, fishing expeditions, and data dredging and mining as questionable research practices. J Clin Psychiatry 2021;82:20f13804).
3.3 Adequacy of title, references, figures, and tables
The title must adequately reflect the contents of the manuscript. References must be as current and complete as possible but avoid the use of multiple references to back up a single fact. Figures and tables must be pertinent to the contents of the manuscript and must not be redundant with information already presented in the text.
4 Manuscript preparation
- Please take the time to read our Best Practices for submitting, reviewing, and publishing in JVDI.
- Microsoft Word file, double-spaced, 12-point Times New Roman font, left-aligned (not justified), 25 mm (1 in.) margin on all sides, pages numbered at the bottom center (i.e., Page X of Y).
- For styles, do not use Heading 1, Use the Normal style setting.
- Number the text lines consecutively throughout the manuscript; begin page 1 with line 1; do not restart numbering on each subsequent page.
- Indent paragraphs and do not include spaces between
- Allow 1 space (not 2) after a word or
- Punctuation: comma or period before superscripts; semicolon after superscripts; no spaces before or between superscripts. We use serial (Oxford) commas.
- Use page breaks (= Ctrl-Enter) rather than a series of hard returns to separate sections of
- No need to capitalize words to abbreviate in capital letters; non-standard abbreviations must be defined on first use—note that the Abstract is independent of the main body of text. See Appendix C.
- Number style is one, 2, 3, 4… within the text, but 1, 2, 3, 4… when in a series in the same
- Number cases starting with case 1 regardless of your particular numbering system and use in all sections of the manuscript (e.g., cases 1–10; cases 2, 3, 5). Laboratory case numbers should not be used in the text, tables, or figures.
- SI units of measurement (International System of Units) must be used (may include conventional units in brackets; see section 4.1.3).
- For anatomic terms, use the English equivalents of terms in Nomina Anatomica Veterinaria. Names of infectious agents should follow the current published standards for viruses (ICTV, International Committee on Taxonomy of Viruses), bacteria (NCBI), and fungi (NCBI).
- Cite the brand and manufacturer in parentheses at the appropriate location within the Do not include the address.
- Reference citations in text are listed as superscripts after the punctuation, as 1,2-4,8
- Arrange references alphabetically, numbered
- Include tables in the main document, but do not embed
- Submit figures as .tiff or .jpg only (see 5). Do not exceed the max. file size of 5 MB per figure.
- Submit supplemental tables in a Microsoft Word file (see section 6). Submit supplemental figures following the figure guidelines (see section 5).
4.1 General format and style
4.1.1 Layout and media
Three manuscript formats are accepted: Reviews, Full Scientific Reports, and Brief Reports (it is strongly encouraged that Brief Reports include a literature review). Supplemental files may be submitted with each of these formats (see sect. 6). Review articles are encouraged provided they cover subjects of current and broad interest to veterinary laboratory diagnosticians. Authors interested in submitting a Review should contact the EIC ([email protected]).
The suitability of Letters to the Editor, Book Reviews, and Commentaries is determined by the EIC, and a pre-submission inquiry to the editor is recommended. Book Reviews should be emailed to Donal O’Toole, the Book Review Editor ([email protected]), and not submitted through the submission site.
4.1.2 Language and style
The American form of English must be used, and manuscripts must be written in a style following the current standards for scientific publications. Editors will reject manuscripts that do not meet a minimum standard for written English. Abbreviations may be used after the first mention with complete spelling; standard abbreviations may be used without definition (see Appendix C). Use Arabic numerals except when a number begins a sentence, in which case spell the number out in full. JVDI number style is one, 2, 3, 4… within the text, but 1, 2, 3, 4… when in a series in the same sentence.
Rather than use the passive voice, use the active voice, personalize, and avoid anthropomorphization. “Studies do not find anything; authors or researchers do. Reports, studies, findings, or tables are not animate entities, and they cannot analyze, show, demonstrate, or reveal results. More correctly, the author(s) took these actions: “We analyzed …”, “We found …”, etc. (Table 1).” (Maxie G. Personalizing science—who did what when? J Vet Diagn Invest 2023;35)
Table 1. Recent examples of statements that were personalized or converted to the active voice.
|
Original |
Edited |
|
The aim of this study was to investigate |
We investigated |
|
In this report, we describe |
We describe here |
|
In this retrospective study, the data were collected |
We collected data |
|
A total of 28 samples were tested |
We tested 28 samples |
|
Known positive and negative samples were used in this study |
We used known positive and negative samples |
|
The sensitivity and specificity of the assay were also evaluated |
We evaluated the sensitivity and specificity of our assay |
|
The developed assay |
Our assay |
|
The study analyzed the data |
We analyzed the data |
|
Table 1 shows that samples were positive. |
Samples were positive (Table 1). |
|
Test results did not show a significant difference (p > 0.05) |
Results were not significantly different (p > 0.05) |
|
The results of this report demonstrated that |
We found |
An editing service, including translation from Spanish, Portuguese, or Chinese, is available from SAGE; use of this service does not guarantee acceptance of the manuscript by the journal.
“Use of the terms first world/third world and developed/developing are not recommended as descriptors when comparing countries or regions. Low-income, limited-income, resource-limited, resource-poor, transitional…” are acceptable (AMA Manual of Style, 11th ed.).
JVDI has accepted the practice of italicizing not only the names of family, genus, and species, but the formal scientific names of all taxa regardless of rank, as is the practice in the International Code of Nomenclature for algae, fungi, and plants (Thines M, et al. Setting scientific names at all taxonomic ranks in italics facilitates their quick recognition in scientific papers. IMA Fungus 2020;11:25).
4.1.3 Units of measurement
SI units of measurement (International System of Units) must be used (may include conventional units in brackets). Express centrifugal speed in relative centrifugal force (× g) and not in revolutions per minute. See Appendix C for abbreviations.
For easy conversions, see AMA Manual of Style: https://academic.oup.com/amamanualofstyle/si-conversion-calculator
For veterinary lab examples, see:
https://www.uoguelph.ca/ahl/content/hematology-reference-intervals
https://www.uoguelph.ca/ahl/biochemistry-reference-intervals
4.1.4 Tumor pathology
JVDI supports the need for standardization of tumor assessment (Meuten DJ, et al. International guidelines for veterinary tumor pathology: a call to action. Vet Pathol 2021;58:766–794; Schulman FY, et al. Reporting guidelines for manuscripts on tumor prognosis. Vet Pathol 2022. doi:10.1177/03009858221082207); checklists are available online in the Supplemental material that accompanies the above-mentioned Meuten article. Submitters are encouraged to follow these guidelines.
