A Step-by-Step Approach for Assessing Human Skin Irritation Without Animal Testing for Quasi-Drugs and Cosmetic Products
Abstract
Introduction:
Materials and Methods:
Results:
Conclusion:
Introduction
Materials and Methods
Physicochemical properties and the ingredient category based on the existing information on the test substance

Predictability of the tests
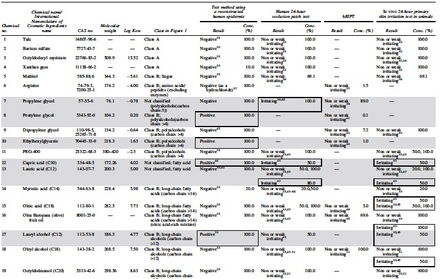 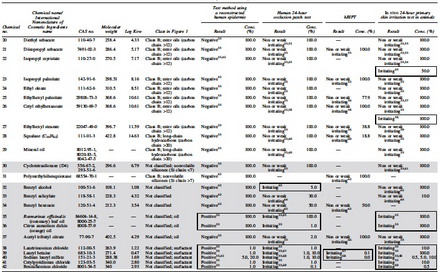 |
Results
Physicochemical properties and ingredient category of the test substance based on existing information
Class A: Component characteristics
Class B: Ingredient category
| “Harmless ingredients,” Class B23–25 | |
|---|---|
| Category | Explanation of each ingredients category21–24 |
| Sugars | Compounds with one carbonyl or aldehyde group and two or more hydroxyl groups with three to six carbon atoms (monosaccharides) and compounds in which two or more of them are bonded by glycoside bonds (disaccharides, oligosaccharides, polysaccharides). In addition, a compound (sugar alcohol) in which the carbonyl group of a monosaccharide is reduced to become a hydroxyl group. |
| Amino acids/peptides (excluding enzymes) | Amino acids are compounds that have a carboxyl group and an amino group in the molecule, and are produced by a synthetic method or a fermentation method. A peptide is a peptide in which two or more amino acids are bound by a peptide bond, and is synthesized by partial hydrolysis of a protein or binding of amino acids. |
| Polyalcohols (carbon chain >4) | A compound that has two or more hydroxyl groups in the molecule. Those with two hydroxyl groups are called dihydric alcohols, and those with three hydroxyl groups are called trihydric alcohols. We limited the number of carbon chains to exclude propylene glycol, because irritation reaction was confirmed in the 24-hour occlusion patch test. |
| Plant extracts (residue on evaporation <5%) | A plant-derived product, which is an ingredient (extract, juice, water) having its origin in a plant (including algae), and the extracts from part or all of the plant, dried products from plants or plant extracts, juices of plants, an aqueous layer obtained by steam distillation of a part or whole extract of a plant. From the information on the origin of plants, it is limited to plants that are not used in medicines and pesticides and have no reports of health hazards such as carcinogenicity, and are limited to plant extracts with an evaporation residue of 5% or less. |
| Long-chain fatty acids (carbon chain >14) | Fatty acid is a general term for carboxylic acids that have chain hydrocarbons. Long-chain fatty acids have an even number of carbon atoms of 14 or more, the hydrocarbon group is linear, and the unsaturated bond present in the hydrocarbon group is the cis type. |
| Long-chain alcohols (carbon chain >12) | A general term for alcohols with a large number of carbon atoms, which refers to aliphatic alcohols having 12 or more carbon atoms. |
| Ester oils (carbon chain >12) | A compound produced by losing water from acid and alcohol. Esters of fatty acids and glycerin exist as animal fats and vegetable oils. |
| Wax | Chemically, it is an ester of a long-chain fatty acid and a long-chain monohydric alcohol and is in a solid state. |
| Triglycerides | Three molecules of fatty acid are ester bonded to one molecule of glycerol. There are an extremely large number of molecular species depending on the type of fatty acid and the bonding position on glycerol, and each exhibits different physicochemical properties. |
| Long-chain hydrocarbons (carbon chain >20) | An organic compound consisting of carbon and hydrogen that exhibits a liquid state at a temperature of 15°C and 1 atm. The number of carbon chains was limited to exclude pristane, which was found to be irritating in the 24-hour occlusion patch test. |
| Nonvolatile silicones (Si chain >7) | With a siloxane bond as the main structure, there are various compounds from low molecular weight compounds (low viscosity volatile liquids) to high molecular weight compounds (resins). Since it cannot be ruled out that volatile silicones may be irritating, volatile silicones such as D4, D5, and D6 were excluded. |
| Test methods | na | Accuracy | False negative ratio | False positive ratio |
|---|---|---|---|---|
| Animal testing | 36 | 69.4% (25/36) | 43.8% (7/16) | 20.0% (4/20) |
| Human patch test | 37 | 97.3% (36/37) | 0% (0/11) | 3.8% (1/26) |
| Human repeated insult patch test | 19 | 84.2% (16/19) | 0% (2/2) | 17.6% (3/17) |
Predictability of the tests
Reliability of the RhE test for predicting skin irritation in the human patch test
| Test methods | na | Accuracy | False negative ratio | False positive ratio |
|---|---|---|---|---|
| Animal testing | 36 | 80.6% (29/36) | 43.8% (7/16) | 0% (0/20) |
| Human patch test | 37 | 89.2% (33/37) | 27.3% (3/11) | 3.8% (1/26) |
| Human repeated insult patch test | 19 | 89.5% (17/19) | 0% (2/2) | 11.8% (2/17) |
Reliability of the RhE test for predicting skin irritation in animal testing
Reliability of the RhE test for predicting skin irritation in a human cumulative skin irritation test
A step-by-step approach for skin irritation testing
Discussion
Conclusions
Acknowledgments
Author Disclosure Statement
Funding Information
Disclaimer
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
Cite
Cite
Cite
Download to reference manager
If you have citation software installed, you can download citation data to the citation manager of your choice
Information, rights and permissions
Information
Published In
Keywords
Authors
Metrics and citations
Metrics
Journals metrics
This article was published in Applied In Vitro Toxicology.
View All Journal MetricsPublication usage*
Total views and downloads: 211
*Publication usage tracking started in December 2016
Publications citing this one
Receive email alerts when this publication is cited
Web of Science: 0
Crossref: 2
- Development of a Vitamin C Derivative Serum for Pre-Aging Skin: Raw Material Screening, Stability and Clinical Evaluation
- Irritation Test of Bawang Dayak (Eleutherine Bulbosa (Mill.) Urb.) Loose Powder for Acne Vulgaris
Figures and tables
Figures & Media
Tables
View Options
View options
PDF/EPUB
View PDF/EPUBAccess options
If you have access to journal content via a personal subscription, university, library, employer or society, select from the options below:
loading institutional access options
Alternatively, view purchase options below:
Purchase 24 hour online access to view and download content.
Access journal content via a DeepDyve subscription or find out more about this option.





