Harmonisation of large-scale, heterogeneous individual participant adverse event data from randomised trials of statin therapy
Abstract
Background:
Methods:
Results:
Conclusion:
Introduction
Methods
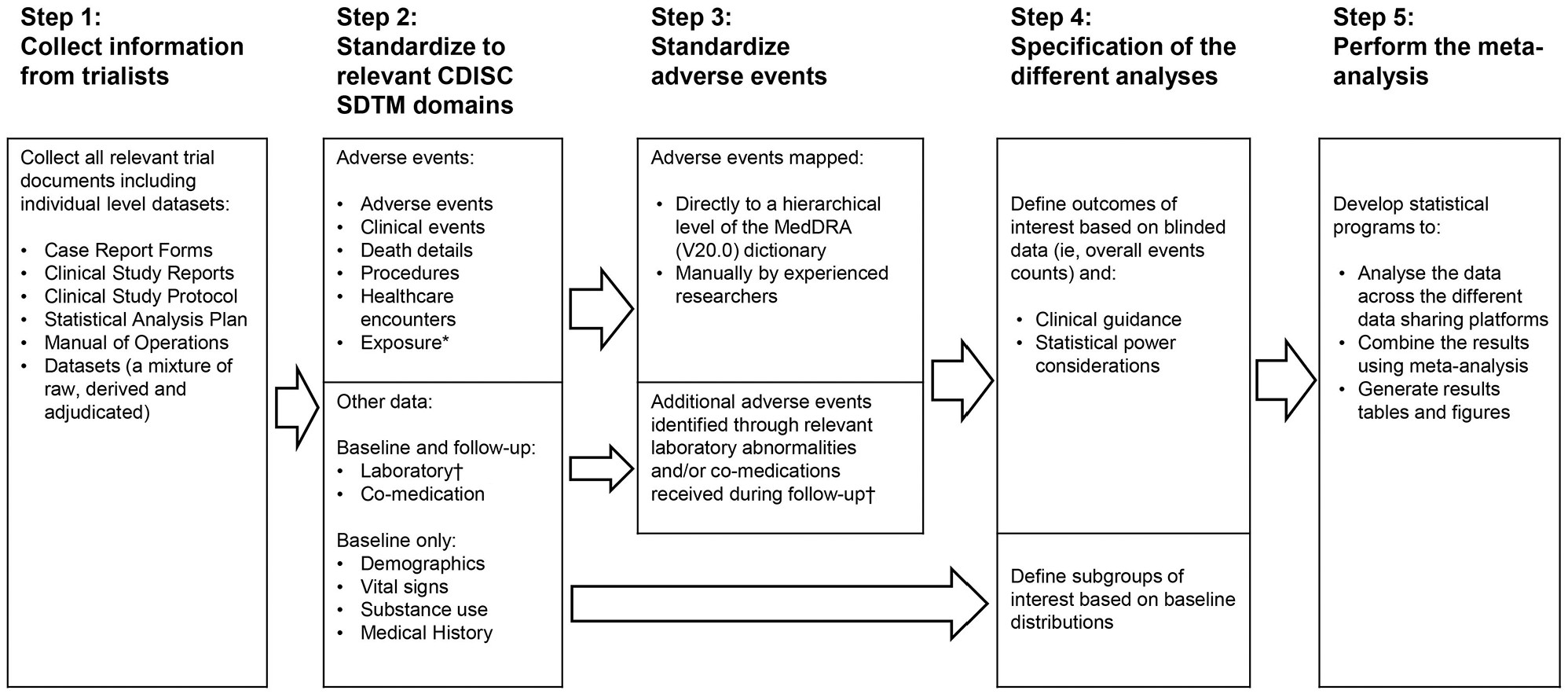
Development of a common data format based on Clinical Data Interchange Standards Consortium Study Data Tabulation Model
| SDTM domain | Description |
|---|---|
| Adverse events | Adverse events which may be reported either as free text or as terms created from a coding system (e.g. MedDRA or International Classification of Diseases). |
| Clinical events | Trial-specific non-fatal events which were not classified as adverse events. |
| Co-medication | Concomitant and prior medications/therapies used by the participant. Examples are the concomitant medications/therapies given on an as needed basis and the usual background medications/therapies given for a condition. |
| Death details | Any fatal events regardless of cause. |
| Demographics | The parent domain for all other domains including a set of essential standard variables that describe each participant in the clinical study. Examples are the participants’ characteristics at baseline (e.g. treatment allocation, age and race). |
| Exposure | Reasons for stopping (or adjusting) study treatment. For our purposes, only adverse events resulting in stopping (or adjusting) study treatment were sought. |
| Healthcare encounters | Inpatient and outpatient healthcare events (e.g. hospitalisations, nursing home stay, rehabilitation facility stays and ambulatory surgery). |
| Laboratory test results | Laboratory test findings including, but not limited to haematology, clinical chemistry and urinalysis data. For our purposes, only the following laboratory tests were captured: (1) alanine aminotransferase, (2) aspartate aminotransferase, (3) creatine kinase, (4) glucose in blood, (6) high-density lipoprotein cholesterol, (7) low-density lipoprotein cholesterol, (8) triglycerides, (9) total cholesterol, (10) creatinine concentration (serum), (11) estimated glomerular filtration rate, (12) glycosylated haemoglobin and (13) platelets (thrombocytes). |
| Medical history | The participant’s prior history at the start of the trial (e.g. general medical history, gynaecological history and primary diagnosis). |
| Procedures | Details describing a participant’s therapeutic and diagnostic procedures (e.g. coronary artery bypass graft, cataract surgery or hip replacement). |
| Substance use | Substance use information that may be used to assess the efficacy and/or safety of therapies that look to mitigate the effects of chronic substance use. For our purposes, only smoking status and alcohol consumption at baseline were collected. |