Report mitotic activity as mitotic count (MC) and not mitotic index (MI; Meuten DJ, et al. Mitotic count and the field of view area: time to standardize. Vet Pathol 2016;23:1–15). In a cellular region at the periphery of the tumor with the most mitotic activity, count the total number of mitotic figures in 2.37 mm2 (10 contiguous fields, no overlapping, high-power field [hpf] 40× objective, 10× ocular field number [FN] 22 mm, field of view [FOV] diameter 0.55 mm at specimen level), avoiding and/or skipping areas of the tumor that are cell-poor from hemorrhage, edema, necrosis, cysts, etc.
4.1.5 Immunohistochemistry
IHC tests reported must be validated in the subject species. Steps are best presented in table format for the various antibodies used, and include primary antibody (target antigen, host, source, clone, dilution), antigen retrieval method (heat-induced epitope retrieval, protease retrieval, no retrieval, other), chromogen, and detection system used (Caswell JL, et al. Observational study design in veterinary pathology, part 2: methodology. Vet Pathol 2018;55:774–785; Ramos-Vara JA, et al. Suggested guidelines for immunohistochemical techniques in veterinary diagnostic laboratories. J Vet Diagn Invest 2008;20:393–413; Ramos-Vara JA, Miller MA. When tissue antigens and antibodies get along: revisiting the technical aspects of immunohistochemistry—the red, brown, and blue technique. Vet Pathol 2014;51:42–87).
Example 1.
Table 1. Antibodies with source, clone and manufacturer, concentration used, retrieval information, and detection method.
|
Antibody |
Host |
Source |
Clone |
Antigen retrieval |
Dilution |
Chromogen |
Autostainer |
Positive canine control |
|
CD204 |
Mouse |
Medicinal Chemistry Pharmaceutical, KT022 |
SRA-E5 |
PT low |
1:1,000 |
DAB |
Bond-Max; Leica |
Histiocytic proliferation |
|
E-cadherin |
Mouse |
BD Bioscience, 610181 |
36/E-cadherin |
ER1 |
1:300 |
DAB |
Bond-Max; Leica |
Skin |
|
Calretinin |
Mouse |
Dako, M7245 |
Dak-Calret-1 |
ER1 |
1:100 |
DAB |
Bond-Max; Leica |
Eye |
|
Pancytokeratin plus |
Mouse |
BioCare, CM162C |
AE1/AE3 + 5D3 |
ENZ3 |
1:200 |
DAB |
Bond-Max; Leica |
Skin |
|
Cytokeratin 5/6 |
Mouse |
Dako, M7237 |
D5/16 B4 |
PT high |
1:50 |
DAB |
Dako Link 48; Agilent |
Skin |
|
Cytokeratin 7 |
Mouse |
Dako, M7018 |
OV-TL 12/30 |
ER2 |
1:75 |
DAB |
Bond-Max; Leica |
Lung |
Example 2.
Table 1. Details of immunohistochemical stains performed on sections of suspected non-visceral leiomyosarcoma in dogs.
|
Antibody |
Source |
Antigen retrieval |
Dilution |
Secondary polymer |
|
SMA |
BioGenex, MU128-UC |
Heat-induced epitope retrieval in a Black & Decker steamer for 30 min at 95°C in EDTA buffer (pH 9.0) |
1:300 |
BioCare polymer-HRP (mouse MC541H) |
|
Desmin |
Cell Marque, 243M-16 |
Heat-induced epitope retrieval in a Black & Decker steamer for 30 min at 95°C in target retrieval solution, pH 6 (Dako, S1699) |
1:100 |
BioCare polymer-HRP (mouse MC541H) |
|
Laminin |
BioGenex, PU078-UP |
10-min incubation in 0.2% protease enzyme diluted in PBS at 37°C |
1:50 |
Rabbit-on-canine HRP polymer (Biocare, RC542H) |
SMA = smooth muscle actin.
4.1.6 Sources and manufacturers
Cite the name of the manufacturer in parentheses at the appropriate location within the text—addresses of suppliers are not required. Use generic names of drugs in the text, with the brand name in parentheses. No need to use ® or ™. No need for Corp., Inc., Ltd., Co., Pty., GmbH, AG, etc. There is no need for catalog numbers, unless similar products have the same name. For free online analyses or software, add the URL in parentheses.
Examples:
The Microflex LT MALDI-TOF mass spectrometer (Bruker Daltonics) was used in our study. The mass spectrometer was calibrated for molecular weights with a range of 3,637–16,952 Da prior to sample testing using the bacterial test standard (Bruker Daltonics), as per the manufacturer’s recommendations.
The amplicons were then purified, sequenced, and further confirmed by BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi).
4.2 Detailed manuscript layout
4.2.1 Title page
Manuscript page 1: See example below. Title of paper, authors’ first name, middle initial, last name (e.g., John D. Doe); name and location of each author’s institution(s); name, postal address, and email of the corresponding author; and a short running head not to exceed 60 characters (including spaces; when published, the first author’s last name + et al. will appear on even-numbered pages and the short running head on odd-numbered pages). Please set up the title and authors as in a recent issue of JVDI. Use sentence case for the Article title.
Example:
Manuscript page 1: See example below. Title of paper, authors’ first name, middle initial, last name (e.g., John D. Doe); name and location of each author’s institution(s); name, postal address, and email of the corresponding author; and a short running head not to exceed 60 characters (including spaces; when published, the first author’s last name + et al. will appear on even-numbered pages and the short running head on odd-numbered pages). Please set up the title and authors as in a recent issue of JVDI. Use sentence case for the Article title.
Example
|
Paeniclostridium (Clostridium) sordellii–associated enterocolitis in 7 horses Akinyi C. Nyaoke, Mauricio A. Navarro, Karina Fresneda, Santiago S. Diab, Janet Moore, Dena Lyras, Milena Awad, Francisco A. Uzal1 California Animal Health and Food Safety Laboratory System, University of California–Davis, San Bernardino (Nyaoke, Navarro, Fresneda, Moore, Uzal) and Davis (Diab) branches, CA, USA; Infection and Immunity Program, Monash Biomedicine Discovery Institute and Department of Microbiology, Monash University, Clayton, Victoria, Australia (Lyras, Awad). 1Corresponding author: Francisco A. Uzal, California Animal Health and Food Safety, University of California–Davis, 105 W Central Ave, San Bernardino, CA 92408, USA. [email protected] Running head: Paeniclostridium sordellii enterocolitis in horses |
4.2.2 Abstract
Manuscript page 2: Limit the abstract to 250 words or fewer (Full Scientific Reports) or 200 words or fewer (Brief Reports) and write as a single paragraph. It must be factual and concise, yet complete enough to stand alone without reference to the text. State conclusions clearly: “Results are discussed.” is unacceptable. Do not use reference citations in the Abstract.
4.2.3 Keywords
Manuscript page 2: Using terms from the medical subject headings (MeSH) list of the U.S. National Library of Medicine, provide an alphabetical list of keywords or phrases not to exceed 80 characters (including spaces). Note keywords directly below, and on the same page as, the Abstract. Spell out non-standard abbreviations at first use. During online submission, SAGE Track limits the number of keywords that can be submitted; this has no bearing on the list included in your manuscript.
4.2.4 Manuscript sections
- Reviews: Abstract (≤250 words) and Keywords; Introduction; and appropriate section headings and subheadings chosen by the author.
- Full Scientific Reports: Abstract (≤250 words) and Keywords; Introduction (no header needed); Materials and methods; Results; Discussion; Acknowledgments (optional); Declaration of conflicting interests; Funding; References; Tables (optional); and Figure legends (if applicable). Do not use subheads in Introduction and Do not summarize your findings (In conclusion…) at the end of the manuscript (avoid a repeat of the Abstract).
- Brief Reports: Abstract (≤200 words) and Keywords; body of manuscript (<2,000 words; no section or subheadings in the main text); Acknowledgments (optional); Declaration of conflicting interests; Funding; References (n ≤ 20); Tables (optional); and Figure legends (if applicable). Do not summarize your findings (In conclusion…) at the end of the manuscript (avoid a repeat of the Abstract).
- Supplemental material is optional but welcomed (see section 6).
Acknowledgments
In the Acknowledgments section, list all contributors who do not meet the criteria for authorship. Examples of those who might be acknowledged include a person who provided purely technical help, writing assistance, or a department chair who provided only general support. Authors are to disclose whether they had any writing assistance and identify the entity that paid for this assistance.
Conflicting interests
Required section. A statement is required from all authors to be carried within the paginated pages of all published articles, under the heading “Declaration of conflicting interests”. If no conflicting interests exist, please use the following text:
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
When making a declaration, the disclosure information must be specific and include any financial relationship that authors of the article have with any sponsoring organization and the for-profit interests the organization represents, and with any for-profit product discussed or implied in the text of the article.
Any commercial or financial involvements that might represent an appearance of a conflict of interest need to be additionally disclosed in the cover letter accompanying your article to assist the Editor in evaluating whether sufficient disclosure has been made within the Declaration of Conflicting Interests provided in the article. For more information, please visit the SAGE Publication Ethics Policies.
Publication of papers dealing with a commercial product or laboratory test does not convey or imply an endorsement by JVDI or AAVLD.
Funding
Required section. Disclose any funding sources, as well as grant numbers. If no outside funding was used, please use the following text:
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
In the text, identify references with superscript numbers. Do not use the names of authors in the text.
Authors are responsible for the accuracy of all references.
Example:
Rarely, the nasal infection caused by P. insidiosum in sheep extends to the rhinopharyngeal region31; the submandibular and parotid lymph nodes are almost always involved.25,27,31
In the list of References, first arrange references alphabetically by the first author, then number them consecutively (see Appendix B).
Personal communications should be listed in the text [e.g., (Maxie MG, pers. comm., 2020 Apr 08)].
Tables
Tables should appear on separate pages following the References. Include a table title directly above the table. Table titles must be free-standing and self-explanatory. Number all tables consecutively with Arabic numerals and cite them consecutively in the text. Insert citations in the text following relevant results (Table 1, Fig. 3), rather than devoting a sentence to referencing the content of the Table or Figure (“… are presented in Table 1.”).
Layout: left-align columns; top-justify cells; sentence case for column heads and entries; set title and table footnotes as text outside of the table; no bolding other than to show abnormal values. Spell out all abbreviations in alphabetical order using table footnotes (e.g., M = male; ND = not determined). Indicate references to items other than abbreviations using the following sequential symbols (*, †, ‡, §, ¦, #, ¶, **).
See example below, as well as the examples in section 4.1.5.
Example:
Table 1. Results of a selective enrichment broth with real-time PCR assay for Salmonella, expressed as cycle threshold (Ct) values, for recent clinical isolates spiked into an environmental matrix from a poultry farm.
|
Ct value for indicated dilution, cfu/mL |
|||||
|
Origin |
Serogroup |
Serovar |
10,000 |
100 |
10 |
|
Bovine feces |
NG |
Cerro |
13.4 |
13.5 |
14.2 |
|
|
K |
Cerro |
15.0 |
14.6 |
15.3 |
|
Bovine small intestine |
B |
4,5,12:i:- |
16.0 |
15.0 |
14.2 |
|
Avian drag swab |
C1 |
Thompson |
16.7 |
18.6 |
17.8 |
|
Reptile feces |
NG |
Arizona III_53:z10:z35 |
14.0 |
14.9 |
17.4 |
|
Bovine lung |
D1 |
Dublin |
29.0 |
35.1* |
* |
NG = non-groupable.
* All samples were culture-positive except those marked by an asterisk.
Figure legends
Every figure must have a corresponding legend. Legends appear on a separate page following the References or Tables. Identify animal species, organ, or tissue, and describe the figure in a continuous phrase or sentence. Indicate stains on light and electron micrographs within the figure legend. See also Image magnification in section 5. Related series of figures may be grouped into plates (see Panel image alignment in section 5).
Example of single-panel image legend:
Figure 1. Granulomatous hepatitis in a llama. H&E. Bar = 20 μm.