| Subject visit | Information about the timing of participant visits that is otherwise spread over domains that include the visit variables. For our purposes, the subject visit domain was created only when imputation methods were required for timing variables. |
| Vital signs | Measurements including but not limited to blood pressure, temperature, respiration, body surface area, body mass index, height and weight. For our purposes, only the following measurements were captured: (1) systolic blood pressure, (2) diastolic blood pressure, (3) heart rate, (4) body mass index, (5) height, (6) weight and (7) waist circumference. |
Use of a common event-coding dictionary based on MedDRA
| MedDRA hierarchical level | Description | Total number of available terms |
|---|---|---|
| Lower level term | A reflection of how observations might be reported in practice, for example, a verbatim term. Each lower level term is linked to only one preferred term. | 77,248 |
| Preferred term | A single descriptor such as a symptom, sign, disease diagnosis, investigation or procedure. Each preferred term has at least one lower level term (itself) as well as synonyms and lexical variants (e.g. abbreviations, different word order). | 22,499 |
| Higher level term | A group of preferred terms that are related based on anatomy, pathology, physiology, aetiology or function. | 1553 |
| Higher level group term | Related higher level terms are linked to higher level group terms based on anatomy, pathology, physiology, aetiology, or function. | 326 |
| System organ class | The final level of the hierarchy where higher level group terms are grouped by aetiology (e.g. infections and infestations), manifestation site (e.g. gastrointestinal disorders) or purpose (e.g. surgical and medical procedures). There is also a system organ class to contain issues pertaining to products and one to contain social circumstances. | 27 |
Organisation of trial data prior to analysis
Results
Summary of received data
| Study | Year ofpublicationof primaryresults | Number of participants | Treatmentcomparison(mg/day) | Medianfollow-up(years) | Meta-dataCRF (F)CSP (P)CSR (R) | Total number of | Adverse event terms | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Datasets | Records | Variables | Main medicalterminologya | Total | Unique | ||||||
| Statin vs placebo, double-blinded trials | |||||||||||
| 4S | 1994 | 4444 | S20-40 vs placebo | 5.4 | FP | 5 | 116,502 | 147 | MedDRA* | 33,661 | 1390 |
| WOSCOPS | 1995 | 6595 | P40 vs placebo | 4.8 | FP | 23 | 671,759 | 509 | BMS ICD-9 | 78,673 | 7978 |
| CARE | 1996 | 4159 | P40 vs placebo | 4.9 | FPR | 9 | 1,849,516 | 275 | BMS ICD-9 | 91,635 | 753 |
| AFCAPS/TexCAPS | 1998 | 6605 | L20-40 vs placebo | 5.0 | P | 5 | 995,156 | 97 | MedDRA* | 8143 | 770 |
| LIPID | 1998 | 9014 | P40 vs placebo | 5.9 | FPR | 28 | 1,347,182 | 592 | ICD-9 | 57,636 | 7075 |
| LIPS | 2002 | 1677 | F80 vs placebo | 4.0 | FPR | 61 | 537,144 | 1145 | ICD-10 | 8867 | 2392 |