Examples of multiple-panel image (plate) legend:
Figure 1. Photomicrograph of the hemisected right globe. A. Microscopic abnormalities outlined in B–F. B. The sclera (box b in A) merges with dermis-like stroma containing hair follicles and sebaceous glands with overlying pigmented squamous epithelium. Bar = 200 µm. C. Deep stroma in the anterior segment (box c in A) resembles uveal stroma, containing large cells resembling bladder cells and covered by cuboidal ciliated cells. Bar = 50 µm. D. Islands of cartilage (box d in A) merge with this stroma. Bar = 200 µm. E. Papillae resembling ciliary body plicae (box e in A) extend from the interior surface of the globe. Bar = 200 µm. F. Pigmented vascularized tissue resembling choroid (box f in A) covers the interior surface of the sclera, with an overlying layer of retinal pigment epithelium.
Bar = 200 µm.
Figures 1–5. Herpesviral meningoencephalitis in cattle. Figure 1. A 7-mo-old male calf was separated from the herd, was somnolent, and stood still for long periods with a low head carriage.
Figure 2. Affected 6-mo-old female calf with serous ocular discharge and drooling. Figure 3. Same calf in Fig. 1 with yellow, mucopurulent nasal discharge. (Image from Pesq Vet Bras 26:123–132, 2006; with permission). Figure 4. A 7-mo-old female calf from the same outbreak in Figs. 1 and 3 was ataxic and had a wide-based stance. Figure 5. Same calf in Figs. 1 and 3 with recumbency.
5 Figures
Failure to submit figures in the required format and resolution will result in immediate rejection. See our Best Practices (slides 19–29) for tips on creating and modifying figures. Figures must NOT be embedded in the text document but submitted separately. Please submit figures UN-flattened to allow for editing if needed. Do not exceed the maximum file size of 5 MB per figure.
Acceptable formats are .tiff and .jpg
- minimum acceptable resolution is 300 dpi (300 pixels/inch, 11 pixels/cm) for photographs, pathology images
- minimum acceptable resolution is 600 dpi (600 pixels/inch, 22 pixels/cm) for line art (e.g., graphs, charts, maps)
- For .tiff files, use LZW When saving a file from any photo-editing program, you should be given the option of compressing the file. LZW compression will reduce the size of your file, increase the speed of upload and download times, and will not affect your image quality negatively.
Sizing and configuration
- Illustrations with figure panels should be reproduced in 2 columns (180 mm wide) or 1 column (90 mm wide).
- Preferred panels are up to 2 figures horizontal and up to 3 figures Other configurations may be accepted at the discretion of the Editor (please inquire before submission). See panel image configuration below.
Group multiple color figures into a composite figure (the JVDI Images Editor will add thin white lines to separate the figure panels), with the individual panels clearly identified (i.e., 1, 2, 3, etc. for different lesions or organs; or A, B, C, etc. for closely related images, e.g., various IHCs of similar sections) in 14-point Arial, located in the bottom left corner of each image. Save figures as separate files with the figure number (Fig 1, Fig 2, Figs 3–5, etc.) as the file name (do not use figure numbers and/or titles as part of the image).
Insets should be placed in the bottom right corner, separated from the main panel (the JVDI Images Editor will add thin white lines to separate the inset from the main figure; see Example 3 below).
Submit pathology images in color.
- In gross pathology images, grass, surgery drapes, bodily fluids, gloves, , must be digitally removed and replaced with a more-or-less uniform background (see Example 3 below).
- In gross anatomic photographs, locate the head to the right of the
- Scale bars, case numbers, or other identification legends are not
- In photomicrographs, orient the surface of the skin or mucous membrane at the top of the Photomicrographs must not contain photographic or tissue artifacts, and the images must be evenly lit with white backgrounds (places where there is no tissue). Limit image modification or enhancement to that obtainable by ordinary photographic techniques or to adjustment of white balance, brightness, or contrast by photo-editing software. If these parameters in your figures are unacceptable, the Images editor may make suitable adjustments.
- There is no requirement to indicate image size in the figure
- Scale bars may be used if deemed necessary by the authors but are not required. Scale bars may be black or white (depending on the background) and are located on the bottom right corner of the image. Please do not place the μm size on the figure or the bar; instead, add the size to the figure legend (e.g., Bar = 20 μm).
- Electron micrographs must have a scale bar in the
Panel image configuration
Example 1. Example 2.
Reprinted from JVDI 2025;37(2). Reprinted from JVDI 2025;37(2).
Example 3.
Reprinted from JVDI 2025;37(3).
Example 4.
Reprinted from JVDI 2025;37(2).
Example 5.
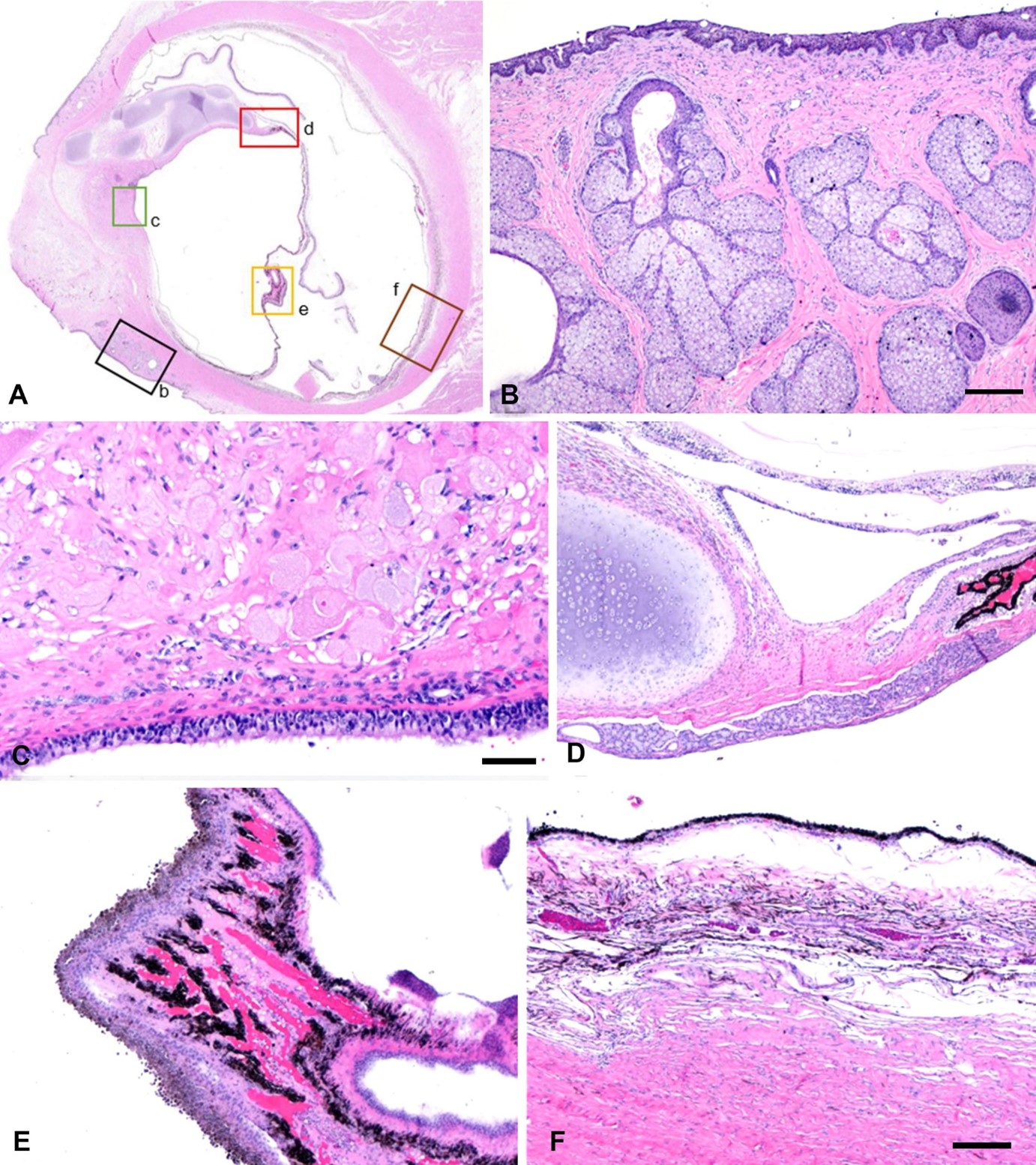
Reprinted from JVDI 2025;37(4).
Example 6.

Reprinted from JVDI 2025;37(3).
Line art guidelines
See our Best Practices for tips on creating and modifying line art. All text in figures (Arial, in Sentence case) must be of sufficient point size to be readable in print.
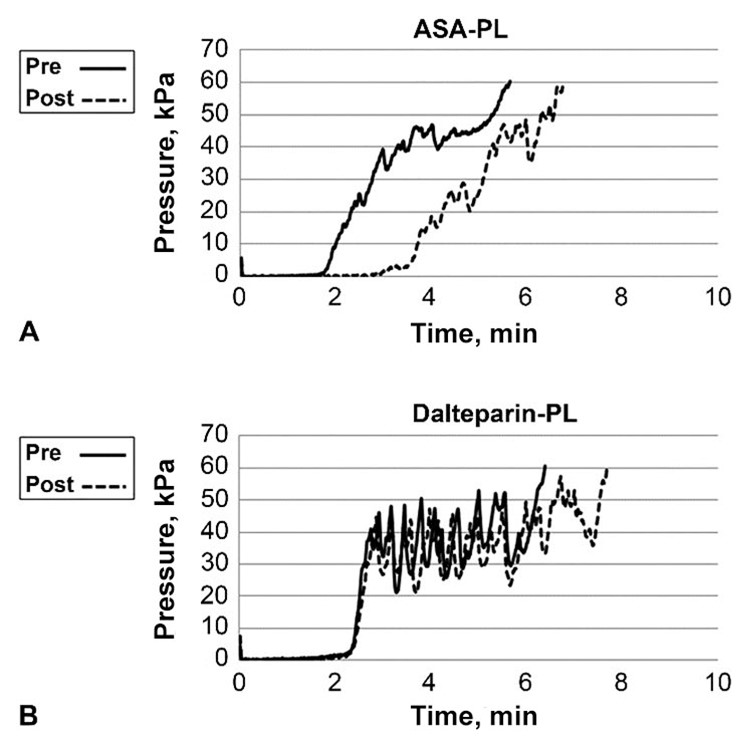
The y-axis of Figures must begin at 0, and the axis can show a break; otherwise, results may be exaggerated inappropriately.
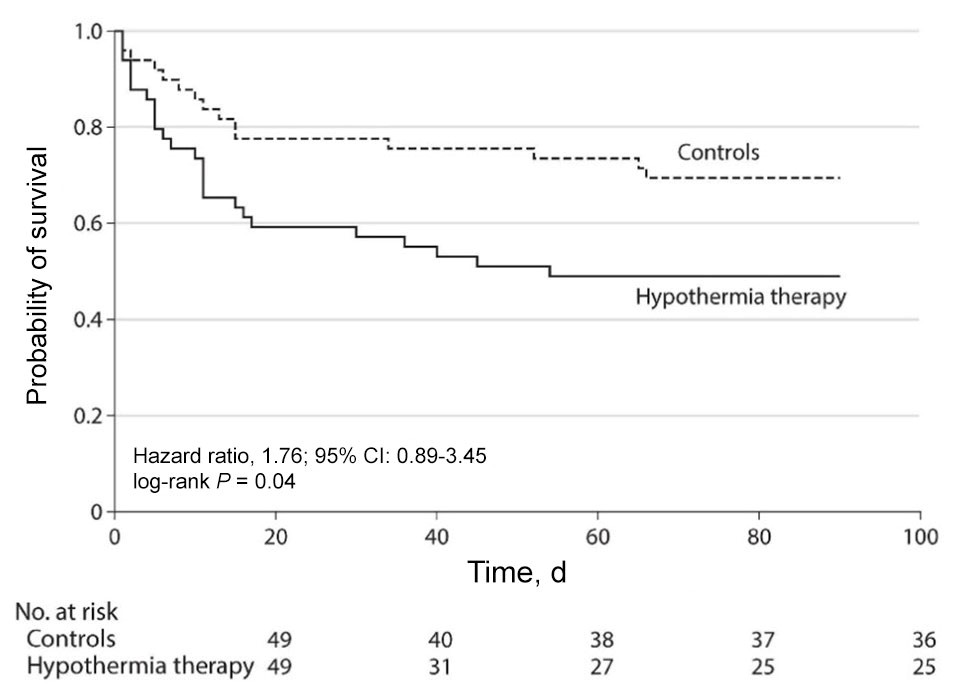
Kaplan–Meier survival curves
A table of the number of animals at risk is required for each graph (AMA Manual of Style, 11th ed.).