| HPS | 2002 | 20,536 | S40 vs placebo | 5.2 | FP | 28 | 8,152,101 | 392 | MedDRA 14 | 76,986 | 2378 |
| PROSPER | 2002 | 5804 | P40 vs placebo | 3.3 | FP | 11 | 628,189 | 485 | Modified ICD-9 | 131,598 | 848 |
| ASCOT-LLA | 2003 | 10,240 | A10 vs placebo | 3.3 | FP | 10 | 710,266 | 466 | ICD-9 | 42,294 | 1896 |
| ALERT | 2003 | 2102 | F40 vs placebo | 5.5 | FPR | 79 | 1,069,747 | 5654 | ICD-9/MedDRA 5.1 | 27,114 | 3589 |
| CARDS | 2004 | 2838 | A10 vs placebo | 4.2 | FP | 17 | 988,712 | 522 | MedDRA 11 | 26,485 | 1890 |
| 4D | 2005 | 1255 | A20 vs placebo | 2.7 | FP | 21 | 302,442 | 632 | MedDRA 11 | 6758 | 461 |
| ASPEN | 2006 | 2410 | A10 vs placebo | 4.0 | FP | 18 | 472,820 | 379 | MedDRA 11 | 20,323 | 2861 |
| SPARCL | 2006 | 4731 | A80 vs placebo | 4.9 | FP | 14 | 900,713 | 512 | MedDRA 11 | 38,811 | 3539 |
| CORONA | 2007 | 4984 | R10 vs placebo | 2.8 | FPR | 62 | 2,155,519 | 1716 | MedDRA 10 | 28,899 | 3450 |
| JUPITER | 2008 | 16,714 | R20 vs placebo | 1.9 | FPR | 33 | 1,540,095 | 2428 | MedDRA 11.1 | 73,526 | 5731 |
| GISSI-HF | 2008 | 4574 | R10 vs placebo | 3.9 | F | 10 | 208,489 | 169 | MedDRA 15.1 | 28,701 | 537 |
| AURORA | 2009 | 2555 | R10 vs placebo | 3.9 | FPR | 42 | 724,490 | 1273 | MedDRA 11.1 | 39,512 | 4013 |
| HOPE-3 | 2016 | 12,705 | R10 vs placebo | 5.6 | FP | 170 | 1,415,309 | 6213 | MedDRA 17.1 | 70,651 | 7631 |
| Subtotal | 123,942 | 646 | 24,786,151 | 23,606 | 890,273 | 40,221 | |||||
| Statin versus other control/usual care, open-label trials | |||||||||||
| POST-CABG | 1997 | 1351 | L40-80 vs L2.5-5 | 4.3 | FP | 83 | 227,740 | 2821 | None | 37,164 | 650 |
| GISSI-P | 2000 | 4271 | P20 vs no treatment | 1.9 | FP | 1 | 4271 | 795 | ICD-9 | 2777 | 228 |
| ALLHAT-LLT | 2002 | 10,355 | P40 vs no treatment | 4.7 | FP | 10 | 381,498 | 103 | ICD-9/ICD-10 | 9932 | 115 |
| ALLIANCE | 2004 | 2442 | A10-80 vs usual care | 4.5 | FP | 15 | 136,557 | 344 | MedDRA 11 | 3717 | 1334 |
| Subtotal | 18,419 | 109 | 750,066 | 4063 | 53,590 | 2311 | |||||
| More vs less statin, double-blinded trials | |||||||||||
| PROVE-IT | 2004 | 4,162 | A80 vs P40 | 2.1 | FP | 24 | 603,289 | 1,161 | ICD-9 | 25,600 | 1551 |
| A to Z | 2004 | 4,497 | S40 then S80vs placebo then S20 | 2.0 | FP | 10 | 386,787 | 405 | MedDRA* | 4615 | 561 |
| TNT | 2005 | 10,001 | A80 vs A10 | 5.0 | FP | 20 | 2,676,152 | 624 | MedDRA 11 | 93,583 | 3735 |
| SEARCH | 2010 | 12,064 | S80 vs S20 | 7.0 | FP | 25 | 7,341,699 | 344 | MedDRA 14 | 61,379 | 1303 |
| Subtotal | 30,724 | 79 | 11,007,927 | 2534 | 185,177 | 6210 | |||||
| More vs less statin, open-label trials | |||||||||||
| IDEAL | 2005 | 8888 | A40-80 vs S20-40 | 5 | FP | 11 | 1,999,354 | 292 | MedDRA 13 | 59,129 | 2281 |
| Subtotal | 8888 | 11 | 1,999,354 | 292 | 59,129 | 2281 | |||||
| Total | 181,973 | 845 | 38,543,498 | 30,495 | 1,188,169 | 45,230 | |||||
Data harmonisation