6 Supplemental material
JVDI, through SAGE Publications, hosts supplemental materials online, alongside the full text of articles. Supplemental materials are generally files that were used to create the research (e.g., datasets, dendrograms, etc.) or additional pieces to the article that are not included in the fee version (e.g., audio/video material, tables, figures, or raw data).
Submit text and tables as .doc or .csv files; submit figures as .tiff or .jpg files. Do not submit supplemental material as a PDF.
For .csv files, supply a title and description for all research data items. These can be included in a separate Word document upon submission.
Please add the following text to the main document following the Funding section. All supplemental data must also be cited in the text (see example below).
Supplemental material
Supplemental material for this article is available online.
Example:
Laboratories were asked to provide a list of 10 most common bacterial isolates on which they performed antimicrobial susceptibility testing across all animal species (Table 2; Suppl. Table 1).
7 New manuscript submission
- Submit all parts of the manuscript via SAGE Track.
- If you have previously submitted or reviewed a manuscript using SAGE Track, use your existing User ID and Password to log in. NOTE: If you are not registered, click “Create an Account” and follow the on-screen instructions.
—Your ORCID iD will not be published in your article unless your existing iD is linked with your ScholarOne account in SAGE Track. To edit your ScholarOne account, log in to SAGE Track and click E-mail/Name from the drop-down menu under your name, which appears in the upper right corner of your screen.
—If you do not have an ORCID iD, you are provided with a link to create an account.

2. Click on Author Center, then Manuscripts with Decisions.
4. As shown in the screenshot below, if you are resubmitting a previously rejected manuscript, you must answer YES and enter your previous manuscript number during the submission If you do not know your previously assigned manuscript number, please contact the Editorial Office
NOTE: For new and revised manuscript submissions, our submission site generates a PDF of your files (main document, figures, suppl. material) at the end of your submission. You are required to check the PDF to verify that all of your files have been uploaded successfully and that the files are in the correct order of appearance. The PDF will display your figures at a smaller size and lower resolution to help the PDF load faster. Your original files are not altered during this process. The Images editor evaluates your figures from your original files, and not from the pdf.
8 Revised manuscript submission
- Use your User ID and Password to log in to SAGE Track.
- Click on Author Center, then Manuscripts with
- Locate the manuscript you wish to revise and click Create a
- You will be prompted to respond to the comments made by the Editor and/or Important: Address each comment and detail your changes in the space provided or by uploading a Word document with the reviewers’ comments listed and your response to each.
- Follow the submission process, providing information when prompted.
All submissions are reviewed by the EIC and may be rejected based on content and/or formatting. Suitable manuscripts are assigned by the EIC to a Section editor, who will oversee the peer-review process. Once peer-reviewed, the revised version of the manuscript is forwarded to the EIC for review and approval (further revision is often required). In the meantime, photo images are reviewed by the Images editor. The EIC then sends the approved manuscript to one of the Managing editors for copyediting, where it will be held in the category “Awaiting Editor Decision” in SAGE Track until edited. After final approval of the copyediting by the EIC and author, the manuscript files are forwarded to SAGE Publications.
9 Page proofs
The Corresponding Author will receive an email from the SAGE Production Editor with a link to access the typeset page within 2–4 wk from time of acceptance.
10 Help
If you experience any problems during the online submission process, please consult the SAGE Track Author Guide and FAQs, which provides detailed submission instructions. Holly Farrell or Barbara Vann, co-Managing editors ([email protected], [email protected], [email protected]) are also available for assistance. Francisco Uzal, the Images editor ([email protected]), is available for specific questions about figures.
APPENDIX A: PCR guidelines—suggested dossier or publication information
|
Sample |
|
Source of known positive and negative samples |
|
Description of sample types and species |
|
Processing procedure |
|
Description of procedure and length of time, if frozen |
|
Name of fixative and length of time, if fixed |
|
Sample storage conditions and duration (especially for FFPE samples) |
|
Nucleic acid extraction |
|
Procedure and/or instrumentation |
|
Name of kit and details of any modifications |
|
Source of additional reagents used |
|
Specify internal control |
|
PCR target information |
|
Name of target gene |
|
Sequence accession number |
|
Coordinates according to accession number and length of amplicon |
|
In silico specificity screen (BLAST), secondary structure analysis, sequence alignment; all are not necessary to show but author should indicate it has been done |
|
Primer sequences, unless evaluating a commercial kit with proprietary information |
|
Primer/probe source |
|
PCR amplification |
|
Primer/probe concentration given in µm in final reaction |
|
Master mix reagents (buffer, enzyme, Mg, dNTPs)* |
|
Additives* |
|
Reaction volume |
|
Complete thermocycling parameters |
|
Manufacturer of qPCR instrument |
|
Controls included: NTC, internal control, amplification control |
|
Analysis methods and software version |
|
Analytical performance characteristics |
|
Limit of detection (LOD) |
|
Efficiency |
|
Linearity |
|
Range |
|
Repeatability |
|
Diagnostic performance characteristics |
Table adapted from MIQE (Bustin SA, et al. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 2009;55:611–622).
* Include vendor/manufacturer.
APPENDIX B: Reference formats
Journal articles
Same author (alphabetical order; date order)
Dubey JP. A review of toxoplasmosis in pigs. Vet Parasitol 1986;19:181–223.
Dubey JP. The history of Toxoplasma gondii—the first 100 years. J Eukaryot Microbiol 2008;55:467–475.
Dubey JP, et al. All about Toxoplasma gondii infections in pigs: 2009–2020. Vet Parasitol 2020;288:109185.
Dubey JP, Jones JL. Toxoplasma gondii infection in humans and animals in the United States. Int J Parasitol 2008;38:1257–1278.
More than 2 authors
Solanki AK, et al. Clostridium perfringens beta toxin DNA prime-protein boost elicits enhanced protective immune response in mice. Appl Microbiol Biotechnol 2017;101:5699–5708.
In a language other than English
Trindade AB, et al. Lipossarcoma em caturrita (Myiopsitta monachus) [Liposarcoma in a caturrita (Myiopsitta monachus)]. Cienc Anim Bras 2010;11:971–976. Portuguese.
In a supplement
Schultz RD, et al. Age and long-term protective immunity in dogs and cats. J Comp Pathol 2010;142(Suppl 1):S102–S108.
Forthcoming
Clift SJ, et al. Polyclonal antibody–based immunohistochemical detection of intra-leukocytic Theileria
parasites in roan and sable antelopes. J Vet Diagn Invest 2021. Accepted.
Epub ahead of print
Songsungthong W, et al. In silico–derived Actinobacillus equuli–specific DNA markers and development of associated PCR assays. J Vet Diagn Invest. Published online 2025 Oct 8. doi:10.1177/10406387251382186
Errata/Corrigendum
Ong CB, Herdt TH, Fitzgerald SD. Hyperplastic goiter in two adult dairy cows. J Vet Diagn Invest 2014;26:810–814. Corrigendum: J Vet Diagn Invest 2018;30:654.
Books
Chapter in a book
Craig LE, et al. Bones and joints. In: Maxie MG, ed. Jubb, Kennedy, and Palmer’s Pathology of Domestic Animals. 6th ed. Vol. 1. Elsevier, 2016:16–163.
Book in a series
Koestner A, et al. Histological Classification of Tumors of the Nervous System of Domestic Animals. 2nd series. (Vol. 5, WHO International Classification of Tumors of Domestic Animals). Armed Forces Institute of Pathology, 1999.
Other media
CLSI document
Clinical and Laboratory Standards Institute (CLSI). Defining, establishing, and verifying reference intervals in the clinical laboratory; approved guideline. 3rd ed. CLSI, 2010. CLSI document EP28-A3C3.
Conference proceedings (add page numbers and URL if available)
Reed KF, et al. Milk urea nitrogen: precision, accuracy, and individual animal variability. In: Proc 82nd Cornell Nutr Conf; Ithaca, NY; 2020:192–200. https://hdl.handle.net/1813/72919
Stoltsz WH, Dunsterville MT. In vitro establishment and cultivation of a Cytauxzoon sp. (Theileria sp.) from a sable antelope (Hippotragus niger, Harris 1838). J S Afr Vet Assoc 1992;63:182. Parasitol Soc South Africa abstract. https://journals.co.za/doi/pdf/10.10520/AJA00382809_2977
Dissertation/thesis (add URL if available)
Roguskie JM. The role of Pseudomonas aeruginosa 1244 pilin glycan in virulence. Master thesis.
Duquesne University, 2005. https://dsc.duq.edu/etd/1119
Zehr ESN. Relatedness of Haemophilus parasuis strains and their proteins’ possible roles as virulence factors. PhD dissertation. Iowa State University, 2008.
European Commission
European Commission. Commission Decision (2002/657/EC) of 12 August 2002 implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results. Off J Eur Commun 2002;L221:8–36.
Newspaper article
Gaul G. When geography influences treatment options. Washington Post (Maryland Ed.). 2005 Jul 24:Sect. A:12 (col. 1).
Online database
Elliott-Smith E, Haig SM. Piping plover. 2020 March 4. Version 1.0. Birds of the World. Cornell Laboratory of Ornithology. https://birdsoftheworld.org/bow/species/pipplo/cur/introduction
Protein [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; [1988]. Accession XP_545130.3, cystatin-A [Canis lupus familiaris]; [cited 2024 Mar 05]. https://www.ncbi.nlm.nih.gov/protein/XP_545130.3https://www.ncbi.nlm.nih.gov/search/all
/?term=XP_545130.3
Patent
Pagedas AC, inventor; ABS Inc., assignee. Flexible endoscopic grasping and cutting device and positioning tool assembly. United States patent US 20020103498. 2002 Aug 1.
Preprint
Hamer SA, et al. Natural SARS-CoV-2 infections, including virus isolation, among serially tested cats and dogs in households with confirmed human COVID-19 cases in Texas, USA. bioRxiv 2020:2020.12.08.416339.
Stenger B. Extraction and PCR for animal samples using the ThermoFisher Scientific TaqPath COVID-19 Combo Kit. Protocols.io. dx.doi.org/10.17504/protocols.io.36wgqjq5ovk5/v1
Scientific report (add report number and URL if available)
Barker B, Degenhardt L. Accidental drug-induced deaths in Australia 1997–2001. University of New South Wales, National Drug and Alcohol Research Centre, 2003.
Kock TJ, et al. Evaluation of water temperature effects on adult sockeye salmon (Oncorhynchus nerka) behavior in the Yakima River, Washington, 2019. U.S. Geological Survey, 2020. Open-file report 2020-1033.
Simpson VR. Health status of otters in southern and south west England 1996–2003. Environment Agency, 2007. Science report SC010064/SR1. https://assets.publishing.service.gov.uk/media
/5a756a9640f0b6360e473f33/scho0307bmkl-e-e.pdf
URL
American Veterinary Medical Association. SNOMED, HL7, LOINC the official informatics standards for veterinary medicine. 2002 May 15. https://www.avma.org/javma-news/2002-06-01/snomed-hl7-loinc-official-informatics-standards-veterinary-medicine
Centers for Disease Control and Prevention. Multistate outbreak of human Salmonella Cotham and Salmonella Kisarawe infections linked to contact with pet bearded dragons (final update). 2014 Aug 20. https://archive.cdc.gov/#/details?url=https://www.cdc.gov/salmonella/cotham-04-14/index.html
Institute of Medicine of the National Academies. Key capabilities of an electronic health record system.
Letter report. The National Academies Press, 2003. https://www.nap.edu/catalog/10781/key-capabilities-of-an-electronic-health-record-system-letter-report
If publication date is not available, add cite date.