Mapping of adverse events to MedDRA
| Type and MedDRA level of coding | Adverse events, N (%) | |
|---|---|---|
| Totala | Unique termsb | |
| Automatically coded events | ||
| Lower level term | 779,449 (65.6%) | 11,965 (26.5%) |
| Higher level term | 6216 (0.5%) | 659 (1.5%) |
| Higher level group term | 820 (<0.1%) | 1 (<0.1%) |
| Subtotal | 786,485 (66.2%) | 12,625 (27.9%) |
| Manually coded events | ||
| Lower level term | 391,338 (32.9%) | 32,458 (71.8%) |
| Higher level term | 7571 (0.6%) | 116 (0.3%) |
| Higher level group term | 2775 (0.2%) | 31 (<0.1%) |
| Subtotal | 401,684 (33.8%) | 32,605 (72.1%) |
| Total | 1,188,169 (100%) | 45,230 (100%) |
Discussion
Acknowledgments
Declaration of conflicting interests
Funding
Footnotes
Data availability statement
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
Cite
Cite
Cite
Download to reference manager
If you have citation software installed, you can download citation data to the citation manager of your choice
Information, rights and permissions
Information
Published In
Keywords
Data availability statement
Authors
Author contributions
Metrics and citations
Metrics
Publication usage*
Total views and downloads: 2892
*Publication usage tracking started in December 2016
Altmetric
See the impact this article is making through the number of times it’s been read, and the Altmetric Score.
Learn more about the Altmetric Scores
Publications citing this one
Receive email alerts when this publication is cited
Web of Science: 5 view articles Opens in new tab
Crossref: 5
- Assessment of adverse effects attributed to statin therapy in product labels: a meta-analysis of double-blind randomised controlled trials
- Safety and Efficacy of Achieving Very Low LDL Cholesterol Concentrations with PCSK9 Inhibitors
- Effects of statin therapy on diagnoses of new-onset diabetes and worsening glycaemia in large-scale randomised blinded statin trials: an individual participant data meta-analysis
- A Pragmatic Method to Integrate Data From Preexisting Cohort Studies Using the Clinical Data Interchange Standards Consortium (CDISC) Study Data Tabulation Model: Case Study
- Endothelial Function in Dyslipidemia: Roles of LDL-Cholesterol, HDL-Cholesterol and Triglycerides
Figures and tables
Figures & Media
Tables
View Options
View options
PDF/EPUB
View PDF/EPUBAccess options
If you have access to journal content via a personal subscription, university, library, employer or society, select from the options below:
loading institutional access options
SCT members can access this journal content using society membership credentials.
SCT members can access this journal content using society membership credentials.
Alternatively, view purchase options below:
Purchase 24 hour online access to view and download content.
Access journal content via a DeepDyve subscription or find out more about this option.