U.S. Department of Agriculture. U.S. cattle production. [cited 2020 April 9]. https://www.ers.usda.gov
/topics/animal-products/cattle-beef/sector-at-a-glance/#production
APPENDIX C: Abbreviations that may be used without expansion
|
Units - abbreviated when used with a numeral; otherwise, spell out |
||
|
base pair, kilobase pair |
bp, kbp |
|
|
beats/min |
bpm |
|
|
colony-forming unit |
cfu |
|
|
plaque-forming unit |
pfu |
|
|
days post-infection |
dpi |
|
|
kilogram |
kg |
|
|
gram |
g |
|
|
milligram |
mg |
|
|
microgram |
μg |
|
|
picogram |
pg |
|
|
nanogram |
ng |
|
|
international units |
IU |
|
|
liter |
L |
|
|
deciliter |
dL |
|
|
milliliter |
mL |
|
|
microliter |
μL |
|
|
femtoliter |
fL |
|
|
meter |
m |
|
|
centimeter |
cm |
|
|
millimeter |
mm |
|
|
micrometer |
µm |
|
|
microkatal |
μkat |
|
|
mole |
mol |
|
|
millimole |
mmol |
|
|
millimole/L, millimolar |
mM |
|
|
micromole |
μmol |
|
|
picomole |
pmol |
|
|
parts per million; -billion; -trillion |
ppm, ppb, ppt |
|
|
relative centrifugal force, RCF |
× g |
|
|
vol/vol, wt/vol, wt/wt |
v/v, w/v, w/w |
|
|
Time |
|
|
|
day(s) |
d |
|
|
week(s) |
wk |
|
|
month(s) |
mo |
|
|
year(s) |
y |
|
|
millisecond(s) |
ms |
|
|
second(s) |
s |
|
|
minute(s) |
min |
|
|
hour(s) |
h |
|
|
morning, afternoon |
AM, PM |
|
|
Routes of administration |
||
|
intradermal |
ID |
|
|
intramuscular |
IM |
|
|
intraperitoneal |
IP |
|
|
intravenous |
IV |
|
|
per os |
PO |
|
|
subcutaneous |
SC |
|
|
Frequency of administration |
|
|
|
every 8 h (not tid) |
q8h |
|
|
every 12 h (not bid) |
q12h |
|
|
every 24 h (not sid) |
q24h |
|
|
Statistical terms |
|
|
|
analysis of variance |
ANOVA |
|
|
confidence interval |
CI |
|
|
coefficient of variation |
CV |
|
|
number |
n |
|
|
probability |
p |
|
|
sample mean |
x̄ |
|
|
standard deviation, standard error standard error of mean |
SD, SE SEM |
|
|
Student t-test |
t-test |
|
|
Other common abbreviations |
|
|
|
adenosine mono-, di-, tri- phosphates; cyclic AMP |
AMP, ADP, ATP, cAMP |
|
|
adrenocorticotropic hormone |
ACTH |
|
|
American Type Culture Collection |
ATCC |
|
|
APHIS |
|
|
|
Bacillus Calmette–Guérin |
BCG |
|
|
Basic Local Alignment Search Tool: https://blast.ncbi.nlm.nih.gov/ |
BLAST |
|
|
cluster of differentiation |
CD |
|
|
central nervous system |
CNS |
|
|
cerebrospinal fluid |
CSF |
|
|
The Commonwealth Scientific and Industrial Research Organisation |
CSIRO |
|
|
complete blood count |
CBC |
|
|
cycle threshold |
Ct |
|
|
degrees of freedom |
df |
|
|
deoxynucleotide triphosphates |
dNTPs |
|
|
deoxyribonucleic acid, ribonucleic acid: c = complementary; |
DNA, RNA: cDNA, mRNA, rDNA, tRNA |
|
|
dichlorodiphenyltrichloroethane |
DDT |
|
|
electrocardiogram, electrocardiographic |
ECG |
|
|
enzyme-linked immunosorbent assay |
ELISA |
|
|
ethylenediaminetetraacetic acid |
EDTA |
|
|
for example [exampli gratia] |
e.g., |
|
|
gauge |
G |
|
|
hematocrit |
Hct |
|
|
hematoxylin and eosin |
H&E |
|
|
hemoglobin |
Hb |
|
|
high-efficiency particulate air filter |
HEPA filter |
|
|
high-power field(s) |
hpf |
|
|
hydrogen peroxide |
H2O2 |
|
|
id est [that is] |
i.e., |
|
|
immunoglobulin A, D, etc. |
Ig, IgA, IgD, IgE, IgG, IgM |
|
|
low-power field |
lpf |
|
|
Madin–Darby bovine kidney cells |
MDBK cells |
|
|
Madin–Darby canine kidney cells |
MDCK cells |
|
|
mass-to-charge ratio |
m/z |
|
|
median lethal dose |
LD50 |
|
|
median tissue culture infective dose |
TCID50 |
|
|
minimum inhibitory concentration, MIC50 = 50% of isolates are inhibited |
MIC, MIC50 |
|
|
magnetic resonance imaging |
MRI |
|
|
nonsteroidal anti-inflammatory drug |
NSAID |
|
|
open reading frame |
ORF |
|
|
packed cell volume |
PCV |
|
|
personal communication |
pers. comm. |
|
|
phosphate-buffered saline |
PBS |
|
|
polyacrylamide gel electrophoresis |
PAGE |
|
|
polymerase chain reaction |
PCR |
|
|
quality assurance |
QA |
|
|
quality control |
QC |
|
|
quantitative PCR |
qPCR |
|
|
red blood cell |
RBC |
|
|
reference interval |
RI |
|
|
revolutions per minute |
rpm |
|
|
ribosomal RNA |
rRNA |
|
|
sodium dodecyl sulfate |
SDS |
|
|
species - single, plural, new sp. subspecies - with organism name |
sp., spp., sp. nov., subsp. |
|
|
trisaminomethane |
Tris |
|
|
unpublished data |
unpub. data |
|
|
United Kingdom; United States - abbreviated when used as an adjective; otherwise, spell out |
U.S. |
|
|
United States Department of Agriculture |
USDA |
|
|
ultraviolet |
UV |
|
|
versus —abbreviated when inside parentheses; otherwise, spell out |
vs. |
|
|
white blood cell |
WBC |
|



 Facebook
Facebook X
X LinkedIn
LinkedIn YouTube
YouTube RSS feed
RSS feed Email alerts
Email alerts